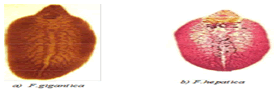Keywords: Bovine; Fasciolosis
Introduction
Bovine fasciolosis is economically important parasitic diseases of cattle caused by digenaen trematodes of genus Fasciola commonly referred to as liver fluke. F. hepatica and F. gigantica are the two species most commonly implicate as the etiological agents of the fasciolosis. F. hepatica has a worldwide distribution while F. gigantica is found on most continents primarily in tropical region [1].
Fasciola hepatica, the common liver fluke, causesfasciolosis or (liver rot). It may lead to secondary bacterial infection such as bacillary icterohemoglobinuria (red water) in cattle. This disease is caused by members of the genus Clostridium, anaerobic spore forming bacteria [2]. It passes its life cycle in two different hosts; those are intermediate and definite hosts (DH). Definitive hosts include cattle, sheep, many other ruminants, equidae, swine and rabbits. The genus Lymnaea in general; and L.truncatula and L. auricularia in particular are the most common intermediate hosts for F. hepatica and F. gigantica respectively. These intermediate hosts serveas means of transmission to animals, that the animal gain accesses their infective stage via feed and water [3].
Pathogenesis of fasciolosis varies according to the parasitic development phases: parenchymal and biliary phases. The parenchymal phase occurs during migration of flukes through the liver parenchyma and is associated with liver damage and hemorrhage. The biliary phase coincides with parasite residence in the bile ducts and results from the haematophagic activity of the adult flukes and from the damage to the bile duct mucosa by their cuticular spines [4]. Chronic fasciolosis is the most common form of the disease in cattle. It occurs when the parasite reaches the hepatic bile duct [5]. Animal become emaciated, have pale eye and gums and typically develop “bottle jaw” due to edema under the jaw [6]. Diagnosis of fasciolosis is based on clinical sign, grazing history and seasonal occurrence, examination of feces by laboratory tests and post mortem examination [3].
Drugs differ in their efficacy, mode of action and cost. Numbers of drugs have been used to control fasciolosis in animals. Triclabendazole is the treatment of choice due to its effectiveness for both the larval and adult flukes and the main control measures are reduction of snail population, use of antihelmintics and immunization and immunity [7]. Control of fasciolosis may be approached by reducing population of the intermediate snail host and by using anthelmintics [3].
The economic losses due to fasciolosis throughout the world are enormous and these losses are associated with mortality, morbidity, reduced growth rate, condemnation of liver, increased susceptibility to secondary infections and expense due to control measures [8]. Ethiopia has substantial livestock resources; its level of productivity is low due to constraints of disease. Out of these diseases, fasciolosis is one of the significant diseases of animals [3].
The objectives of this review paper are:
To review about bovine fasciolosis
To provide some information about epidemiology, treatment and control of the disease.
Cattle Fasciolosis
Definition
Fasciolosis is also known as, fasciolasis, distomatosis and liver rot. Fasciolosis is a parasitic disease of sheep, goat and cattle. It occasionally affects humans, hence considered as a zoonotic disease [9, 10]. They are responsible for wide spread morbidity and mortality in cattle characterized by weight loss, anemia and hypoproteinemia [3].
Etiology
Fasciolosis is caused by different species of trematodes (commonly called “flukes”) of the genus Fasciola [11]. The taxonomic classification of theorganisms that cause fasciolosis is presented as follows: Phylum: Platyhelminthes, Class: Trematoda, Sub class: Digenea, Order: Echinostomida, Super family: Fascioloidea, Genus: Fasciola, Species: F. hepatica and F. gigantica [4].
Morphology
Fasciola hepatica is a leaf shaped, fluke with broad and cone shaped anterior projection. It is grayish brown in color changing to gray when preserved. The tegument is armed with sharp spines. The young fluke at the time of entry in to the liver is 1-2 millimeter in length and lancet like when it has become fully mature in the bile ducts. It is leaf-shaped gray brown in color and is around 3.5cm in length and 1cm in width [3]. The eggs of F. hepatica are oval in shape, brownish or yellowish brown in color. The eggs have an indistinct operculum and develop only after the eggs have been laid.
Figure 1: Adult stages of Fasciola spp. Source: [4].
