Keywords
Clinical practice guidelines; Randomized
controlled trials; Fragility index; Fragility quotient; Risk of
bias; Bowel preparation; Colonoscopy; Colon cancer
Key Points
• What is already known about the subject?
The fragility index demonstrates the number of events that
would need to change to alter the significance of a reported result. Several studies have already suggested baseline
acceptable values for fragility index.
• What are the new findings?
Our investigation demonstrated that RCTs found in Bowel
Preparation Guidelines have moderate fragility and high risk of
bias, particularly in regard to selective reporting of outcomes.
• How might it impact clinical practice in the foreseeable
future?
Reporting study fragility and preemptive assessment of bias
would allow practitioners to assess the weight of results in
RCTs and thus have more confidence in guideline
recommendations.
Introduction
Clinical practice guidelines (CPGs) are essential to evidencebased
clinical decision making. CPG recommendations are
often based on a systematic survey of the literature and are
graded on the strength of the recommendation and quality of
evidence. Typically, the strongest recommendations are
supported by methodologically robust evidence, like that from
randomized controlled trials (RCTs). However, not all RCTs are
equally robust. The strength of CPG recommendations
depends on the robustness of the RCTs used as evidence.
In the field of gastroenterology, colonoscopy is a key step in
the evaluation of colorectal carcinoma (CRC). CRC is the
second most commonly diagnosed cancer in women and the
third in men worldwide with an estimated 1.4 million new
cases each year [1]. The incidence of CRC has declined in
recent years within the US, likely due to increased screening
and new technologies. To continue this trend, evidence-based
guidelines with robust underlying evidence are needed.
The American Gastroenterological Association (AGA)
guidelines for bowel cleansing for colonoscopy provide 30
evidence-based recommendations to endoscopists [2]. In
accordance with the Grades of Recommendation Assessment,
Development and Evaluation (GRADE) system, authors of CPGs
and the GRADE system recognized RCTs as the highest level of
evidence, along with systematic reviews [3]. Concerning,
however, is the recent demonstration that the results of RCTs
are often fragile [4,5]. The application of the fragility index (FI) and fragility quotient (FQ) has shown that the statistical
significance of clinical trial results often relies on relatively few
patient events. To calculate the FI, one must iteratively remove
one patient event from a study arm and add it to the other
until the statistical significance of a clinical endpoint is
nullified. The FQ contextualizes the FI by dividing the FI by the
RCTs sample size. RCTs with fragile outcomes are less
trustworthy, and the findings may be irreproducible.
Furthermore, bias may compromise the trustworthiness of
RCT outcomes. To ensure trustworthy findings and improve
transparency in RCTs, the Cochrane Risk of Bias tool 2.0 was
developed [6]. The tool was designed to objectively analyze
study elements to identify potential sources of bias. This tool
assesses bias over five domains: randomization, deviation
from intended interventions, accounting for missing data,
measurements, and selectively reporting results. Each of the
five domains requires equal attention to mitigate bias in RCTs.
Our primary objective in this study is to examine the fragility
of the RCT endpoints that underpin the AGA guideline on
bowel cleansing for colonoscopy using the FI and FQ. Our
secondary objective was to assess the methodological quality
of the RCTs using the Cochrane Risk of Bias Tool 2.0.
Methods
Identification of studies
Using the AGA CPGs, we identified all RCTs cited within the
document. Using Pubmed and Google, we located the articles
eligible for review. Two investigators (C.C., C.W.) screened,
reviewed, and included all studies that met eligibility criteria.
Eligibility criteria
To be evaluated for fragility, studies must possess the
following three characteristics: randomization between two
patient groups in a 1:1 distribution, a parallel two group
design, and at least one statistically significant dichotomous
endpoint.
Data collection
Data was extracted from the articles using a Google form.
Data collected includes the sample size for each group,
number of patients lost to follow-up, the reported outcome,
the rates of the reported outcome within the groups,
statistical significance value, and the method for determining
statistical significance. We prioritized primary outcomes, but
included the secondary outcome if it met the criteria for our FI
analysis and the primary outcome did not.
Fragility index and fragility index quotient
Fragility index was calculated from the extracted outcome
data using an online calculator [7]. This calculator requires the
number of events in each arm and the respective sample size.
