Keywords
PCI; Vascular closure; Claims data; Expenditures
Introduction
In 2013 over 350,000 people died of cardiovascular diseases in Germany making it the leading cause of death [1]. To diagnose and treat cardiovascular diseases often a femoral cardiac catheterization like a coronary angiography (diagnosis) or a percutaneous coronary intervention (PCI) is performed. In 2008 around 845,000 diagnostics and 304,000 therapeutic catheterizations were performed in Germany [2].
These kinds of procedures leave an access puncture site in the femoral artery that can be closed by either manual compression (MC) or by using a Vascular Closing Device (VCD). MC was the standard method used since the beginning of femoral catheterization. VCDs were developed to achieve a shorter time to hemostasis and to shorten the time that the patient must lie still and therefore enhance patient comfort. Furthermore, they are expected to cause less access site complications. Combining these positive aspects should then result in cost savings surpassing the costs of the closure device itself.
However, some studies found differing results concerning complication rates. On the one hand, VCDs are associated with significantly shorter time to hemostasis, and ambulation, but no significant difference in the complication rate [3,4]. On the other hand, Biancari et al. identified a significant increase in severe complications, such as groin infections, after deployment of a VCD [5]. In several cost effectiveness studies VCDs were shown to be more cost-effective than MC [6-10]. However, those studies were mostly single centered with only a small sample size, they only looked at one study-specific VCD and also only included highly selective populations. A recent study by Kerré et al. compared direct medical costs in an all-comers PCI population in a single center. They found a significant difference in medical costs between a group of patients with VCD and another group with MC [11]. But, so far, no study investigated therapeutic and diagnostic cardiac catheterizations separately in a wide population and for a bigger range of VCDs (Figure 1).
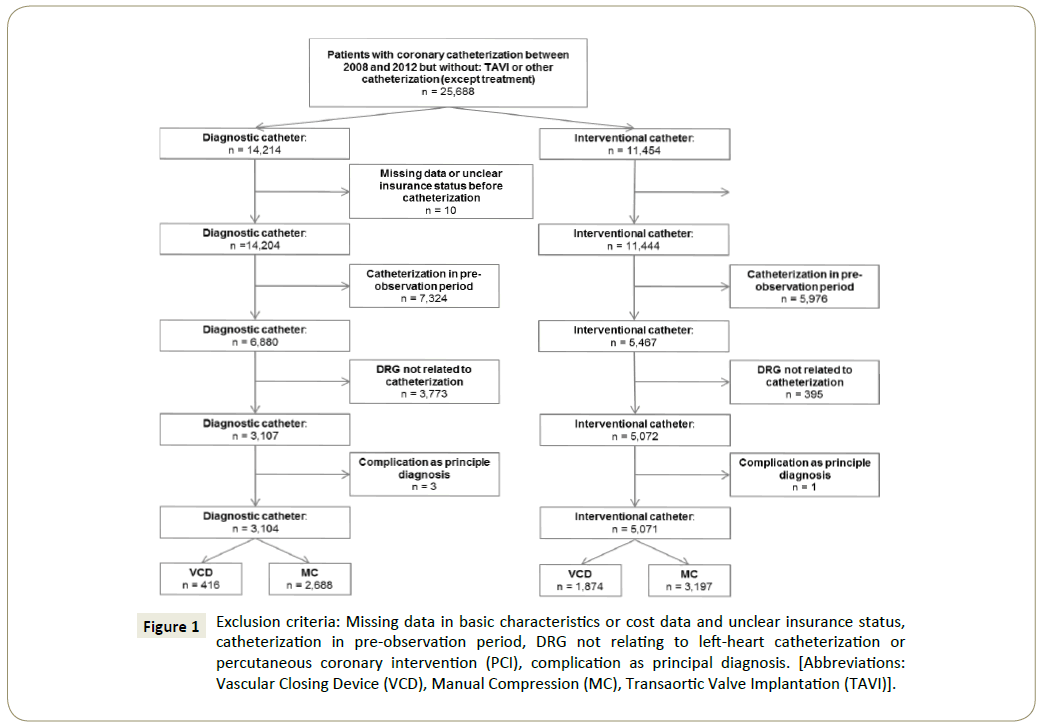
Figure 1: Exclusion criteria: Missing data in basic characteristics or cost data and unclear insurance status, catheterization in pre-observation period, DRG not relating to left-heart catheterization or percutaneous coronary intervention (PCI), complication as principal diagnosis. [Abbreviations: Vascular Closing Device (VCD), Manual Compression (MC), Transaortic Valve Implantation (TAVI)].