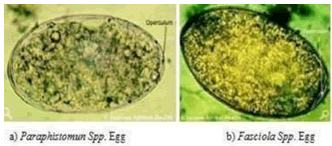
Fasciola eggs should be distinguished from the eggs of other flukes, especially from the large eggs of paramphistomum. Fasciola eggs have yellowish brownshell with an indistinct operculum and embryonic cells where as paramphistomum eggs have transparent shell, distinct operculum with embryonic clear cells and possess a small knob at their posterior ends [4].
gigantica is larger than F. hepatica and canreach up to 7.5cm length. The shape is more of leaf like, the conical anterior end is very short and the shoulder characteristic of F. hepatica is barely perceptible. The eggs are larger than those of F. hepatica, measuring 190x100 micrometer (µm) [3, 4].
Epidemiolog
Geographical Distribution: Fasciolosis is considered as important limiting factor for bovine and ovine production. This fluke’s distribution is worldwide in areas where cattle, sheep and goat are raised and there is a niche for Laymnaeid snail [2]. F. hepatica, which is a temperate species, is the mostly important trematode of domestic ruminant and common causes of liver fluke disease in temperate areas of the world. Thus, it is found in Southern and Northern America, Europe, Australia and Africa. F. gigantica, on the other hand, is economically important and widely distributed in tropical countries of Africa and Asia [12]. In Ethiopia, F. gigantica is found at altitudes below 1800 meter below sea level while F. hepatica is found at altitudes between 1200-2560meters above sea level. Mixed infection by the two species can be encountered at 1200-1800 meter above sea level [8].
Risk Factors: The main factors determining the timingand severity of fasciolosis depend on the number of metacercaria accumulating on herbage. Particularly temperature and rainfall affect both the spatial and temporal abundance of snail hosts and the rate of development of fluke eggs and larvae. There are three most important factors that influence the occurrence of fasciolosis [13].
Availability of Suitable Snail Habitat: One of the most important factors that influence the occurrence of fasciolosis in an area is availability of suitable snail habitat. The snail habitat may be permanent or temporary. The availability of these intermediate hosts depends on climatic conditions for instance, L. truncatuala prefers wet mud to free water and permanent habitat includes the ban of ditches or streams and edges of small ponds. Fields with clumps of rushes are often suspect site. Through a slightly acid pH environment is optimal for L. truncatula, excessively acid pH levels are detrimental [3].
Temperature : It is an important factor affecting the rate ofdevelopment of snails and the stages of the parasite outside the final host. A mid -day or night temperature of 10°c or above is necessary for both snails to breed and for the development of F. hepatica within the snail and all activity cease at 5°c [3]. Fasciola cercaria and Lymnaea snails have been found to survive better at 25-30°c which explains in parts at least, the much higher prevalence in autumn compared to other seasons [13].
Moisture : The ideal moisture conditions for breeding anddevelopment of Fasciola within snails are provided when rainfall exceeds transpiration and field saturation is attained. Such conditions are also essential for the development of fluke eggs, for miracidium to search for snails and for the dispersal of cercariae shed from the snails [3].
Host range
Definitive host: Its definitive hosts include cattle, sheep, many other ruminants, equidae, swine and rabbits. Finally it should be remembered that F. hepatica can infect wide range of mammals and is possible that on occasions these hosts may act as reservoirs of infection, but F. gigantica infect ruminants [3].
Intermediate host: Its Intermediate hosts includemolluscs of the family Lymnaeidae such as Lymnaea. Snails of the genus Lymnaea, the most common L.truncatula, are an amphibious snail with a widedistribution throughout the world. Other important Lymnaea vectors of F. hepatica outside Europe are L.tomentosa in Australia and New Zealand, L. cumella inNorth America, Australia and New Zealand, L. bulimoidis in Southern USA and the Caribbean, L. humlis in North America, L. viator in South America and L. diaphena in South America. On the other hand, snails of the genus Lymnaea, L. auricularia, other snails L. natalensis, L. rufescens and L. rubiginas are IH of F. gigantica. All areprimarily aquatic and found in streams, irrigation channels and marshy swamps [3].
Life cycle
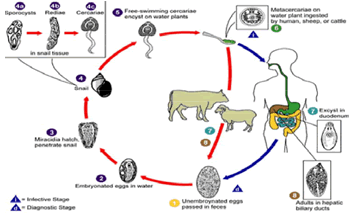
Adult flukes in the bile duct shed eggs into the bile, which enter into the intestine. Eggs reach outside by passing down the common bile duct and being voided with feces. They are undeveloped when passed and require minimum of 10 days to reach miracidial stage [2-4].
The eggs of flukes passed in the feces of mammalian host develop and hatch releasing motile, ciliated miracidium. These takes 9 days at optimum temperature of 22-26°c and little development occurs below 10°c [4].