It first calculates a p-value based on the event rates using
Fisher’s exact test. If the p-value is statistically significant, the calculator iteratively subtracts and adds one patient event at a
time until the p-value is non-significant. If the p-value is not
statistically significant, the FI is 0. In such a case, the original
statistically significant p-value was often obtained from a test
other than Fisher’s exact. The FQ was then calculated from the
FI and total sample size (Table 1).
| Randomized controlled trial (Author) |
Outcome |
Sample Size |
Patients Lost to Follow Up |
Intervention Group |
Control Group |
Outcome: Intervention |
Outcome: Control |
Fragility Index |
Fragility Quotient |
| Abdul-Baki, et al. |
"Satisfactory" bowel cleanliness |
382 |
0 |
199 |
183 |
177 |
78 |
72 |
0.188 |
| Abut, et al. |
"Satisfactory" or "unsatisfactory" bowel cleanliness |
80 |
4 |
41 |
39 |
41 |
23 |
12 |
0.15 |
| Amato, et al. |
Presence of moderate to severe bowel pain |
296 |
0 |
151 |
145 |
9 |
22 |
3 |
0.01 |
| Arezzo |
"Good" or "Medium"bowel preparations |
200 |
0 |
100 |
100 |
77 |
95 |
9 |
0.045 |
| Berkelhammer, et al. |
"Good" or "Fair" Bowel Cleanliness |
300 |
0 |
140 |
160 |
132 |
117 |
24 |
0.08 |
| Cesaro, et al. |
"Excellent" Bowel Cleanliness |
99 |
0 |
50 |
49 |
35 |
24 |
1 |
0.01 |
| Cohen et al. |
"Excellent" bowel cleanliness |
181 |
0 |
143 |
138 |
93 |
55 |
19 |
0.068 |
| Cohen, et al. |
"Excellent" bowel preparation |
107 |
3 |
52 |
55 |
36 |
21 |
7 |
0.065 |
| Delegge, et al. |
"Satisfactory" bowel preparations |
506 |
0 |
284 |
222 |
89 |
82 |
0 |
0 |
| El-Baba, et al. |
"Excellent" bowel preparations |
62 |
0 |
36 |
26 |
18 |
5 |
2 |
0.032 |
Haappama
ki, et al. |
"Poor" or"inadeqaute" bowel cleanliness |
399 |
85 |
203 |
196 |
22 |
8 |
3 |
0.008 |
| Law, et al. |
"Excellent" or"good" bowel cleanliness |
207 |
13 |
101 |
106 |
77 |
57 |
11 |
0.053 |
| Lee, et al. |
Number of patients with an unsatisfactory bowel cleansing |
104 |
0 |
51 |
53 |
23 |
42 |
8 |
0.077 |
| Malik, et al. |
"Adequate" Bowel Cleanliness |
81 |
0 |
40 |
41 |
39 |
31 |
3 |
0.037 |
| Marmo, et al. 2010 |
"Satisfactory" Bowel Cleanliness |
870 |
27 |
437 |
433 |
327 |
141 |
157 |
0.18 |
| Park, et al. |
"Excellent" Bowel Cleanliness rated as excellent on Aronchik scale |
152 |
38 |
73 |
79 |
55 |
40 |
8 |
0.053 |
| Parra-Blanco, et al. |
Failed bowel preparations |
177 |
20 |
89 |
88 |
11 |
2 |
2 |
0.011 |
| Picchio, et al. |
"Adequate" Bowel cleanliness |
182 |
0 |
94 |
88 |
67 |
46 |
5 |
0.027 |
| Radaelli, et al. |
"Excellent" or "good"bowel cleansing |
383 |
5 |
191 |
192 |
173 |
153 |
8 |
0.021 |
| Repici, et al. |
Bowel Cleanliness with BPPP scores of 6 or greater. |
377 |
28 |
187 |
190 |
148 |
133 |
1 |
0.003 |
| Rex, et al. |
"Excellent" bowel preparations |
130 |
7 |
63 |
67 |
45 |
23 |
14 |
0.108 |
| Rex, et al. |
"Satisfactory" bowel cleanliness |
601 |
2 |
304 |
297 |
256 |
221 |
11 |
0.018 |
| Samarasena, et al. |
"Excellent" bowel preparations |
222 |
0 |
105 |
117 |
83 |
30 |
48 |
0.216 |
| Saunders,et al. |
"Good" or"Excellent" bowelcleanliness |
89 |
0 |
44 |
45 |
34 |
43 |
3 |
0.034 |
| Sharara, et al. |
"Excellent" or better bowel cleanliness |
99 |
0 |
49 |
50 |
31 |
17 |
5 |
0.051 |
| Tae, et al. |
Patients receiving agood (>5) rating forbowel prep on BBPSscale. |
200 |
5 |
102 |
98 |
95 |
80 |
3 |
0.015 |
| Tajika, et al. |
Scores of excellent/good/fair vs poor/inadequate |
244 |
0 |
119 |
125 |
39 |
24 |
3 |
0.012 |
| Vradelis, et al. 2009 |
Adequate" colon cleansing |
342 |
3 |
182 |
160 |
148 |
108 |
8 |
0.023 |
| Young, et al. |
"Good" bowel cleanliness |
323 |
0 |
169 |
154 |
144 |
105 |
13 |
0.04 |
| Zwas, et al. |
Presence of aphthoid lesions |
97 |
0 |
53 |
44 |
13 |
1 |
3 |
0.031 |
Table 1: Fragility assessment.
