Research Article - (2018) Volume 8, Issue 4
Shafeeque K* and Hashim KM
Uwin Life Sciences, Malappuram, India
Corresponding Author:
Shafeeque K
Uwin Life Sciences, Malappuram, India
Tel: +919567859670
E-mail: shafeequekalody@gmail.com
Received Date: July 25, 2018; Accepted Date: Aug 07, 2018; Published Date: Aug 25, 2018
Citation: Shafeeque K, Hashim KM (2018) Comparative Anti-Angiogenesis Study between Allicin Nanoparticle and Normal Allicin from Garlic (Allium Sativum Linn). Eur Exp Biol Vol. 8 No. 4:27. doi:10.21767/2248-9215.100068
Copyright: © 2018 Shafeeque K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
The current research article deals with anti–angiogenesis study between allicin nano particle and normal allicin from Garlic (Allium sativum L). Since last year’s Garlic has been used for treating various purposes and its consumption is said to reduce health risk and its organosulfur components effectively block cancer cell growth. Allicin is the major biologically active organo sulfur compound present in garlic. In-vitro anti angiogenesis assay was carried out and significant activity of Nano allicin found for the cancer treatment. Cinnamaldehyde was used as positive control and found that allicin nano particle process more antiangiogenesis activity than the normal allicin.
Keywords
Angiogenesis; Nanoparticle; Allicin; Organosulfur; Allium sativum
Introduction
Nanotechnology is current trend in research field, using small size of materials with wide range of application [1]. The word Nano particle means, very small size of particle. In nanotechnology treatment of atom, molecules or compounds to produce nanoparticle with different properties .The size and structure of nanoparticle is similar to most biological molecules, therefore Nano particle can be useful for in research. Application of nanoparticle with science led to development of medical research, drug delivery system, diagnosis devices and analytical tools etc. There are many types of nanoparticles: organic, inorganic, nanocrystals, nanotubes, polymeric structures such as dendrimers, etc. They are used primarily in research and drug delivery systems [2]. Nowadays design and development of herbal nano medicine has become a frontier in the nano formulation research field. Recent research proved that herbal nano medicine very useful for treatment of cardiovascular disease, cancer therapy, proliferative diseases, and diabetes, anti –osteoporosis and like [3]. Application of nanotechnology has enabled the delivering drug to specific target using Nano devices [4]. Target drug delivery, effective dose, and specific size are leads to reduce the side effects of drug with decrease in treatment cost and consumption. It enables on maximum bio availability, this can be achieved by application Nano technology [5,6].
Advantage of using Nanotechnology for medical purpose is that tiny devices and can possibly inserted inside the body. This type of Nano devices very efficient than typical drug delivery [7]. Targeted delivery system or nano particle sized medicine can be improved pharmacokinetic profile and bioavailability of drug [8,9]. The pharmacokinetics and pharmacodynamics of nano medicine is highly variable from person to person [10]. Nano medicines have desirable properties, it can be used for to become drug better drug delivery. Drug with poor solubility can be solved by drug delivery system where both hydrophobic and hydrophilic environment exist [11]. Side effects and tissue damage may prevent by drug delivery system through effective dose and size [12]. However a lot of Research is still on ongoing to understand pros and cons of nano particle drug delivery system [13].
Since earliest time, Garlic has been used as medicinal purposes throughout the world, mainly to prevent coronary thrombosis, atherosclerosis and stroke and it used for the treatment of infections and disease .it can reduce serum cholesterol level and inhibit cancer cell growth [14]. Ancient people used garlic (Allium sativum L) for centuries for curing various illness such as heart disease, arthritis, pulmonary complaints, diarrhea and worm infection countries like in India, China, Greek and Egypt [15]. A recent research proved that both organ sulfur compounds from garlic and aqueous garlic extract might influence its anticancer activity by inducing programmed cell death. Therapeutic advantage of organosulfur compounds from garlic across the world and role that it was examined to play in prevention and treatment of various illness. Among organosulfur compounds from garlic, Allicin is the major biologically active organosulfur compounds present in the garlic. Allicin is important compounds exist in crushed garlic with much advantage [16]. Action allinase on allin from garlic leads to formation of allicin and gets processed to very quickly into other compounds such diallyl sulfide, diallyl disulfide, diallyl trisulfide, ajoene, S-allyl mercaptocysteine, S-allyl cysteine and vinyl dithiines [17]. Organosulfur compounds found in Garlic, Allicin (3-prop-2-enylsulfinylsulfanylprop-1-ene) Possess anti microbial activity. Recent studies have proved that allicin keep anti thrombotic, anti oxidant and anti cancer effects [18].
