Research Article - (2016) Volume 6, Issue 6
Kumari A1*, Kumar R1, Maurya S2 and Pandey PK1
1University Department of Botany, Ranchi University, Ranchi, India
2ICAR Research Complex for Eastern Region, Research Centre, Plandu, Ranchi, India
Corresponding Author:
Anjali Kumari
University Department of Botany
Ranchi University, Ranchi
India
Tel: +919704412434
E-mail: anjalikumariharp@gmail.com
Received date: October 21, 2016; Accepted date: November 23, 2016; Published date: November 30, 2016
Copyright: © 2016 Kumari A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Trichoderma spp. is the most promising and effective bioagents against several soil borne plant pathogenic fungi. In present study, 26 isolates of Trichoderma were isolated from soil of Jharkhand and screened for their antagonistic and antibiosis efficacy against Rhizoctonia solani by dual culture. Result indicated that the antagonistic potential of 26 isolates of Trichoderma spp. against R. solani were varied which inhibited R. solani ranges 33-54%. Among isolates of Trichoderma, seven isolates showed strong antagonistic potential which inhibited >50% mycelial growth of R. solani, viz., RCT1 (53.71%) followed by RCT22 (52.6%), RCT3 (51.85%), RCT7 (51.11%), RCT10 (50.37%), RCT 8 (50%) and RCT14 (50%). Moreover, seventeen (17) isolates were also showed inhibitory but their antagonistic potential <50% of the mycelial growth while two isolates (RCT12 and RCT17) showed <40% mycelial growth. These potential isolates of Trichoderma may be further exploited as biocontrol agent against R. solani as well as other Soilborne phytopathogenic fungi.
Keywords
Biological control; Trichoderma; Antagonistic potential; Antibiosis; Inhibition
Introduction
Rhizoctonia solani is an important soilborne plant pathogenic fungus which distributed worldwide which has wide host range [1-3]. It causes numerous diseases in crop plants viz. dampingoff, black spot and root rot diseases [4]. R. solani is a fast growing necrotrophic and sclerotial fungus which survives in the soil as hard, resistant sclerotial bodies [5]. Management of R. solani by chemical fungicides was expensive and tedious. No effective fungicides are available in the market. Moreover the negative effect of chemical fungicides causes phytotoxicity and environmental pollution [6]. Intensified use of fungicides has resulted in the accumulation of toxic compounds potentially hazardous to humans and environment also in the build-up of resistance of pathogens [7]. In order to tackle these national and global problems, alternatives of chemical control are investigated by the use of antagonistic microbes [8]. In this regard biological control offers an alternative solution for long term sustainability and effective management of soil borne diseases [9,10]. Among the microbes, Trichoderma spp. is a common saprophytic filamentous fungi which habitat in soil and rhizospheric soil. It acts as a bio control agent against various plant pathogenic causes several diseases in mono and dicotyledonous crop plants [11-13].
The effectiveness of biocontrol agents depends on several parameters,that include specific pathogen, soil texture, water content, pH, temperature and crop history [14,15], therefore their application should consider the environmental stress that could affect their ability to maintain their biocontrol capacity. It has very wide range in modes of action viz. Plant growth promotion, mycoparasitism, antibiosis, competition for nutrients and space, tolerance to stress through enhanced root and plant development [16-19]. Looking these potential systemic resistances Trichoderma against soil borne phytopathogen, the present experiment were designed to explore novel and potential isolates of Trichoderma spp. from the soil of Jharkhand in the biocontrol of R. solani and other soilborne phytopathogens.
Material and Method
The present experiments were conducted in ICAR, RCER, Research Centre, Plandu, and Ranchi, India.