Risk of bias assessment
We devised a Google Form based on the Cochrane Risk of
Bias Tool 2.0 as a framework to assist in data collection. This
tool examines the study design of randomized controlled trials
and the way data is handled within those trials to determine if
a potential for bias exists. Specific sources of bias are assessed
over five domains: randomization, deviation from intended
interventions, accounting for missing data, measurements, and
selectively reporting results.
The tool provides a flowchart for each of the domains and
one of three grades is given for each of the domains
depending on the available information. The grades are as
follows: low risk, some concerns, and high risk of bias. Two
investigators (C.C., B.H.) reviewed articles independently
before sharing notes. Conflicts were resolved by consensus.
We assigned an overall grade for each of the RCTs after
comparing the individual grades for the five domains. A study
was considered to have a low risk of bias only if given low risk
grades across all five domains. If the study has one domain
that has some concern for bias but all other domains were low
risk, then the study was classified as having some concern for
bias. A final grade of high risk of bias was assigned to any study
with 2 or more some concerns domains or any study with at
least one high risk domain. Risk of bias assessments were
limited to the 30 studies assessed for fragility.
Graphical representation of the data
Graphical representation of the data was done using R
Studio software and the ggplot2 package [8,9].
Results
Study selection
The AGA’s guideline on bowel preparation prior to
colonoscopy contained 253 references. Of these, 133 were
RCTs. Thirty met inclusion criteria and were included in the
final analysis of fragility (Figure 1). Of these, 27 (90.0%)
primary outcomes and 3 (10.0%) secondary outcomes were
recorded.
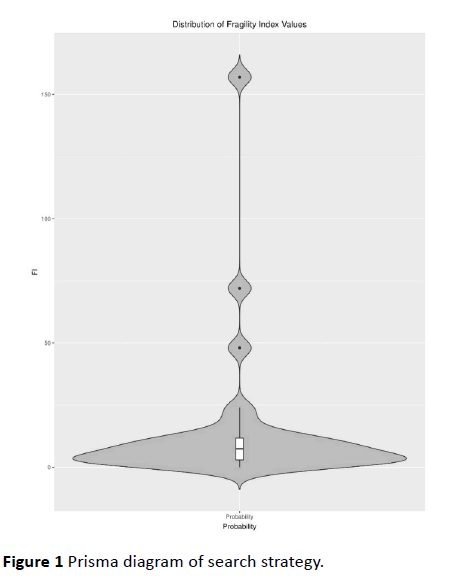
Figure 1 Prisma diagram of search strategy.