The process of the formation new blood vessels from preexisting cells referred to as angiogenesis, it is vital process for many physiological mechanisms such as wound healing, inflammation, embryonic development and organogenesis. In the last decade, role of the angiogenesis in metastasis and malignant tumor has been much studied. A therapeutic strategy to suppress angiogenesis has been examined on experimental tumor models with successful outcomes. Based on these experimental data, some anti-angiogenic agents have been tested in clinical trials. Also it plays a key role in tumor growth and metastasis as well as other physiological and pathological processes such as embryonic development, chronic inflammation, and wound healing. The phenomenon of angiogenesis controlled by various factors such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), tumor necrosis factor (TNF) and Interleukin-8(IL-8) [19]. Vasculogenesis is process of development of first vessel in embryo followed by angiogenesis Responsible for most of the blood vessel growth. [20]. Important role of angiogenesis on malignant tumor metastasis first described by Judah Folkman [21]. Phenomenon of angiogenesis must need steady an availability of amino acids, nucleic acids, carbohydrates, oxygen, and growth factors. Malignant tumor instigate angiogenesis, then only growth of new blood vessels from preexisting ones so as to obtain these nutrients while process of angiogenesis vasodilatation and increased permeability of preexisting vessels occurs followed by activated endothelial cells release proteases to degrade matrix afterward endothelial cells proliferate and migrate. This process needed to Differentiation and maturation of blood vessels present in the body. Since last decades people have been using different kind of antiangiogenic drugs. Era of antiangiogenic drugs started with Judah Folkman’s hypothesis in early 1970. In 1989 Dr. Napolene Ferra first identified and isolated vascular endothelial growth factor (VEGF). In 2004 Food drug administration approved first antiangiogenic drug to treat colorectal cancer named avastin. Different antiangiogenic drugs varying in targets such as neovasculator and preexting vasculator [22-26].
It has been found that anti-angiogenic drugs might potentially interfere with many physiological process such as blood pressure, wound healing, fetal development, reproduction and increase chance of clot in arteries that may lead to stroke or heart attack [27-30]. A typical example for anti-angiogenic therapy, hyper tension is one of the side effects of systemic inhibition of VEGF signaling. Under normal physiological condition VEGF is responsible to release vasodilator nitric oxide (NO) in blood vessels wall by upregulating endothelial nitric oxide synthase (eNOS) and prostacyclin (PGI2), resulting in vasodilation, followed by the activation of the mitogen-activated protein kinase (MAPK) and phosphotidylinositiol 3kinase (P13K) downstream pathways. Therefore by suppression of VEGF, which will lead to vasoconstriction and elevated blood pressure [31-35]. It is cleared that the inhibition of VEGF, that disturb angiogenesis process or delay wound healing process [36]. Nevertheless, cancer treatment with anti-angiogenic drugs that could results for improved delivery of drugs [37-44].
Materials and Methods
Alcohol extraction and isolation of allicin
We took 500 g fresh garlic, peeled gloves and put in hot air oven at 45°C to become crispy form for 3 days. Using mortar and pestle crushed to get coarse powder. The shade powdered sampled 35 gm were taken with accuracy weight. It was then extracted using alcohol using reflux method for three times. This solution completely transferred to conical flask through Whatman Filter Paper contained 100 g Silica gel SiO2.To get pure allicin it is need to purify faster than it can degrade into other chemical species. We used 3:1 proportion Petroleum ether and isopropanol in column chromatography.
Extract purification
Each aqueous extract was filtered and diluted to 50 ml with water. The diluted extract (10 ml) was saturated with sodium chloride and subsequently extracted with 10 ml of dichloromethane (3 times). Each fraction were dried over anhydrous sodium sulfate and desiccated by means of rotaryevaporation.
UV spectroscopic analysis
A preliminary analysis of standard allicin using a UV-visible detector with spectral range 200-600 nm was made under the chromatographic conditions. The UV spectrum was recorded for each purified extract in order to ascertain the presence of pure allicin by confirming the Lambda Max of the compound. Further confirmation of the identity of the compound proceeded to the LCMS analysis with integrated library.