The liberated miracidium has a short life span and must locate a suitable snail within 3 hours and successful penetration of the tissue of snail occurs. In infected snails, development proceeds through the sporoyst and redial stage to the final stage in the IH, the cercaria; these are shed from the snail as motile forms which attach themselves to frame surface, such as grass blades and encyst there to form the infective metacercariae [6, 11]. It takes a minimum of 6-7 weeks for completion of the development from miracidium to metacercariae [4].
Metacercariae are ingested by the final host, excyst in the small intestine, migrate through the gut wall, cross the peritoneum and penetrate the liver capsule. The young flukes tunnel through the liver parenchyma for 6-8 weeks and then enter into the bile ducts where they migrate to the large ducts and occasionally the gall bladder. The prepatent period is 10-12weeks [4]. The minimal period for the completion of one entire life cycle of F. hepatica is therefore 17-18 weeks. The longevity of F. hepatica in untreated cattle is usually less than one year [3].
Animals are infected by F. gigantica at water holes; infection depends on several factors related to the biology of the vector, biology of the parasite and management of flocks and herds. Human beings are occasionally infected by ingestion of metacercariae which encysted in water plants. Transmission by consuming raw liver dishes prepared from fresh livers of infected with immature Fasciola species is also possible. Major symptoms include intestinal discomfort, painful liver regions and anemia [3, 7].
Figure 2: Life Cycle of fasciola source: [5]
Pathogenesis
The pathogenesis of fasciolosis variesaccording to the phase of parasite development in the liver and species of host involved. Essentially the pathogenesis has two phases. The first phase occurs during migration in the liver parenchyma and is associated with liver damage and hemorrhage. The second occurs when the parasite is in the bile duct and result from the haematophagic activity of the adult fluke and from damage to the biliary mucosa by their tegumental spines [3, 4].
Pathogenesis as a direct result of the fluke’s activity may be either acute or chronic. Acute fasciolosis occurs during the pre-adult migration of the flukes in the parenchyma of the liver. In chronic fasciolosis, the damage is primarily to the liver. Unlike the acute phase of the disease, the damage occurs from the fibrosis (scar tissue), blockage of the bile duct and inflammation of the bile ducts [3].
The pathogenesis of bovine fasciolosis is similar to that in sheep but has the added features of calcification of the bile ducts and enlargement of gall bladder. Although acute and sub-acute disease may occasionally occur under condition of heavy challenges especially in young calves, chronic form of the disease is by far the most as sheep seen in the late winter or early spring [3, 4].
Aberrant migration of the flukes is more common in cattle and encapsulated parasites are often seen in the lungs ([4]. Migrating F. hepatica through hepatic tissue containing quiescent spores of Clostridium nobyi may cause the development of infectious necrotic hepatitis in sheep and cattle. This migration has been thought to stimulate the development of occasional causes of bacillary haemoglobinuria in cattle. Immature flukes are tissue feeders, but may accidentally ingest some blood and the minor degree anemia that develops in the tracts 4-5 weeks of ingestion probably reflects the loss of blood into migratory tracts of young flukes [14].
Mechanical (obstruction of parenchyma andblood vessels of the liver by immature flukes burrowing through the liver and irritation of the epithelia lining of the bile ducts by the adult); toxic (by secretary and excretory product of the fluke); and loss of blood resulting from hemorrhage in the liver (acute form) and hematophagous feeding habits of the flukes [7]. Light infection due to F. hepatica may be asymptomatic. However they mayproduce hepatic colic with coughing and vomiting, generalized abdominal rigidity, head ache and sweating, irregular fever, diarrhea, pipe clay and anemia [15].
Clinical signs
The clinical features of fasciolosis may vary depending on forms of the disease (acute, sub-acute and chronic).
Acute fasciolosis: Occur rarely in cattle. It is less common than the chronic form and hepatitis is caused by simultaneous migration of large number of immature flukes. Sudden death may occur in acute fasciolosis [3, 16]. It is responsible for wide spread morbidity and mortality in cattle characterized by weight loss, anemia and hypoproteinemia.
Sub-acute fasciolosis: Is caused by ingestion of a moderate number of metacercaria and is characterized by anemia, jaundice and ill-thrift [3, 15]
Chronic fasciolosis: This is the most common form of the disease in cattle. It occurs when the parasite reaches the hepatic bile duct [5]. Animal become emaciated, have pale eye and gums and typically develop “bottle jaw” due to edema under the jaw [6]. Depending on the severity of the disease, death in untreated animals follows in about two to three months although many survive longer than this and may eventually recover if not re infected. Cattle with chronic fasciolosis may develop diarrhea but tend to lose most of their infections after about six months [6].