Collection of soil sample
Soil samples (pH-acidic, N-medium, P-low, K-Sufficient, OClow) were collected from different localities of Agricultural and vegetable growing areas of Ranchi district of Jharkhand, at the depth of 5-7 cm of soil surface The composite soil samples were collected from a particular field in the polyethylene bag and labeled carefully (Table 1).
| Sl. No | Code | Place of collection (Blocks of Ranchi dist.) | Research centre code | Soil sample detail (Rhizospheric soil) |
|---|---|---|---|---|
| 1. | T1 | Namkum | RC T1 | Sapota |
| 2. | T2 | Namkum | RC T2 | Litchi |
| 3. | T3 | Mander | RC T3 | Mango |
| 4. | T4 | Jonha | RC T4 | Guava |
| 5. | T5 | Nagri | RC T5 | Ladyfinger |
| 6. | T6 | Bero | RC T6 | Tomato |
| 7. | T7 | Ratu | RC T7 | Onoin |
| 8. | T8 | Burmu | RC T8 | Pea |
| 9. | T9 | French bean | RC T9 | Angara |
| 10. | T10 | Ormanjhi | RC T10 | Pea |
| 11. | T11 | Lapung | RC T11 | Wheat |
| 12. | T12 | Tamar | RC T12 | Brinjal |
| 13. | T13 | Bero | RC T13 | Potato |
| 14. | T14 | Chanho | RC T14 | Cabbage |
| 15. | T15 | Namkum | RC T15 | Dioscoria |
| 16. | T16 | Namkum | RC T16 | Elephant yam |
| 17. | T17 | Namkum | RC T17 | Tephrosia |
| 18. | T18 | Jonha | RC T18 | Gram |
| 19. | T19 | Namkum | RC T19 | Pea |
| 20. | T20 | Namkum | RC T20 | Mango |
| 21. | T21 | Kanke | RC T21 | Cabbage |
| 22. | T22 | Kanke | RC T22 | Beat |
| 23. | T23 | Pithoria | RC T23 | Potato |
| 24. | T24 | Angara | RC T24 | Paddy |
| 25. | T25 | Silli | RC T25 | Mango |
| 26. | T26 | Namkum | RC T26 | Litchi |
Table 1: Collection of soil samples for isolation of Trichoderma from locality of Ranchi, Jharkhand.
Isolation of Trichoderma from soil
Isolation of different isolates of Trichoderma was made by the collected soil serial dilution technique of the soil sample. One (1) ml of 10-3 dilution was poured on to Trichoderma selective Medium (MgSO4: 0.20 g, KH2PO4: 0.90 g, NH4NO3: 1.0 g, KCl: 0.15 g, Glucose: 3.0 g, PCNB: 20 g, Rose Bengal: 0.15 g, Chloramphenicol: 0.25 g, Agar-agar: 15 g, Metalaxyl: 30 g, Distilled water: 1 L) for selective isolation of Trichoderma and after the appearance of the colonies of Trichoderma on Petri dishes purified by hyphal tip isolation techniques. Trichoderma spp. was identified, picked on the basis of their morphological and microscopic characteristics. The purified and identified cultures of Trichoderma spp. were maintained on Potato Dextrose Agar (PDA) medium and stored at 4°C for further experimentation (Table 2).
