Background: Type 2 diabetes mellitus (DM) is the most common cause of end- stage renal disease. Albuminuria is the foremost commonly utilized marker to anticipate onset of diabetic nephropathy (DN) without sufficient affectability and specificity to identify early DN.
Aim: This study aimed to evaluate plasma α-Klotho as a new biomarker for early DN.
Methods: This cross sectional study included 125 Egyptian subjects attending the out Patients Clinic of the Department of Internal Medicine, 10Th of Ramadan city Health Insurance Hospital and divided into:-control group, patient with diabetic mellitus, patients with Diabetic nephropathy and patient with diabetic nephropathy and other complications. Patients were subjected to measurement of plasma α- Klotho, FBS, HbAIC, serum Creatinine, serum urea, serum uric acid, k, Na, serum phosphorus, Albumin: Creatinine Ratio, GFR, Chol, TG, LDL HDL, AST, ALT, T.BIL, D.BIL ALB, TP, GLB and A/G ratio.
Results: Results showed that plasma a-klotho was significantly correlated with haemoglobin A1C, potassium, GFR, Albumin, TP and GLB. Meanwhile, plasma α - klotho was negatively correlated with duration of DM, CR, Urea, UR.A, Na, phosphorus, ACR, Chol, TG, LDL, AST, ALT, T.BIL, and D.BIL. However, there were no significant correlations between plasma a-klotho and FBS, HDL and A/G ratio. At cut-off level ≥2.6, plasma a-klotho had 95% sensitivity and 81% specificity for diagnosing diabetic nephropathy.
Conclusion: α-klotho may be the early markers for predict in renal injury in patients with type 2 diabetes.
Keywords
α-klotho; Diabetes Mellitus; Nephropathy
Introduction
Type 2 diabetes mellitus (DM) is the most common single
cause of End -Stage Renal Disease (ESRD) [1]. ESRD in nearly half
of patients is due to diabetic nephropathy (DN), and these cases
have the most exceedingly bad result compared to patients with
other causes of ESRD. In spite of the fact that there are
numerous novel drugs for DM, there are no particular healing
medicines however for DN.
Reasons for destitute result incorporate insufficient markers
and the complicated components of DN [2]. Now, severity of this
disease is decided agreeing to the levels of albuminuria.
Albuminuria is the foremost commonly utilized marker to
foresee onset and movement of DN clinically. In any case, this
conventional marker for DN needs both affectability and
specificity to identify early organizes of DN [3].
However, some DN patients with ESRD do not present with
significant albuminuria [4-6]. There is lack of association
between Glomerular Filtration Rate (GFR) and albuminuria
suggests that an alternative to this albuminuria-based staging
system is needed. Some studies have noted the existence of
pathological change before micro albuminuria. Therefore, even
if micro albuminuria can be regarded as the earliest
manifestation of DN, it is possible that a new biomarker for DN
exists. Recently, different markers of DN were reviewed
including fibroblast growth factor 23, tubular markers
inflammatory markers (Interleukin 6 [IL-6], IL-8, monocyte
chemo attractant protein 1, and interferon γ–inducible protein)
urinary 8-hydroxy-20- deoxyguanosine, serum cystatin C, and so
on. Among these, genetic susceptibility almost always leads to
irreversible DN, and detection of the clinical markers mostly
occurs too late to diagnose and monitor the progression of DN.
As such, it is crucial to find an earlier and reliable marker for DN.
Earlier diagnosis and intervention may provide an opportunity to
stop the permanent damage caused by DN. Although a-klotho
was first described as an anti-aging factor, recent experimental
and clinical studies suggest a-klotho also has important pleotroic
effects on the kidneys. Soluble a-klotho is derived from the
proteolytic cleavage of the extracellular portion of the
membrane-bound a-klotho; alternatively, it can be generated
directly by the alterative splicing of the a-klotho transcript. It can
be measured in blood, urine, and cerebrospinal.
Animals with chronic kidney disease have very low renal,
plasma, and urinary a-klotho levels. Furthermore, humans with
chronic kidney disease exhibit markedly reduced a-klotho in serum and urine in the early stages of kidney disease, progressively decreasing in more advanced stages. However,
with regard to diabetic nephropathy, the role of aklotho in the
pathogenesis of kidney injury has not been fully studied. Renal
a-klotho expression is markedly decreased in diabetic
nephropathy in humans and mice. A similar decline is observed
in kidney cells treated with methylglyoxal-modified albumin.
These findings collectively suggest renal a- klotho deficiency is
part of an underlying mechanism involved in diabetic kidney
injury. However, the actual role of soluble a-klotho in diabetic kidney disease has not been evaluated. So this study aimed to
evaluate Plasma a-klotho as biomarkers in chronic diabetic
nephropathy.
Aim of the work
The study aimed to evaluate Plasma a-klotho as biomarkers in
chronic diabetic nephropathy.
Patients and Methods
Study design
Cross sectional study, aiming to evaluate plasma a-klotho as
biomarkers in chronic diabetic nephropathy.