The main objective of our study was to:
(1) Compare hospital expenditures of using a VCD versus MC in both diagnostic and interventional catheterization, and (2) compare the probability of complications of VCD use versus MC in both diagnostic and interventional catheterizations, adopting a payer perspective.
Materials and Methods
In a retrospective cross-sectional claims data-based study we analysed a cohort of patients undergoing a coronary catheterization, who either received VCD or MC to achieve hemostasis. Data on socio-demographics, diagnostic and procedural codes, and inpatient healthcare expenditures came from health insurance data. The codes used in the analysis are given in the supporting information.
Study population
Data for the study came from AOK Baden Württemberg, the 5th biggest Statutory Health Insurance fund (SHI) in Germany currently insuring 3.9 million citizens [12]. In total around 70% of German citizens are insured in a SHI. Insurance contributions and healthcare services covered by the SHIs are regulated by law.
AOK Baden Württemberg approved the use of their data for the following analyses which were conducted according to national data protection laws. According to a self-disclosure of the ethics committee of the Bavarian State Medical Association, explicit ethics approval is categorically not required for this type of studies.
Data on diagnoses and resource utilization were provided on a patient basis for the years 2008 to 2011. We identified catheterizations using OPS-codes (German Version of the International Classification of Procedures in Medicine). Inclusion criterion was having either a PCI or a diagnostic cardiac catheterization in the years 2008 to 2011. In case of multiple catheterizations over the years only the first procedure was looked at. The diagnostic catheterization group included patients having one or more OPS coding of diagnostic catheterizations only during the same hospital stay, whereas the interventional catheterization group included patients with one or more OPS codes of an interventional procedure. Here additional codes for diagnostic catheterizations were allowed, since 94.5% of our cases with an interventional catheterization had also received a diagnostic one during the same hospital stay. Our initial data set included 25,688 catheterized individuals. To reach the final analysis set we subsequently excluded individuals according to following exclusion criteria (Figure 1):
(1) Undergoing a trans aortic valve implantation (TAVI) during the same hospital stay, as this is associated with a comparably large puncture site so that the relevant comparator would be vascular suture, rather than MC.
(2) Having any other catheterization, like right-heart catheterizations or percutaneous interventions on the vessels of the lung, during the same hospital stay, because complications could not be allocated to either the coronary catheterization or the other procedure.
(3) Observations with missing information about expenditures and basic characteristics or unclear insurance status before the hospital stay with the catheterization.
(4) Observations with an inpatient or outpatient catheterization in the first two quarters of 2008. This allowed us to use this period as a pre-observation period for determining a history of comorbidities.
(5) All subjects having a diagnosis related group (DRG) that was not related to cardiac catheterization to keep the emphasis on coronary catheterizations.
(6) Observations with a complication as principal diagnosis. Having one of the complications as a principal diagnosis could mean that the diagnosis was present before the procedure and not arising from it.
In total, we ended up with a sample size of 8,175 subjects, of which 3,104 (38%) had a diagnostic and 5,071 (62%) an interventional procedure. We analysed diagnostic and therapeutic catheterizations separately as patients undergoing the two types of catheterizations differ in their characteristics and comorbidity profiles. Also, the risk of complications in interventional procedures is higher than in diagnostic ones.
Measurement of VCD usage
The intervention of interest was the usage of a VCD to achieve hemostasis identified by the corresponding OPS code. We considered every observation of the study cohort that had this OPS code during the hospital stay with the femoral catheterization as having received a VCD. MC does not have an OPS code, therefore we assigned everybody else to the MC group.
Measurement of expenditures and complications
The main outcomes in our analysis were the expenditures for the hospital stay with the femoral catheterization, and whether a complication related to the method of puncture site closure occurred. Data on healthcare expenditures came from the dataset directly as total expenditures for the SHI for the hospital stay, inflated to the year 2011–the last year of the observation period-by using the German Consumer Price Index (CPI). Expenditures for readmissions relating to complications were automatically included due to case consolidation. We identified complications arising from either VCD or MC by using corresponding International Classification of Disease Codes (ICD). We agreed upon the following complications and ICDcodes identified by literature search and after consultation of a clinician and a coder: hematoma and bleeding [3], arteriovenous fistula (AVF) [13], pseudo aneurysm [13], retroperitoneal haemorrhage (RPH) [14], thrombosis [14], infection of access site [14] and other complications as indicated by a ICD code relating to a complication occurring from a procedure. The risk for complication was assessed binary by either having one of these complications or having no complications at all. To detect complications all ICD codes that were coded during the hospital stay and that were not a principle diagnosis were evaluated.