| Isolates | Periodical growth in(mm/day) | |||||||
|---|---|---|---|---|---|---|---|---|
| 24Hrs |
48Hrs |
72Hrs | 96Hrs | |||||
| T | R | T | R | T | R | T | R | |
| RC T1 | 29.33 ± 0.66 | 25.00 ± 0.57 | 48.33 ± 0.88 | 39.66 ± 0.88 | 49.00 ± 0.57 | 40.66 ± 1.45 | 49.00 ± 0.57 | 41.66 ± 0.88 |
| RC T2 | 28.00 ± 1.15 | 23.00 ± 1.52 | 41.00 ± 0.57 | 48.66 ± 0.66 | 44.66 ± 0.88 | 49.33 ± 0.66 | 46.00 ± 0.00 | 47.33 ± 1.76 |
| RC T3 | 27.00 ± 0.57 | 29.66 ± 0.33 | 44.33 ± 0.33 | 47.00 ± 0.00 | 45.33 ± 0.33 | 48.00 ± 0.00 | 47.66 ± 0.33 | 43.33 ± 0.88 |
| RC T4 | 24.00 ± 1.15 | 26.66 ± 0.33 | 45.33 ± 0.33 | 45.33 ± 0.33 | 46.33 ± 0.33 | 47.00 ± 0.57 | 45.33 ± 1.33 | 47.00 ± 0.57 |
| RC T5 | 28.66 ± 0.66 | 27.33 ± 0.33 | 48.00 ± 1.52 | 43.66 ± 0.33 | 46.66 ± 1.66 | 46.33 ± 0.66 | 48.33 ± 1.33 | 46.33 ± 0.66 |
| RC T6 | 27.66 ± 0.33 | 28.00 ± 1.52 | 43.66 ± 0.88 | 45.00 ± 0.00 | 44.66 ± 0.33 | 46.33 ± 0.66 | 46.33 ± 0.33 | 45.33 ± 0.88 |
| RC T7 | 25.66 ± 0.33 | 30.33 ± 0.33 | 44.66 ± 0.33 | 44.00 ± 0.57 | 47.33 ± 0.88 | 42.66 ± 0.88 | 46.00 ± 0.57 | 44.00 ± 0.57 |
| RC T8 | 30.00 ± 0.00 | 33.00 ± 1.00 | 46.33 ± 0.66 | 45.00 ± 0.00 | 46.66 ± 0.33 | 48.33 ± 0.66 | 44.66 ± 0.88 | 45.00 ± 0.57 |
| RC T9 | 25.00 ± 1.00 | 34.00 ± 0.57 | 42.33 ± 1.33 | 47.00 ± 1.00 | 44.66 ± 0.66 | 49.00 ± 1.52 | 46.66 ± 0.66 | 47.33 ± 1.76 |
| RC T10 | 27.33 ± 1.15 | 32.66 ± 0.66 | 45.66 ± 0.33 | 43.00 ± 1.00 | 45.66 ± 0.66 | 47.00 ± 1.00 | 47.33 ± 3.33 | 44.66 ± 2.33 |
| RC T11 | 27.33 ± 0.88 | 33.33 ± 1.66 | 41.66 ± 1.20 | 48.33 ± 0.88 | 46.00 ± 2.08 | 49.33 ± 0.33 | 45.00 ± 2.51 | 51.33 ± 2.96 |
| RC T12 | 13.00 ± 1.52 | 34.00 ± 1.00 | 23.33 ± 1.76 | 55.00 ± 2.51 | 28.33 ± 1.20 | 64.00 ± 3.05 | 32.66 ± 1.76 | 60.33 ± 1.45 |
| RC T13 | 24.00 ± 1.73 | 33.33 ± 0.33 | 41.00 ± 1.20 | 46.00 ± 0.00 | 44.66 ± 1.33 | 48.00 ± 0.00 | 45.00 ± 2.51 | 48.00 ± 0.57 |
| RC T14 | 26.00 ± 0.57 | 33.00 ± 1.15 | 44.00 ± 0.57 | 44.66 ± 1.45 | 46.00 ± 1.15 | 47.66 ± 1.45 | 46.66 ± 1.66 | 45.00 ± 2.51 |
| RC T15 | 25.33 ± 0.33 | 36.33 ± 0.88 | 47.33 ± 1.20 | 46.66 ± 0.88 | 47.66 ± 1.45 | 46.66 ± 0.33 | 48.00 ± 2.08 | 46.33 ± 0.66 |
| RC T16 | 26.00 ± 2.08 | 34.66 ± 0.33 | 44.33 ± 2.08 | 48.33 ± 0.33 | 43.66 ± 0.88 | 47.66 ± 0.33 | 43.66 ± 0.33 | 45.66 ± 1.33 |