LC MS analysis of allicin
The LCMS analysis were carried out using Shimadzu LC 10 AVP, Japan with a L-7110 pump and a column of C18 with dimensions 250 × 4.6 mm i.d., 5 μm particle size. The column was operated in isocratic mode (50:50 MeOH:H2O) at a flow rate of 0.5 ml/ min. The mobile phase which we maintained in an isocratic mode was 50:50 ratio of MeOH and H2O. The column temperature was adjusted to 25°C. The PDA detector showed a correct resolution at 254 NM and selected for the entire scanning for the development of the mass spectrum of the compound so as to confirm its identity. The library confirmation was done using Metwin 2.0 version with the mass fragmentation matching [45-47].
Nano particle preparation
In this study Emulsification Solvent diffusion method used chitosan nano particle preparation .Chitosan natural polymer obtained from Chitin by deacetylation. After cellulose chitin is the second most abundant polysaccharide in nature, derived from crustacean shells such as lobster, crab and shrimps. Chitosan natural polymer is also found in some yeast and fungi. Monomer of Chitosan combined by (1,4) glycosidic linkage, forming along chain linear polymer. Nano particle formation depends upon molecular weight and degree of deacetylation. Chitosan such as Parenteral administration, peroral administration, Non-viral gene delivery vector and ocular administration. Emulsification solvent diffusion method based on partial miscibility of an organic solvent with water. In this technique injection of an organic phase into chitosan solution, containing a stabilizing agent, an oil water emulsion is obtained under mechanical stirring followed by high pressure homogenization. As result of the diffusion of organic solvent into water, polymer precipitation occurs that leads to formation of Nano particle. Allicin is hydrophobic compound and showed high percentage of drug entrapment. Nevertheless some drawbacks associated with this method, include harsh process condition and the high shear forces using chitosan nano particle preparation [48].
In vitro anti- angiogenesis assay
Different animal model were used, to understand process of angiogenesis [49-52]. The Fertilized egg obtained from chicken farm Palakkad of Kerala State, India. In this study we took 4 egg, head part of egg broke with surgical blade and 2 micro gram of nano particle allicin, normal allicin and positive control Cinnamaldehyde added through surgical syringe to each egg. After that broken part of egg sealed, and put it incubator with negative control were maintained at 37°C for 17 days (Figures 1 and 2).
Figure 1: Allicin structure (A) and 3D Conformer (B).
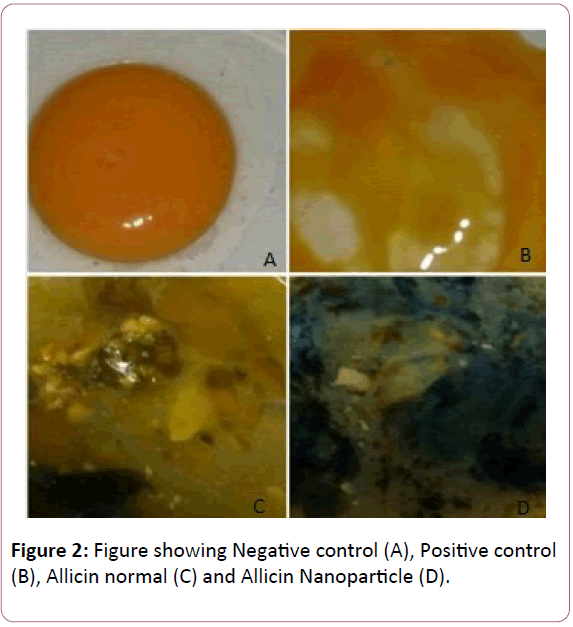
Figure 2: Figure showing Negative control (A), Positive control (B), Allicin normal (C) and Allicin Nanoparticle (D).
Results and Discussion
The above picture shows that the allicin in nano particle is having significant activity even when compared to that of the positive control. The normal particle showed a moderate antiangio- genesis activity.
Conclusion
From the present investigation we found high antiangiogenesis activity of the allicin nanoparticle at 2 mg. Thus we conclude that the allicin nanoparticle is very much potential in treating the angiogenesis. Further studies like in-vivo and chemical studies using the Nano particle of allicin may leads to the development of a new drug from the natural resource and will be a turning point the entire pharma industry.