Measurement of confounder
We identified the following risk factors for complications by literature search: increasing age, female gender, obesity [15], hypertension [16], Peripheral Vascular Disease (PVD), Coronary Artery Disease (CAD) [17], diabetes mellitus [18], renal dysfunction [16] and the intake of antiplatelet and anticoagulant medication [17,19]. In the dataset, we identified these confounding variables through ICD and OPS codes from inpatient and outpatient records of the insurance company. For each patient and comorbidity, we only used those codes coded in the two quarters prior to the index hospital admission to address risk factors prevalent at the time of procedure and included them in the model as dummy variables. In the outpatient sector, we used confirmed diagnoses only.
Statistical analysis
Cohort characteristics were reported as means and proportions with p-values from t-tests for continuous variables (age) and from Chi-square tests for binary variables (gender and comorbidities). We used logistic regression to model the influence of VCDs on the risk of complications reported as odds ratios with p-values and confidence intervals.
As healthcare expenditures did not show a normal Gaussian distribution using linear regression (OLS) was not possible. Therefore, we used a Generalized Linear Model (GLM) with gamma distribution and log link function. This kind of model can handle data that are right-skewed and eliminates heteroscedasticity [20]. The derivate of the parameter estimates from gamma regression represent the additive effect (in healthcare expenditures) that a change in this variable would cause. To more easily interpret these results, healthcare expenditures were also presented as recycled predictions and adjusted differences with confidence intervals and p-values. Recycled predictions are used to understand the marginal effect of independent variables on a dependent variable. They are obtained from the gamma regression model by averaging predicted scores, after fixing the value of one independent variable (either VCD or MC), and using observed values on the remaining independent variables. The recycled predictions then provide adjusted means for both VCD and MC groups, where the difference is calculated [21].
Confidence intervals and p-values of the adjusted means and difference were based on non-parametric bootstrapping (1,000 bootstrap repetitions, percentile method).
The underlying null hypothesis for all models was that employing a VCD has no influence on healthcare expenditures per hospital stay or on the probability of complications. As we analysed the influence of VCD on these two effects in the same sample of patients, we used a Bonferroni-adjusted significance level of 2.5% to account for multiple testing. All regression models were adjusted for the confounders mentioned earlier. All analyses were performed using software of the SAS System for Microsoft, Version 9.3 copyright (c) 2002-2010 by SAS Institute Inc., Cary, NC, USA; tables and figures were created in Microsoft Excel and PowerPoint.
Sensitivity analysis
In a sensitivity analysis, we performed a comparison of healthcare expenditures and the probability of complications in a subgroup of patients with Diagnosis Related Groups (DRG) indicating catheterizations that are less complex or involve fewer and less severe comorbid conditions. We estimated healthcare expenditures and complications with the same methods as described above. However, for this analysis we could not perform logistic regression for the effect complications in diagnostic catheterization, as we had quasi-complete separation of data points, due to no complications in the VCD group.
Results
Sample characteristics
Sample characteristics of the patients with VCD and with MC divided according to type of catheterization are displayed in Table 1. All in all, we found 416 people that had a VCD employed in the diagnostic group and 1,874 in the interventional group amounting to a percentage of 13.4% and 37.0% respectively. Between the two groups of catheterizations differences were found in the proportion of males undergoing the procedure and in the prevalence of CAD, with a higher percentage of males in the interventional group and also a higher prevalence of CAD. Between the groups of VCD and MC in both types of catheterization gender and age, as well as most comorbidities were distributed almost equally between VCD use and MC. For PVD the prevalence was higher in the MC group.
|
|
Diagnostic catheterization |
Interventional catheterization |
| VCD(n=416) |
MC(n=2,688) |
P-value |
VCD(n=1,874) |
MC(n=3,197) |
P-value |
| Male gender as % (n) |
54.1(225) |
52.6(1,415) |
0.58 |
69.6(1,304) |
67.8(2,168) |
0.19 |
| Mean age in years, (SD) |
68.2(11.8) |
67.4(11.8) |
0.21 |
67.0(11.6) |
67.8(11.2) |
0.02 |
| Peripheral vascular disease as % (n) |
18.0(75) |
22.6(608) |
0.04 |
24.2(454) |
29.4(940) |
<0.0001 |
| Diabetes as % (n) |
32.9(137) |
36.2(927) |
0.20 |
38.4(720) |
40.2(1,285) |
0.21 |
| Renal disease as % (n) |
12.5(52) |
13.3(357) |
0.66 |
13.4(251) |
13.1(418) |
0.75 |
| Hypertension as % (n) |
75.5(314) |
77.1(2,073) |
0.46 |
72.6(1,360) |
75.0(2,399) |
0.05 |
| Obesity as % (n) |
22.1(92) |
22.8(612) |
0.77 |
20.0(374) |
21.1(673) |
0.35 |
| Coronary artery disease as % (n) |
58.7(244) |
60.3(1,620) |
0.53 |
79.5(1,489) |
80.1(2,562) |
0.56 |
Table 1: Means and proportions of sample characteristics according to the different types of catheters and Vascular Closing Devices(VCD) versus Manual Compression(MC).