| RC T17 | 15.33 ± 1.20 | 23.33 ± 2.40 | 25.33 ± 1.33 | 59.33 ± 1.76 | 32.00 ± 0.57 | 59.00 ± 0.57 | 33.00 ± 0.57 | 59.66 ± 0.33 |
| RC T18 | 19.00 ± 1.00 | 34.33 ± 0.66 | 33.33 ± 2.18 | 49.66 ± 0.88 | 37.00 ± 0.00 | 53.33 ± 0.88 | 40.33 ± 0.88 | 53.00 ± 1.00 |
| RC T19 | 27.66 ± 0.33 | 34.33 ± 0.66 | 43.66 ± 1.33 | 45.66 ± 1.33 | 44.33 ± 0.33 | 47.33 ± 0.66 | 46.33 ± 0.66 | 46.00 ± 0.00 |
| RC T20 | 25.33 ± 0.33 | 35.00 ± 0.00 | 46.00 ± 2.00 | 49.00 ± 0.57 | 45.00 ± 0.57 | 49.66 ± 0.33 | 47.00 ± 1.00 | 47.66 ± 1.33 |
| RC T21 | 30.33 ± 0.33 | 23.66 ± 0.88 | 44.00 ± 0.57 | 45.33 ± 0.33 | 50.33 ± 0.33 | 46.00 ± 0.57 | 55.33 ± 0.66 | 46.33 ± 0.66 |
| RC T22 | 28.66 ± 0.88 | 28.33 ± 2.18 | 45.66 ± 0.33 | 45.33 ± 0.33 | 45.66 ± 0.66 | 47.00 ± 0.57 | 46.33 ± 1.66 | 42.66 ± 0.66 |
| RC T23 | 27.66 ± 0.33 | 29.33 ± 3.17 | 46.66 ± 0.33 | 46.00 ± 0.57 | 48.00 ± 0.00 | 47.00 ± 1.52 | 49.33 ± 0.66 | 47.33 ± 0.33 |
| RC T24 | 28.33 ± 0.33 | 31.00 ± 2.00 | 44.00 ± 0.00 | 45.66 ± 0.33 | 45.00 ± 0.57 | 47.66 ± 0.33 | 45.00 ± 0.00 | 45.66 ± 0.33 |
| RC T25 | 30.33 ± 0.33 | 35.00 ± 0.00 | 45.00 ± 0.00 | 44.66 ± 0.33 | 44.33 ± 0.33 | 46.66 ± 0.66 | 44.33 ± 0.33 | 46.66 ± 0.33 |
| RC T26 | 26.33 ± 0.33 | 32.33 ± 0.66 | 43.00 ± 0.57 | 45.00 ± 0.00 | 45.66 ± 0.33 | 45.33 ± 0.33 | 44.66 ± 0.66 | 45.66 ± 0.88 |
± standard error of mean, T= growth of Trichoderma isolates, R= growth of Rhizoctoniasolani
Table 2: Periodical growth of different isolates of Trichoderma and Rhizoctoniasolani in dual culture
Isolation of sclerotial fungus Rhizoctonia solani
Damping off diseased affected plants were collected from vegetable growing area of Ranchi, Jharkhand. Then after the collected sample were surface sterilized by dipping in 0.1% HgCl2 for 5-10 second followed by three subsequent washing with sterilized distilled water and then after they were placed in Petri plates containing Potato Dextrose Agar (PDA) medium and incubated at 25°C ± 2°C for 36-48 h. The culture were purified by hyphal tip isolation and maintained on PDA slants for further experimentation as well as visually appeared sclerotia were collected, wash and inoculated on the petridishes of containing PDA and after 48-72 h colonies appeared on the petridishes (Figure 1).