Study setting
The study was carried out at Clinic of the Department of
Internal Medicine, 10Th of Ramadan city Health Insurance
Hospital.
Target population
Diabetic patients attending the Out Patients Clinic of the
Department of Internal Medicine, 10Th of Ramadan city Health
Insurance Hospital. This study included 125 Participants who
were divided into:-
1- Control group:- 20 healthy subjects whose age ranged
between 30-50 years old were taken as control group.
2- Study group:- including 105 patients divided into.
Group 1 :- 20 patient with diabetic mellitus whose age ranged
between 30-50 years old
Group 2 :- 65 patients Diabetic nephropathy whose age
ranged between 30-50 years old
Group 3:- Diabetic nephropathy and other complications
whose age ranged between 30-50 years old.
Inclusion criteria
Patients were free from infectious disease.
Patients were free from inflammatory disease.
Patients were free from liver disease.
Patients were free from malignancy.
All were non-smokers.
Exclusion criteria
Patients with active urinary tract infection.
Patients with renal disease other than diabetic nephropathy;
neoplastic disorders; severe liver dysfunction; active or chronic
infection or inflammatory disorders.
Pregnancy.
Patients with a recent (i.e., within 6 months) history of acute
myocardial infarction, stroke, or occlusive peripheral vascular
disease.
All patients were subjected to the following:
Collection of demographic data as required in the attached
sheet including age, occupation, anthropometric measurements
of height, weight, waist circumference, and history of disease.
Collection of morning urine samples in vacutaniner cup and
also collection of 10 venous blood samples from the overnight
fasted 5 ml blood were collected on plane tubes and other 5 ml
blood were collected on EDTA tubes by vacutaniner system
under complete aseptic conditions and HbAIC first done and the
samples centrifuged for 10 min at 2,500 g within 30 min
separated serum and plasma were stored at 20°C for the
measurement of measurement of plasma a-klotho
concentration, serum fasting glucose concentration, serum
Creatinine, serum urea n, serum uric acid, serum potassium k,
serum sodium Na serum phosphorus, Albumin: Creatinine Ratio,
GFR concentration, serum cholesterol and serum triglyceride.
AST, ALT, T.BIL, D.BIL ALB, TP, GLB and A/G ratio.
The collected data was revised, coded, tabulated and
introduced to a PC using Statistical package for Social Science
(SPSS 23). Data was presented and suitable analysis was done
according to the type of data obtained for each parameter. The
following tests were used:
Descriptive statistics
Mean, Standard deviation (± SD) and range for parametric
numerical data, while Median and Interquartile range (IQR) for
non-parametric numerical data. Frequency and percentage of
non- numerical data.
Analytical statistics
ANOVA test of significance was used when comparing
between means of more than two groups.
Post-hoc test after ANOVA for significance between each two
groups.
Chi-Square test was used to examine the relationship
between two qualitative variables.
Fisher’s exact test was used to examine the relationship
between two qualitative variables when the expected count is
less than 5 in more than 20% of cells.
Correlation analysis (using Pearson's method) to assess the
strength of association between two quantitative variables. The
correlation coefficient denoted symbolically "r" defines the
strength (magnitude) and direction (positive or negative) of the
linear relationship between two variables.
Results
A total of 125 subjects were enrolled in this study; their mean
age was 55.8610.4 years (range, 24–82 years), and there were
71 men and 54 women. Age, BMI, Duration of D.M, F.B.G, C.P.A, HBAIC, S. Creatinine, S. urea, UR.A, Na, ACR, GFR, Cholesterol,
Triglycerides, HDL, LDL, AST, ALT, ALB, T.BIL, and D.BIL were
significantly higher in diabetic patients than non-diabetic
control. Meanwhile, K, Ph, T.P, AG ratio and KL were significantly
lower in diabetic patients than in non-diabetic controls. Other
parameters did not differ significantly between the diabetes
group and non-diabetic controls.
Age, BMI, Duration of D.M, F.B.G, C.P.A, HBAIC, S. Creatinine,
S. urea, UR.A, Na, ACR, GFR, Cholesterol, Triglycerides, HDL, LDL,
AST, ALT, ALB, T.BIL, D.BIL, K, Ph, T.P, AG ratio and KL were
significantly different between four groups. Plasma a-klotho was
significantly correlated with haemoglobin A1C, potassium, GFR,
Albumin, TP and GLB. Meanwhile, plasma a-klotho was
negatively correlated with duration of DM, CR, Urea, UR.A, Na,
phosphorus, ACR, Chol, TG, LDL, AST, ALT, T.BIL, and D.BIL.
However, there were no significant correlations between plasma
a-klotho and FBS, HDL and A/G ratio.