Primary analysis
In diagnostic catheterization healthcare expenditures were almost equal for VCD (€2,657) and MC (€2,664) with an adjusted difference of €6 (CI= [-141.5, 121.7], p-value=0.92). In interventional catheterization healthcare expenditures between VCD (€4,380) and MC (€4,352) were comparable with an adjusted difference of €28 (CI= [-107.0, 150.2], p-value=0.62). Table 2 shows all factors influencing healthcare expenditures according to the gamma regression. Male gender, increasing age, the intake of antiplatelet and anticoagulant medication, as well as the comorbidities diabetes and renal failure had a significant influence on increasing healthcare expenditures in diagnostic catheterization. Patients with the comorbidity CAD had significantly lower healthcare expenditures. In interventional catheterization factors having a significant influence on healthcare expenditures were age, intake of antiplatelet medication, CAD, obesity, diabetes, PVD, and renal failure. Patients with CAD and obesity had significantly lower healthcare expenditures; all other significant factors resulted in higher healthcare expenditures.
|
|
Diagnostic catheterization |
Interventional catheterization |
| exp ß |
P-value |
exp ß |
P-value |
| Intercept |
1,709.40 |
<0.0001 |
3,710.79 |
<0.0001 |
| Employment of VCD |
1.00 |
0.95 |
1.01 |
0.57 |
| Male gender |
1.09 |
<0.0001 |
0.98 |
0.12 |
| Age group (in decades) |
1.06 |
<0.0001 |
1.02 |
<0.0001 |
| Intake of antiplatelets |
1.20 |
<0.0001 |
1.23 |
<0.0001 |
| Intake of anticoagulants |
1.18 |
<0.0001 |
1.04 |
0.16 |
| Hypertension |
0.98 |
0.29 |
0.99 |
0.69 |
| CAD |
0.88 |
<0.0001 |
0.90 |
<0.0001 |
| Obesity |
1.00 |
0.81 |
0.95 |
0.0003 |
| Diabetes |
1.05 |
0.002 |
1.05 |
0.0001 |
| PVD |
1.04 |
0.05 |
1.05 |
<0.0001 |
| Renal failure |
1.24 |
0.0001 |
1.17 |
<0.0001 |
VCD=Vascular Closing Device, CAD=Coronary Artery Disease, PVD=Peripheral Artery Disease
Table 2: Parameter estimates from gamma regression of costs by type of catheterization.
The probability of having a complication was significantly lower when a VCD was used in diagnostic catheterization with an odds ratio of 0.31 (p-value=0.02). In the interventional group, the odds ratio was 0.98 (p-value=0.90) indicating no difference. In both types of catheterization male gender was significantly associated with a lower probability of complications. The intake of antiplatelet medication and renal failure as comorbidity was associated with a significantly higher probability for complications. Odds ratios of all factors from logistic regression are displayed in Table 3.