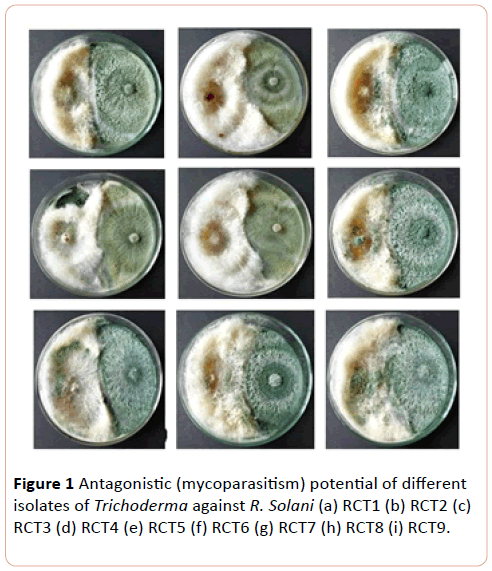
Figure 1: Antagonistic (mycoparasitism) potential of different isolates of Trichoderma against R. Solani (a) RCT1 (b) RCT2 (c) RCT3 (d) RCT4 (e) RCT5 (f) RCT6 (g) RCT7 (h) RCT8 (i) RCT9.
Identification of Trichoderma species
Fungal hyphae of Trichoderma species are septet, hyaline and smooth-walled which produces numerous conidiophores are highly branched. Normally, the branches will form at or near 90° with respect to the main branch. The typical conidiophore terminates with one or a few phialides that usually arising directly from the axis near the tip. Phialides, also known as conidiogenous cells, are typically enlarged in the middle like a flask-shape, and may be cylindrical or nearly sub-globose. Conidia are one-celled, and either ellipsoidal (3-5 × 2-4 μm, L/W ≥ 1.3) or globose (L/W< 1.3). They are typically light to dark green, or sometimes colourless, greyish or brownish which typically smooth surface.
Some strains are also produces chlamydospores which play important role in survival. They are normally found as thickwalled, enlarged vegetative cells with condensed cytoplasm which are unicellular, globose to subglobose chlamydospores are either formed within hyphae or at the hyphal tips [20-22].
Typically, they are colourless, pale yellowish or greenish. Morphological characters was compared with morphologically and molecularly identified strains T. asperellum (NAIMCCF- 03167) (Table 3).
| Strains | Growth of R. solaniin Treatment | Growth of R. solaniin control | Inhibition (%) |
|---|---|---|---|
| RC T1 | 41.66 ± 0.88 | 90.00 ± 0.00 | 53.71 |
| RC T2 | 47.33 ± 1.76 | 47.41 | |
| RC T3 | 43.33 ± 0.88 | 51.85 | |
| RC T4 | 47.00 ± 0.57 | 47.77 | |
| RC T5 | 46.33 ± 0.66 | 48.52 | |
| RC T6 | 45.33 ± 0.88 | 49.63 | |
| RC T7 | 44.00 ± 0.57 | 51.11 | |
| RC T8 | 45.00 ± 0.57 | 50.00 | |
| RC T9 | 47.33 ± 1.76 | 47.41 | |
| RC T10 | 44.66 ± 2.33 | 50.37 | |
| RC T11 | 51.33 ± 2.96 | 42.96 | |
| RC T12 | 60.33 ± 1.45 | 32.96 | |
| RC T13 | 48.00 ± 0.57 | 46.66 | |
| RC T14 | 45.00 ± 2.51 | 50 | |
| RC T15 | 46.33 ± 0.66 | 48.52 | |
| RC T16 | 45.66 ± 1.33 | 49.26 | |
| RC T17 | 59.66 ± 0.33 | 33.71 | |
| RC T18 | 53.00 ± 1.00 | 41.11 | |
| RC T19 | 46.00 ± 0.00 | 48.88 | |
| RC T20 | 47.66 ± 1.33 | 47.04 | |
| RC T21 | 46.33 ± 0.66 | 48.52 | |
| RC T22 | 42.66 ± 0.66 | 52.6 | |
| RC T23 | 47.33 ± 0.33 | 47.41 | |
| RC T24 | 45.66 ± 0.33 | 49.26 | |
| RC T25 | 46.66 ± 0.33 | 48.15 | |
| RC T26 | 45.66 ± 0.88 | 49.26 |
RCT12,RCT17b) RCT11,RCT18c) RCT20d)RCT23,RCT25
Table 3: Inhibition of Rhizoctonia solani by different isolates of Trichoderma in dual culture (in mm).