Overall fragility index and fragility quotient
The median FI for the 30 trials was 7.5 events (IQR 3 -
11.75). One study (1/30, 3.3%) had a FI of 0, indicating that the
reported outcome was not significant according to Fisher's
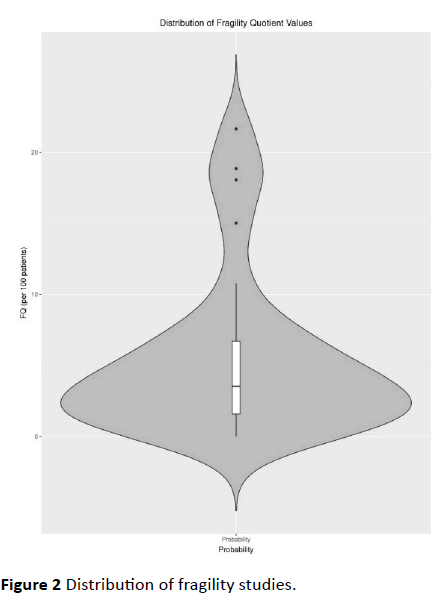
exact test. The median FQ for the trials was 0.035, meaning 3.5
per 100 patients (IQR 1.5 - 6.7 per 100) were needed to nullify
the statistical significance of the RCT endpoints in our study.
The median sample size was 200 (104.7 - 337.2). The number lost to follow up was greater than or equal to the FI in 5 trials
(5/30, 16.7%). Complete FI and FQ results can be found in
Appendix 1 (Figure 2).
Figure 2 Distribution of fragility studies.
Risk of bias assessment
Our analysis of the trials revealed 16 (53.3%) at high risk, 2
(6.7%) with some concerns for bias, and 12 (40.0%) at low risk
of bias. High risk of bias most commonly came from selection
in the reported result (30.0%) followed by deviations from
expected interventions (20.0%). Cumulative risk of bias data is
shown in Table 2 with assessments for each RCT included in
supplemental Appendix 2.
| Risk of Bias Category (n=30) |
Low Risk |
Some Concerns |
High Risk |
| Arising from the Randomization process |
21 (70%) |
8 (26.7%) |
1 (3.3%) |
| Due to deviations from intended interventions |
24 (80%) |
0 |
6 (20%) |
| Due to missing outcome data |
30 (100%) |
0 |
0 |
| Measurement of the Outcome |
29 (96.7%) |
0 |
1 (3.3%) |
| Selection of the reported Result |
21 (70%) |
0 |
9 (30%) |
| Overall bias of trial |
12 (40%) |
2 (6.7%) |
16 (53.3%) |
Table 2 General risk of bias assessments.
Discussion
Overall, our results show that RCTs in AGA Bowel Cleansing
Guidelines have moderate fragility and a relatively high risk of
bias, most often due to biased reporting of outcomes and bias
arising from differences in care provided between treatment
groups. Regarding fragility, the reported median FI across all 30
RCTs was 7.5 events. This implies that a median of only 7-8
events would be required to reverse the significance of a
certain result. These results are consistent with previous
fragility studies, which reported median fragility indices of 7
and 8 [4,5].
The FIs calculated in our investigation, in conjunction with
FQs and risk of bias, raise questions about the robustness of
the evidence underpinning the AGA’s Bowel Cleansing
Guidelines. Furthermore, 5 of the 30 (16.7%) trials reported a
number lost to follow up greater than or equal to the fragility
index. These lost participants may have been able to provide
data that affected the occurrence of study outcomes thus
altering the significance of reported results. Previous FI
investigations have emphasized the effect that patients lost to
follow up have on trial results and suggested that studies with
FIs lower than the number of participants lost to follow up are
more fragile [4,10]. Our findings highlight the utility of the FI
and FQ by guideline panels who may wish to investigate the
robustness of statistically significant endpoints. If guideline
developers report FI and FQ values alongside p-values, physician readers would be better equipped to make confident
clinical decisions.
For guideline developers to implement and for physicians to
understand the FI and FQ, they must understand the
relationship between the two measures. For example, within
our analysis, the FI ranged from 0 to 157. One may assume
that the larger the FI, the more robust the trial outcome, since
more patient events are necessary to nullify the statistical
significance. However, differences in characteristics of each
study affect the magnitude of FI. To contextualize the
magnitude of the FI, one must calculate the FQ — the FI divided by the trial sample size [11]. Consider an example from
our analysis; one RCT outcome had an FI of 157, while another
had an FI of 48. But, the first trial had a FQ of 18 and the
second had an FQ of 21. So, in fact, fewer patients per 100
were needed to nullify the statistical significance of the first
trial, despite the magnitude of its FI.