|
|
Diagnostic catheterization |
Interventional catheterization |
| OR [CI] |
P-value |
OR[CI] |
P-value |
| Intercept (basecase) |
0.03 [0.006, 0.16] |
<0.0001 |
0.05 [0.02, 0.14] |
<0.0001 |
| Employment of VCD |
0.31 [0.10, 0.98] |
0.02 |
0.98 [0.73, 1.32] |
0.90 |
| Male gender |
0.49 [0.29, 0.84] |
0.003 |
0.49 [0.37, 0.66] |
<0.0001 |
| Agegroup (in decades) |
0.99 [0.79, 1.24] |
0.91 |
1.12 [0.98, 1.28] |
0.05 |
| Intake of antiplatelets |
2.44 [1.02, 5.85] |
0.02 |
1.53 [1.11, 2.09] |
0.003 |
| Intake of anticoagulants |
1.45 [0.66, 3.18] |
0.29 |
0.74 [0.37, 1.47] |
0.32 |
| Hypertension |
1.35 [0.67, 2.72] |
0.34 |
1.00 [0.70, 1.42] |
0.99 |
| CAD |
1.11 [0.64, 1.92] |
0.66 |
0.73 [0.52, 1.03] |
0.04 |
| Obesity |
0.68 [0.35, 1.32] |
0.19 |
0.78 [0.54, 1.15] |
0.15 |
| Diabetes |
0.79 [0.46, 1.37] |
0.34 |
0.73 [0.54, 1.00] |
0.03 |
| PVD |
1.24 [0.69, 2.23] |
0.42 |
1.03 [0.74, 1.44] |
0.84 |
| Renal failure |
2.26 [1.22, 4.19] |
0.003 |
1.57 [1.06, 2.33] |
0.01 |
VCD=Vascular Closing Device, CAD=Coronary Artery Disease, PVD=Peripheral Artery Disease,
OR=Odds Ratio, CI=97.5% Confidence Interval
Table 3: Parameter estimates of logistic regression of complications by type of catheterization.
Sensitivity analysis
Table 4 shows results from a sensitivity analysis in a subgroup with less complex procedures. Sample size was 494 for diagnostic catheters with VCD usage of 51 (10.3%) and 2,239 interventional catheters with 839 (37.5%) VCDs employed. No significant differences in either the probability of complications or healthcare expenditures were found. Results showed that again male gender significantly lowered the probability of complications in interventional catheterization.
| Costs |
Diagnostic catheterization |
Interventional catheterization |
| VCD (n=51) |
MC (n=443) |
VCD (n=839) |
MC (n=1400) |
| Adjusted costs |
1,287€ |
1,257€ |
3.384€ |
3.354€ |
| Adjusted difference [CI] |
30€ [-31.5, 92.4] |
30€ [-84.7, 145.2] |
| P-value |
0.28 |
0.55 |
| Complications |
OR [CI] |
P-value |
OR [CI] |
P-value |
| Intercept |
- |
- |
0.25 [0.04, 1.60] |
0.09 |
| Employment of VCD |
- |
- |
1.10 [0.64, 1.89] |
0.69 |
| Male gender |
-- |
-- |
0.56 [0.32, 0.97] |
0.02 |
| Age |
-- |
-- |
0.89 [0.70, 1.15] |
0.32 |
| Intake of antiplatelets |
--- |
-- |
1.39 [0.63, 3.08] |
0.35 |
| Intake of anticoagulants |
- |
-- |
0.76 [0.20, 2.92] |
0.65 |
| Hypertension |
-- |
-- |
0.88 [0.46, 1.68] |
0.67 |
| CAD |
-- |
-- |
0.47 [0.24, 0.90] |
0.01 |
| Obesity |
-- |
-- |
0.92 [0.46, 1.78] |
0.77 |
| Diabetes |
-- |
-- |
0.96 [0.55, 1.67] |
0.85 |
| PVD |
-- |
-- |
0.94 [0.50, 1.77] |
0.83 |
| Renal failure |
-- |
-- |
0.81 [0.30, 2.20] |
0.64 |
Table 4: Odds ratio(OR) of complications from logistic regression and adjusted predictions of costs and cost difference with 97.5% confidence interval(CI), from gamma regression with 1,000 bootstraps, in the group of Vascular Closing Devices (VCD) versus manual compression(MC), by type of catheterization. Adjusted by: age, gender, intake of antiplatelet or anticoagulant medication during hospital stay, the comorbidities hypertension, Coronary Artery Disease (CAD), Peripheral Vascular Disease (PVD), renal failure, diabetes and obesity.
Discussion
In our study, we found that VCDs might be the safer option in diagnostic catheterization, in interventional ones there was no significant association concerning the probability of complications. Healthcare expenditures were not significantly influenced in either type of catheterization. Specifically, we found that VCDs were significantly associated with a lower probability of having a complication in diagnostic catheterizations, whereas healthcare expenditures were almost the same and differed only by €6. In interventional catheterizations, the probability of developing a complication was comparable and healthcare expenditures also only differed on average by €28. These results were also mirrored in a sensitivity analysis of less complex cases. Interestingly, in the sensitivity analysis the frequency of VCD usage in diagnostic catheterization was lower than in the primary analysis, indicating that in case of less complex procedures a VCD is used less often.