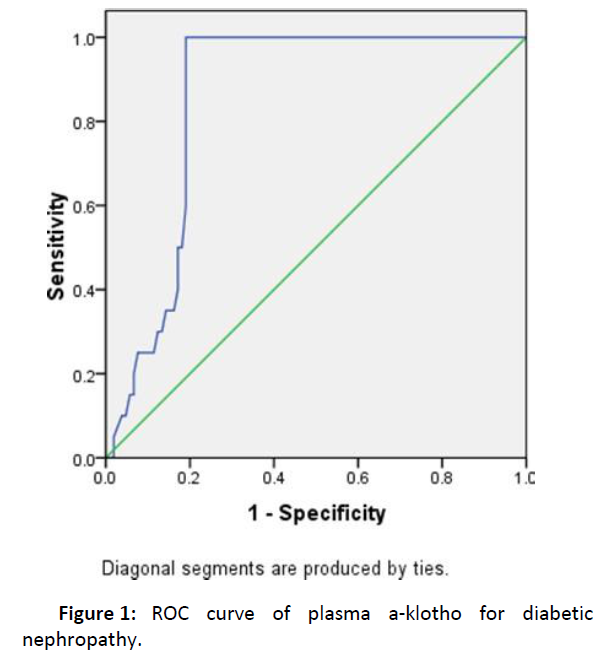
Figure 1 show that at cut- off level ≥ 2.6, plasma a-klotho had
95% sensitivity and 81% specificity for diagnosing diabetic
nephropathy.
Figure 1: ROC curve of plasma a-klotho for diabetic nephropathy.
Discussion
α-Klotho is a single-pass transmembrane protein that is highly
expressed in the kidneys and is known to act as a co-receptor for
fibroblast growth factor-23. 19Circulating soluble α-klotho can
be generated directly by the alterative splicing of the α-klotho
transcript or the extracellular domain of membrane α-klotho can
be released from membrane-anchored α-klotho on the cell
surface .
Unlike membrane α- klotho, which functions as a co- receptor
for fibroblast growth factor- 23, soluble α-klotho acts as a
hormonal factor and plays important roles in anti-aging, antioxidation,
ion transport modulation, and Wnt signalling?
Previous studies aiming to clarify the role of α-klotho as a
potential biomarker of kidney injury show the blood and urinary
concentrations of α- klotho decrease early in the course of
chronic kidney disease in mice with experimentally induced
chronic kidney disease as well as humans.20 As blood α-klotho
concentration was found to be linearly associated with eGFR in
previous studies , plasma α-klotho was associated with the eGFR
in the present study (r=0.888, p<0.001).
Meanwhile, little is known about circulating α-klotho levels in
diabetes- related nephropathy. Recent studies in patients with
diabetes report conflicting data. One study found serum α-
klotho level was not significantly different between patients
with diabetes without nephropathy and non-diabetic controls.
The findings that both exogenous soluble α-klotho
administration and overexpression of membranous α-klotho in
kidney cell culture suppress NF-κB activation and subsequent
inflammatory cytokine production in the response to TNF-α
stimulation suggest α-klotho serves as an anti- inflammatory
modulator. Therefore, preventing deceases in α-klotho and α-
klotho supplementation are potential novel therapeutic
strategies for early diabetic nephropathy. In multiple
experimental models of chronic kidney disease, the replacement
or endogenous up regulation of α-klotho protects the kidneys
from renal insults, preserves kidney function, and suppresses
renal fibrosis. Thus, α-klotho is a highly promising candidate
early biomarker as well as a novel therapeutic agent for chronic
kidney disease. Blood α-klotho concentrations can easily be
checked and used to assess the development of diabetic
nephropathy prior to the onset of micro albuminuria, which is
the earliest sign of diabetic nephropathy in clinical settings. To
best of our knowledge, this study is the first study to assess
validity of α-klotho for diagnosing diabetic nephropathy.
Conclusion
In conclusion, the results of the present study suggest plasma
and urinary a-klotho may be the early markers for predicting
renal injury in patients with type 2 diabetes and we need to do
long-term prospective study in order to elucidate the role of aklotho
in the pathophysiological mechanisms of the
development and progression of albuminuria in type 2 diabetes.
References
- Hostetter TH (2001) Prevention of end-stage renal disease due to type 2 diabetes. N Engl J Med 345: 910–912.
- Arora MK, Singh UK (2103) Molecular mechanisms in the pathogenesis of diabetic nephropathy: an update. Vascul Pharmacol 58: 259–271.
- Halimi JM (2012) The emerging concept of chronic kidney disease without clinical proteinuria in diabetic patients. Diabetes Metab 38: 291–297.
- Tramonti G, Kanwar YS (2013) Review and discussion of tubular biomarkers in the diagnosis and management of diabetic nephropathy. Endocrine 43: 494–503.
- Kramer HJ, Nguyen QD, Curhan G (2003) Renal insufficiency in the absence of albuminuria and retinopathy among adults with type 2 diabetes mellitus. JAMA 289: 3273–3277.
- MacIsaac RJ, Tsalamandris C, Panagiotopoulos S, Smith JT, McNeil JK,et.al. (2004) Non albuminuric renal insufficiency in type 2 diabetes. Diabetes Care 27: 195–200.