Screening of antagonistic potential of Trichoderma with sclerotial fungi
Screening of antagonistic potential of different isolates of Trichoderma against R. solani was assessed by dual Culture in Petri dishes [23]. A mycelial bits of 5 mm diameter obtained from the margin of 3 day old actively growing colony of test fungus (R. solani) was place on a fresh PDA plate 2 cm from the centre of petriplate and antagonist (Trichoderma spp.) were placed opposite to test fungus at 4 cm apart in petriplate in triplicate. R. solani alone inoculated in PDA plate served as control. The radial growth of the pathogens in dual culture and control plates was measured periodically which incubated at 24°C ± 2°C in BOD [24]. The percent inhibition of mycelia growth over control was calculated by following equation [25].
I%=C-T/C × 100
Where, I=Percent inhibition of mycelial growth, C=Growth of mycelium in control.
T=Growth of mycelium in treatment in R. solani.
Statistical analysis
Data were analyzed using statistical package SPSS version 20.
Result and Discussion
Native strains of Trichoderma were isolated from 26 different rhizospheric soils from sites selecting from different blocks of Ranchi district of Jharkhand and coded (Table 1) and individual culture of every isolate was maintained for further experimentation. The isolates were studied for species identification and the same revealed that they belong to Trichoderma Isolates. The isolates of Trichoderma were cultured in Petri plates individually and also in dual culture with R. solani and growth data has been recorded (Table 2). Further, the percent inhibition by all the 26 Trichoderma isolates against R. solani has been evaluated (Table 3). Among isolates of Trichoderma, seven isolates showed strong antagonistic potential which inhibited >50% mycelial growth of R. solani, viz., RCT1 (53.71%) followed by RCT22 (52.6%), RCT3 (51.85%), RCT7 (51.11%), RCT10 (50.37%), RCT 8 (50%) and RCT14 (50%). Moreover, seventeen (17) isolates were also showed inhibitory but their antagonistic potential < 50% of the mycelial growth while two isolates (RCT12 and RCT17) showed < 40% mycelial growth. Trichoderma species was found to be an effective biological control agent for protecting a number of crop plants from damaged induced by S. rolfsii and R. solani under both greenhouse and field conditions in the study conducted by [26,27]. Various agro products is used for biomass production and applied as a biocontrol agent. Several mechanisms may explain the biocontrol activity of these strains [28,29]. Hyperparasitism and volatile metabolites may be involved in the inhibition of R. solani [30]. Cell wall degrading enzymes (CWDEs) such as chitinase, glucanase and proteases are thought to be closely related to the mycoparasitism of Trichoderma strains [17-32]. Inhibitory volatile substances such as alkylpyrons may also contribute to the biocontrol activity of some Trichoderma strains [33-35]. Harman et al. reported that the Trichoderma have ability to antagonized soilborne phytopathogens as well as it also induced plant growth promotion and protect plants form biotic and abiotic stresses [36,37]. Thus, it can be concluded that Trichoderma isolates proves to be effective biocontrol agent and native isolates of it may be further explored as biocontrol agent against R. solani.
Conclusion
Twenty six (26) native strains of Trichoderma were isolated from acid soil of Jharkhand and were characterized on the basis of their morphological features, antagonistic potential and compared with known strains of T. asperellum. Seven (7) isolates of Trichoderma showed strong antagonistic potential which inhibited >50% mycelial growth and seventeen (17) isolates were also showed inhibitory which antagonized < 50% of the mycelial growth of R. solani while two (2) isolates only showed < 40% antagonistic potential. As per result, seven potential isolates of Trichoderma may be further exploited as biocontrol agent against R. solani as well as other Soilborne phytopathogenic fungi.
Acknowledgements
The authors are thankful to Dr. Arun Kumar Singh, Principle Scientist & Head, and ICAR Research Complex for Eastern Region, Research Centre, and Ranchi for providing necessary laboratory facilities and guidance during the present research.