Overall, in diagnostic catheterization the lower risk of complications with VCD was not mirrored in the analysis of healthcare expenditures. VCD usage did not result in significantly lower healthcare expenditures. A reason for this could lie in the German DRG system where the use of a VCD in general is not reimbursed differently than MC. However, differences could have occurred due to longer or shorter length of stay resulting in surcharges or deductions of the DRG amount. Other factors (intake of antiplatelet medication and renal failure) that significantly increased the probability of complications also significantly increased healthcare expenditures in diagnostic catheterization. In interventional catheterization, the intake of antiplatelet medication and the comorbidity renal failure were significantly correlated with an increasing probability of complication. The same factors were also significantly correlated with increasing healthcare expenditures. Surprisingly, CAD corresponded with a lower probability and significantly lower healthcare expenditures. In general, one would assume that a cardiac comorbidity would not have this protective effect. In this case a possible explanation could result from the stability or acuity of the patient. A patient already diagnosed with CAD might come to the hospital for a scheduled procedure rather than an emergency. But for patients presenting with chest pain diagnostic work-up, subsequent hospitalization, and treatment plan for the presenting complaint may take longer and result in higher risks and higher healthcare expenditures than for a stable patient who returns to the hospital for his scheduled catheterization.
Our study shows different results than a study conducted in Belgium by Kerré et al. [11] in 2014. They found a significant reduction in vascular complications when using a VCD, 3% versus 1.5% in PCIs, whereas we found equal probabilities. Costs were on average 498€ lower when a VCD was employed and mainly attributable to a reduction in the length of stay. However, their study population was healthier than ours according to the prevalence of comorbidities. Also, complication rates were lower giving a possible explanation for this difference in costs. A meta-analysis from 2004 Nikolsky et. al. [4] found no significant differences in complications in both diagnostic and interventional catheterization. Hermanides et. al. [22] conducted a randomized comparison in the Netherlands in 2010 also resulting in no significant differences in the occurrence of vascular complications in interventional procedures. In line with that, Koreny et al. [3] did not find significant differences concerning individual complications in their meta-analysis from 2004, they even found a trend of increasing risk for hematoma and pseudo aneurysm. However, a differentiation between diagnostic and interventional procedures was not included here. Another meta-analysis conducted by Biancari et. al. [5] in 2010 found a significantly higher risk of groin infections after using a VCD, but no significant differences in other complications.
The transferability of the results for the economic effects to other countries is limited as accounting systems and costs differ. However, as to our knowledge there were no published studies relating to healthcare expenditures in Germany, we used a study from the US and one from Switzerland for comparison. Resnic et. al. [6] conducted a decision analytic model in patients undergoing PCI in 2007 in a US hospital setting. They found that VCD usage resulted in a cost reduction of around €40. In another study in 2006, Schoenenberger et. al. [10] compared costs of 43 patients undergoing elective PCI using MC or VCD in Switzerland. They found significant differences in total costs, costs for physicians, nurses and costs on the ward. Compared with our study, expenditures were lower, with €425 for VCD and €934 for MC, but the inclusion criteria were vastly different from our study and they analysed costs from the perspective of a hospital.
A limitation of our study is generated by the characteristics of analysing claims data. As patients are not randomized we could only adjust for confounders that are known and measurable by the data available. Also, complications can be derived only from ICDs coded during the hospital stay from hospital records and ICDs coded by a doctor in the outpatient sector after discharge. We considered also including complications and healthcare expenditures that occurred after discharge in the outpatient sector. However, due to the German accounting and reimbursement system in this sector, with quarterly accounts per case, it was not possible to link ICDs and healthcare expenditures directly to the index case. Another difficulty concerning ICD and OPS codes used in this study was that they were not assigned to a specific date and time. OPS codes in general are assigned to a specific date; however, in our dataset we did not have this information, so we can only assume that the complication arose after the procedure. However, a large part of ICD codes used for the analyses were so-called T-codes that already imply being a complication arising from a procedure. Also, in some cases patients had more than one catheterization during a hospital stay and also more than one code for a VCD, so a definite link from the method of closure to the possible complications cannot be established in every case. Another limitation that might bias the results of this study is that from OPS codes it was not possible to distinguish between catheterizations that were performed through the femoral or the radial artery. Compared with the femoral approach, the radial approach is associated with a significantly decreased risk for bleeding complications [23,24]. The access puncture site closure with this method is done by MC or a compression device [25]. Patients with a radial access would in our analysis appear in the MC group and could bias the result towards fewer complications in this group. However, a large proportion of subjects in our analysis underwent their procedure in the year 2008, when this approach was not yet widely used. In a study by Dehmer et al. on US registry data from 2010 and 2011 the femoral approach was still used in over 90% of cases [26]. Another limitation is that we could not identify when a VCD was employed unsuccessfully. Therefore, patients that were treated with MC after failure of the VCD were counted as VCD in our study. This could cause a bias toward more complications in VCDs, if a complication actually arose from the MC afterwards but was allocated to VCD in our analysis. However, according to a study by Biancari et al. [5] failure occurs only in around 3.6% of cases and moreover we followed the approach of intention to treat. A last limitation lies in the generalizability of claims data. AOK Baden Württemberg is the 5th biggest SHI in Germany, but despite that insures only about 6% of the population. Also, to be able to generalize the results the population of AOK insures has to be representative for the whole population. However, we adjusted for an influence of age, sex and comorbidity in the analysis so these factors shouldn’t play a role in the interpretation of our results. Furthermore, because of the focus of the analysis on expenditures for the hospital stay socioeconomic factors shouldn’t play a big role so at least for the statutory health insurance system the results should be representative.