Diagnosis
Diagnosis of fasciolosis is based on clinical sign, grazing history and seasonal occurrence, examination of feces by laboratory tests and post mortem examination [3].
History and clinical sign: Infection with F. hepaticaisusually associated with herds and flocks grazing wet, marshy land. On the other hand, F. gigantica occurs during livestock drinking water from snail infected watering places as well as with grazing wet land which may be seasonally not dated [17]. In acute case of fasciolosis sudden death and severe anemia occur due to migrating young flukes through the liver; however no fluke’s eggs are passed in the feces. Sub-acute case cause signs of rapid loss of condition, severe anemia, high fluke egg count and death occurs 12-30 weeks after infection and in chronic fasciolosis gradual wasting, severe anemia with ascites, bottles jaw and very high fluke egg count which may lead to death more than 20 weeks after infection [3].
Fecal Examination: Chronic fasciolosis is diagnosed byfinding eggs in the feces by using sedimentation technique. However they must be distinguished from the eggs of the other flukes especially the large eggs of paramphistomum Paramphistomum. Examinationemploying sedimentation technique Fasciola eggs have high specific gravity and sedimentation is preferred to floatation [18]. The oval operculated golden eggs of F.hepatica appear in the feces 10 weeks after infection,while F. gigantica eggs only appear 15 weeks after infection. Excretion of fluke eggs shows considerable day to day and within day variation and the distribution of eggs in feces are irregular; single fecal egg count assay may lead to incorrect conclusion [7].
Serology: In vivodiagnosis of mild and prepatentinfection is possible serologically. For example, detection of antibodies by ELISA in serum or milk is available and particularly useful for diagnosis of infection in cattle in an individual or herd basis. A rise in antibodies can be detected by two weeks after infection and keeps rising until week six [7, 19].
Figure 3: Rumen and Liver Fluke Eggs Source: [18].
Necropsy: The detection of adult flukes in the liver atnecropsy is the most reliable method to confirm fasciolosis. Prevalence studies should be based on abattoir survey other than coproscopic investigation [16]. Acute fasciolosis is characterized by a badly damaged, swollen liver. The peritoneal cavity may contain an excess of blood-stained serum. The liver capsule shows many small perforations, sub capsular hemorrhage and the parenchyma shows tracts of damaged tissue and much more friable than the normal [19].
Chronic fasciolosis is characterized by the presence of leaf-like flukes in grossly enlarged and thickened bile ducts particularly in the ventral lobe of the liver. Calcification of bile duct walls is a common finding in cattle. The hepatic parenchyma is extensively fibrosed and the hepatic lymph nodes are dark brown in color [19].
Importance of Fasciolosis
Public Health Importance: Human fasciolosis has been reported from countries in Europe, America, Asia, Africa and Oceania. The incidence of human case has been increasing in the 51 countries of 5 continents. A person must ingest the metacercaria to become infected [20]. Human acquire infection through ingestion of metacercaria that are attached to certain aquatic plant and vegetable. In addition experimental studies suggested that human consuming raw liver dish from liver infected with juvenile flukes could become infected [4].
Economic Importance: Fasciolosis causes major economicloss in cattle, goat, buffalo and sheep [21]. The disease causes considerable impact on the economy of the livestock industry. The economic losses consist of costs of anthelmintics, drenches, labor, liver condemnation at meat inspection; and losses in production due to mortality, reduction in meat, milk and reduction in growth rate, fertility and decreased feed intake, conversion and lower resistance to other disease [22].
Treatment
Not all compounds are equally effective against all stages of development of F. hepatica in the body. For the treatment of acute fasciolosis, it is essential to choose a product which is highly effective against the juveniles that damage the liver parenchyma. For chronic disease a compound active against adult fluke is required [19].
Triclabendazole (12 mg/kg) is considered as the most common drug due to its high efficacy against adult as well juvenile flukes. It is effective against adult F. hepatica at a dose rate of 10mg/kg in cattle. It is ovicidal and will kill any F. hepatica eggs present in the bile duct or the alimentary tract at the time of treatment supplied in combination with ivermectin for combined fluke and round warm control in cattle.
Nitroxynil is given subcutaneously at 10mg/kg and has good efficacy against the adult fluke but the dose has to be increased by up to 50% to obtain adequate control of acute disease [19].