Our results reveal several studies with low FI values, with
one in particular having an FI of 0. The FI calculator begins by
calculating a p-value using Fisher’s exact test. For there to be
an FI of zero, Fisher’s exact test would have to yield a nonsignificant
p-value. Therefore, the original p-value was
significant due to choice of statistical test and likely
irreproducible. For such an outcome to underpin CPG
recommendations is concerning, again supporting the utility of
the FI and FQ as standard measures for RCT authors and CPG
developers. We assessed the FQ for the 5 studies with an FI
less than 3 (the lower bound of the median FI IQR). All of these
studies had an FQ less than or equal to 3 in 100 patients. In
comparison, the studies with FI greater than the upper bound
of the IQR had FQ values around 20 per 100 patients. While it
is important to note that the studies with higher FQs require
roughly 7x as many event changes to alter the significance of
the reported result, few studies have reported FQ values.
Therefore, normal FQ values across RCTs have not been clearly
established.
Our investigation also revealed a high risk of bias in a large
portion of RCTs. Following evaluation by the Cochrane Risk of
Bias 2.0 Tool, 16 of the 30 RCTs (53.3%) were found to be at
high risk of bias. High risk of bias was most frequently due to
the selection in the reported result (30.0%) followed by
deviations from expected interventions (20.0%). Selective
reporting bias in research has been shown to contribute to
misleading information and recommendations that are based
off faulty evidence [12]. Preconceived notions or an interest in
showing benefit of a particular intervention may lead
researchers to selectively report on outcome measurements
that are favorable to the intervention [6]. Our assessment of
risk of bias emphasizes the importance of carefully examining
RCTs for bias before clinical trial interventions are
implemented into practice.
The main limitation to our study is that the results may not
be generalizable to all 133 RCTs from the guideline. The
inclusion criteria for the fragility arm allowed evaluation of
only 30 of 133 RCTs. Due to the nature of the fragility analysis,
this limitation was unavoidable. Despite this limitation, our
results show that analyzing studies for fragility and ROB will
help authors provide the best recommendations from the
available data.
Conclusion
Making clinical decisions affects patients and as such it is
crucial that practitioners have confidence in the
recommendations in clinical practice guidelines. It is
understood that readers should not rely on any one particular
value, however we advocate for an exhaustive approach when
evaluating RCTs and their contribution to CPGs. Inclusion of the FI, FQ, and ROB for RCTs that provide the evidence for
CPGs would improve the confidence in the data and
recommendations. Based on our investigation, it is clear that
RCTs in the ACG Bowel Preparation guidelines exhibit
moderate fragility and a relatively high risk of bias and this
should be taken into account when implementing these
recommendations into clinical practice.
References
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, et al. (2015) Global cancer statistics, 2012. CA: CA Cancer J Clin 65: 87-108.
- Johnson DA, Barkun AN, Cohen LB, Dominitz JA, Kaltenbach T, et al. (2014) Optimizing adequacy of bowel cleansing for colonoscopy: recommendations from the US multi-society task force on colorectal cancer. Am J Gastroenterol 109: 1528.
- https://training.cochrane.org/path/grade-approach-evaluating-quality-evidence-pathway.
- Matics TJ, Khan N, Jani P, Kane JM (2017) The fragility index in a cohort of pediatric randomized controlled trials. J Clin Med 6: 79.
- Walsh M, Srinathan SK, McAuley DF, Mrkobrada M, Levine O, et al. (2014) The statistical significance of randomized controlled trial results is frequently fragile: A case for a fragility index. J Clin Epidemiol 67: 622-8.
- Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, et al. (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. Bmj 343: d5928.
- Kane SP (2017) Fragility Index Calculator. 2017. https://clincalc.com/Stats/FragilityIndex.aspx.
- R-Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
- Wickham H (2009) ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York, USA.
- Mazzinari G, Ball L, Neto AS, Errando CL, Dondorp AM, et al. (2018) The fragility of statistically significant findings in randomised controlled anaesthesiology trials: A systematic search of the medical literature. Br J Anaesth.
- Ahmed W, Fowler RA, McCredie VA (2016) Does sample size matter when interpreting the fragility index? Crit. Care Med 44: 1142-3.
- Kirkham JJ, Dwan KM, Altman DG, Gamble C, Dodd S, et al. (2010) The impact of outcome reporting bias in randomized controlled trials on a cohort of systematic reviews. BMJ 340: 365.