A strength of our analysis is that it is, to our knowledge, the first study in Germany to compare healthcare expenditures and complications concerning VCDs from the perspective of SHI. As they have to bear the biggest proportion of costs in the German healthcare sector their perspective is of high importance.
Interestingly, in a similar study [27] conducted by our research team we compared costs from the perspective of a hospital and found significant costs differences between VCD use and MC, resulting in higher costs for the latter. Comparing these results could lead to the conclusion that in fact using a VCD saves costs, however SHIs do not benefit from these savings.
In comparison to other studies we had a larger sample size by using data from the 5th biggest SHI. Also, analysing claims data automatically provides a multicentre study. Most other studies done in this field so far are rolled out in one specific hospital and hence are possibly biased by certain rules and approaches. In our study, multiple hospitals of differing sizes were taken into account which provides a more realistic picture of daily routine care. Similar to other studies we found factors that significantly influence the occurrence of complications like e.g., the protective effect of male gender and an increasing risk with the comorbidity renal failure [15,16], indicating the external validity of our analysis. Another strength is that we did separate analyses on diagnostic and interventional procedures. Often only interventional procedures are taken into account, however as we can derive from our results, VCD use in diagnostic procedures has a significant impact on complication rates, but is only used in around 13% of cases.
Conclusion and Future Research
Our study indicates that there is no significant association between the use of a VCD and resulting healthcare expenditures from the perspective of the German SHI system. However, VCD use is significantly associated with fewer complications in diagnostic catheterization, which could be considered as a first indication that a more extensive use of VCDs in this area should be considered. So far VCD use is not reflected in DRGs in the German system, so their usage especially in diagnostic procedures, where they seem to have more advantages, is not encouraged by the authorities. However, to support an explicit recommendation regarding the usage, these results need to be confirmed by prospective randomized clinical trials.
Future research should be done in assessing possible long-term effects like PVD on a larger scale. Also, a higher focus on who benefits the most from using a VCD in terms of complications would be useful in order to make resource allocation more beneficial.
References
- Todesursachen (2013) In: Gesundheit Todesursachen in Deutschland. Wiesbaden: Statistisches Bundesamt 4.
- Van BF (2010) Bericht über die Leistungszahlen der Herzkatheterlabore in der Bundesrepublik Deutschland. Der Kardiologe 4(6): 502-508.
- Koreny M, Riedmuller E, Nikfardjam M, Siostrzonek P, Mullner M (2004) Arterial puncture closing devices compared with standard manual compression after cardiac catheterization: A systematic review and meta-analysis. JAMA 291(3): 350-357.
- Nikolsky E, Mehran R, Halkin A, Aymong ED, Mintz GS, et al. (2004) Vascular complications associated with arteriotomy closure devices in patients undergoing percutaneous coronary procedures: A meta-analysis. J Am Coll Cardiol 44(6): 1200-1209.
- Biancari F, D'Andrea V, Marco DC, Savino G, Tiozzo V (2010) Catania: Meta-analysis of randomized trials on the efficacy of vascular closure devices after diagnostic angiography and angioplasty. Am Heart J 159(4): 518-531.
- Resnic FS, Arora N, Matheny M, Reynolds MR (2007) A cost-minimization analysis of the angio-seal vascular closure device following percutaneous coronary intervention. Am J Cardiol 99(6): 766-770.
- Juergens CP, Leung DY, Crozier JA, Wong AM, Robinson JT, et al. (2004) Patient tolerance and resource utilization associated with an arterial closure versus an external compression device after percutaneous coronary intervention. Catheter Cardio Inte 63(2): 166-170.