Until recently treatment was not highly successful due to the in efficiency of the older drugs against the early parenchymal stages. However, efficient drugs are now available and the one of the choice is triclabendazole which removes all developing stages over one week old.
Other drugs are Rafoxanide (7.5 mg/kg), Closantel and Nitroxynil, which will remove flukes over four weeks old [3].
Control and prevention
Program charts for fasciolosis control can be produced based on average rainfall and temperature records of any geographic region [14]. Reduction of snail population and use of anthelmintics are the two major control and prevention strategies against fasciolosis [19].
Reduction of Snail Population: Before any scheme ofsnail control is under taken a survey of the snail habitat should be made to determine whether they are localized or wide spread. The best long term method for reducing mud snail population such as L. truncatula ensures permanent destruction of snail habitat. The snail habitat is limited by simple method of fencing these areas or treat annually with a molluscicide [2, 3]. Control of snail by chemical such as niclosamide, copper sulphate focally and seasonally possible, however, usually not practical due to labor, high cost, environmental consideration and rapid colonization of snail habitats [2].
Use of anthelmintic: It is true that seasonal strategicapplication of effective anthelmintics which is specific for trematode as well as timely prophylactic and curative treatment play an important role in the control of liver fluke infection [23]. The prophylactic use of anthelmintic aiming to reduce pasture contamination by fluke eggs at times most suitable for development of fluke, April to august and removing fluke population at times of heavy burdens or at periods of nutritional stress to animal. Prophylactic treatment in cattle is therefore directed at reducing the fluke burdens in the winter at a time when the nutritional status of the animal is at its lowest level [3]. Other control methods include environmental sanitation and manipulation (draining, swamps, building sewage system and providing clean water supplies), rotational grazing and also avoiding mixed grazing of animals of different age groups (young animals are generally susceptible to helminthes infection [23].
A great number of chemicals have been used as molluscicides in past, but at present, Niclosamide and Copper sulphate are used in different parts of African countries [24].
Host range, longevity and egg production of liver fluke in definitive host have implication for control. In Sheep it is estimated that one liver fluke produces about 20,000 eggs per day. Cattle, on the other hand, are relatively poor hosts in that the infection usually retain only for months. Egg production by F. hepatica is also poorer than in sheep. Pasture contamination by cattle therefore increase more slowly and ends sooner than with Sheep [2].
Immunity and Immunization: It has been suggested thatnatural immunity is expressed both during the migratory parenchymal and adult bile duct stages of the infection. This is considered to be related to the distribution and amount of connective tissue in the hosts liver parenchyma. Cattles are more resistance because of the relatively large amount of connective tissue in their liver. Possibly the connective tissue helps to trap young migrating flukes. Immunity to F. hepatica has been demonstrated and antibodies can be found in the blood of infected animal. Observation in the field indicated that older animals become resistant to infection [3].
hepatica has a number of survival mechanisms for evading host immune responses, including changing its surface antigen during migration, releasing a proteolytic enzyme that can cleave immunoglobins and modulating the host immune response [19].
Vaccine for F. hepatica is under development. One of these which use recombinant fluke cathepsin L proteinase has given up to 79% protection against infection in cattle and sheep. Successful vaccination strategies elicit Th1 rather than Th2 immune response induced by natural infection [19].
Conclusion
Bovine fasciolosis causes severe liver damage especially in calves and increases the susceptibility of cattle to secondary bacterial infections. The liver damage results in failure to gain weight in young animals and weight losses in older animals. The disease constitutes a major impediment to livestock production owing to the direct and indirect losses. Fasciolosis is now recognized as an emerging human disease. Natural immunity is expressed both during the migratory parenchymal and adult bile duct stage of the infection. There is a risk of development of drug resistance in Fasciola through frequent use of single anthelmintics. The fluke worldwide distribution occurs in areas where cattle and sheep are raised and there is a niche for Lymnaeid snail. Mostly in developing countries including Ethiopia, cattle management systems are extensively rearing which make the animal to be easily exposed to the disease.
From the above conclusion, the following recommendations are forwarded:
Farmers should be trained about the zoonotic and the economic impact of the disease.
Education of the farmers should be carried out about the management system of their animals to minimize its risk of occurrence in their livestock population.
Control should be on preventive rather than treatment. Anthelmintics treatment should be combined with improved pasture management. Strategic anthelmintics treatment with appropriate flukicidal drugs should be practiced twice a year; before and after rainy seasons to eliminate fluke burden of the host animal and minimize pasture contamination by fecal egg shedding thus interrupting the life cycle. Cook water-grown vegetables thoroughly before eating.