- Rickli H, Unterweger M, Sutsch G, Brunner La HP, Sagmeister M, et al. (2002) Comparison of costs and safety of a suture-mediated closure device with conventional manual compression after coronary artery interventions. Catheter Cardio Inte, 57(3): 297-302.
- Carere RG, Webb JG, Buller CE, Wilson M, Rahman T, et al. (2000) Suture closure of femoral arterial puncture sites after coronary angioplasty followed by same-day discharge. Am Heart J 139(1): 52-58.
- Schoenenberger U, Ammann P, Maeder M, Rickli H (2006) Angio-Seal vascular closure device: an evaluation of cost effectiveness. Cardiovasc Med 9:257-264.
- Kerre S, Kustermans L, Vandendriessche T, Bosmans J, Haine SE, et al. Cost-effectiveness of contemporary vascular closure devices for the prevention of vascular complications after percutaneous coronary interventions in an all-comers PCI population. Euro Soc Cardio 10(2): 191-197.
- Geschäftsbericht der AOK Baden-Württemberg (2013/14). In.: AOK Baden-Württemberg.
- Kolluri R, Fowler B, Nandish S (2013) Vascular access complications: Diagnosis and management. Curr Treat Opt Cardio med 15(2): 173-187.
- Chevalier B, Lancelin B, Koning R, Henry M, Gommeaux A, et al. (2003) Effect of a closure device on complication rates in high-local-risk patients: results of a randomized multicenter trial. Catheter Cardio Inte 58(3): 285-291.
- Schwartz BG, Burstein S, Economides C, Kloner RA, Shavelle DM, et al. (2010) Review of vascular closure devices. J inva Cardiol 22(12): 599-607.
- Merriweather N, Sulzbach HLM (2012) Managing risk of complications at femoral vascular access sites in percutaneous coronary intervention. Crit Care Nurs 32(5): 16-29.
- Byrne RA, Cassese S, Linhardt M, Kastrati A (2013) Vascular access and closure in coronary angiography and percutaneous intervention. Nature Rev Cardiol 10(1): 27-40.
- Ates M, Sahin S, Konuralp C, Gullu U, Cimen S, et al. (2006) Evaluation of risk factors associated with femoral pseudoaneurysms after cardiac catheterization. J Vascular Surg 43(3): 520-524.
- Hamm CW, Albrecht A, Bonzel T, Kelm M, Lange H, et al. (2008) Diagnostic heart catheterization. Clin Res Cardiol, 97(8): 475-512.
- Kilian R, Matschinger H, Loeffler W, Roick C, Angermeyer MC (2002) A comparison of methods to handle skew distributed cost variables in the analysis of the resource consumption in schizophrenia treatment. J Ment Health Policy Econ 5(1): 21-31.
- Graubard BI, Korn EL (1999) Predictive margins with survey data. Biometrics 55(2): 652-659.
- Hermanides RS, Ottervanger JP, Dambrink JH, De Boer MJ, Hoorntje JC, et al. (2010) Closure device or manual compression in patients undergoing percutaneous coronary intervention: a randomized comparison. Inva cardiol, 22(12): 562-566.
- Rao SV, Ou FS, Wang TY, Roe MT, Brindis R, et al. (2008) Trends in the prevalence and outcomes of radial and femoral approaches to percutaneous coronary intervention: a report from the National Cardiovascular Data Registry. JACC Cardio Inte 1(4): 379-386.
- Jolly SS, Amlani S, Hamon M, Yusuf S, Mehta SR (2009) Radial versus femoral access for coronary angiography or intervention and the impact on major bleeding and ischemic events: A systematic review and meta-analysis of randomized trials. Am Heart J 157(1): 132-140.
- Balaji NR, Shah PB (2011) Cardiology patient page. Radial artery catheterization. Circulation 124(16): e407-e408.
- Dehmer GJ, Weaver D, Roe MT, Milford BS, Fitzgerald S, et al. (2012) A contemporary view of diagnostic cardiac catheterization and percutaneous coronary intervention in the United States: A report from the CathPCI Registry of the National Cardiovascular Data Registry, 2010 through June 2011. J Am Coll Cardiol 60(20): 2017-2031.
- Walter J, Vogl M, Holderried M, Becker C, Brandes A, et al. (2016) Manual compression versus vascular closing device for closing access puncture site in femoral left-heart catheterization and percutaneous coronary interventions: A retrospective cross-sectional comparison of costs and effects in inpatient care. Value Health 20(6): 769-776.