References
- Ahmed, MA (2009) Prevalence of ovine fasciolosis in and around Kombolcha. DVM thesis FVM, University of Gondar, Gondar, Ethiopia.
- Marquardt WC, RS Demaree, RB Grieve (2000) Parasitology and Vector Biology. 2nded. London: Harcourt Academic press: 273-279.
- Urquhart, G.M., J.L. Amour, A.M. Dunn and F.W. Jennings (1996). Veterinary Parasitology. 2nded.Oxford: Blackwell publishing 103-112.
- Taylor, M.A., R.L. Coop and R.L. Wall (2007) Veterinary parasitology. 3rd ed. Oxford: Blackwell Publishing 85-87.
- Michael A (2004) Infectious prevalence of ovine Fasciolosis in irrigation schemes along the upper Awash River Basin and effect of strategic anthelmintic treatment in selected up stream areas. MSC thesis, Addis Ababa University, school of Graduate studies, Department of Biology, Addis Ababa, Ethiopia.
- Hunter A (1994) Fasciolosis in animal health. Specific Disease in Tropics: 1st ed. London: CTA, Macmillan, 2: 149-154.
- Kassai, T., 1999. Veterinary Helmintology. University of science, Oxford: Butterworth Heinemann. pp: 9.
- Yilma JJ, B. Malone (1998) A geographical information System forecast model for strategic control of fasciolosis in Ethiopia: Veterinary parasitology, pp: 103-12.
- Andrews, SJ (1999) The life cycle of F. hepatica. 3rded. Wallingford: CABI publishing, pp: 1-30.
- WHO (1995) Control of food borne trematode infections. Technical Report series, 849: 64.
- Scott, JM, PH Goll, (1997) The epidemiology and Anthelmintic control of Ovine Fasciolosis in Ethiopian central highlands. British veterinary Journal, 133: 273-280.
- Khan, E.M (2005) The Merck Veterinary Manual. 9th ed. USA: Merck Inc : 269.
- Maqbool, A.C.S., H.A. Akhtar and K.A. Hashm. (2002). Epidemiology of fasciolosis in Buffaloes under different management conditions. Vet. arhiv., 72: 221-228.
- Wakuma, M., (2009) Prevalence and economic significance of bovine fasciolosis at Bedele municipal abattoir. DVM thesis, FVM, Jimma University, Jimma, Ethiopia.
- Behm, C.A. and N.C.Sangster, (1999). Pathology, Pathophysiology and clinical aspects of fasciolosis. 2nd ed. Welling ford: CABI publishing, pp: 185-224.
- Kaufmann, J., (1996) Parasitic Infection of Domestic Animals. A diagnostic manual: 1sted. Berlin: Birhouser Verlag., pp: 90-92.
- Payne, W.T.A., (990) Introduction to Animal Husbandry in the Tropics. 4th ed. Oxford: Blackwell science, pp: 47-74.
- Sloss, M.W., R.L. Kemp and A.M. Zajac, 1994. Veterinary clinical parasitology. 6thed. London: Blackwell publishing, pp: 90-92.
- Radostits, O.M., C.C. Gay, K.W. Hinchclitt and P.D. Constable, 2007. Veterinary Medicine, a text book of the disease of cattle, horses, sheep, goats and pigs. 10thed. New York: Elsevier, pp: 1576-1579.
- Mas-Coma, S., M.D. Barguest and J.G. Esteban, 1999. Human Fasciolosis. In Dalton, J.P. (eds.): Welling Ford, CABI publishing, UK, pp: 411-434.
- Food Agricultural Organization of United Nations (FAO), 1994. Diseases of domestic animals caused by flukes. Epidemiology, diagnosis and control of Fasciola, Paramphistome, Dicrocoelium, Eurytrema and Schistosome infections of ruminants in developing countries, Rome, Italy, pp: 49.
- Assefa, M., 2005. Parasitic causes of carcass or organ condemnation at Assela municipality abattoir. DVM thesis, Addis Ababa University, Faculty of veterinary Medicine: Debre Zeit, Ethiopia.
- Hansen, J. and B. Perry, 1994. The epidemiology, Diagnosis and control of Helminth parasites of Ruminants. 4th ed. Rome, Italy, pp: 72.
- Brown, D.D., 2003. Parasitology for Veterinarians. 8thed. New York: Sounders Elsevier. pp: 115-122.