Research Article - (2011) Volume 1, Issue 1
1Dept of Microbiology, Maulana Azad College, Aurangabad, Maharashtra, India
Textile dye Reactive orange 16 was selected for biotransformation studies by Enterococcus faecalis YZ 66.Optimization of parameters for dye decolourization were studied under static anoxic condition. Under optimized condition decolourization of Reactive orange 16 by Enterococcus faecalis YZ 66 was found to be 77.73% in 80 minutes. Degradation of the dye was confirmed by UV- Visible spectophotometric, TLC, and HPLC analysis. The isolate had potential to decolourize mixture of five dyes. This indigenous isolate could be a potential organism for bioremediation of textile wastewater carrying dyes.
RO 16, Enterococcus faecalis, SM
A great variety of synthetic dyes are used for textile dyeing and other industrial applications. The structural diversity of dyes is attributed to the presence of different chromophoric groups such as azo, triphenyl methane and phthalocyanine. Such synthetic dyes are released into the environment from textile and other dyestuff industries. [1] Azo dyes represent about half of the dyes used in the textile industry and, as a consequence, a relevant problem of pollution related to the release of these products in the environment is taking place.[2] Reactive dyes are extensively used for dyeing process in textile. They exhibit a wide variability in chemical structure, primarily based substituted aromatic and hetero cyclic groups. Since reactive dyes are highly soluble in water, their removal from waste water is difficult by conventional coagulation and the activated sludge process. [3]
The textile industries daily discharge million liters of untreated effluents in the form of wastewater into public drains that eventually empty into rivers. [4] The textile industries produce effluents that contain several types of chemicals that are toxic and mutagenic and /or carcinogenic.[5,6]The traditional textile finishing industry consumes about 100 liters of water to process about 1 Kg of textile material. During industrial processing up to 40% of the used dyestuff are released into the process water producing highly coloured wastewater that affects aesthetics, water transparency and gas solubility in water bodies. [7-9]
Dyes released from the textile processing and dye stuff manufacturing industries results in increase in organic load of natural reservoirs. Generally, various dyes found in industrial effluents, ultimately, enter the aquatic ecosystem and can create various environmental hazards. These have very adverse and sometimes irreversible effects on other animals and plants as well. The main purpose of wastewater treatment is the removal of these toxic substances and colour and try to make the water usable for industrial or domestic use. [10] The dyes are toxic and carcinogenic in nature and environmental contamination by these toxic chemicals is emerging as a serious global problem. Coloured solution containing dyes from industrial effluents of textile, dyeing and printing industries may cause skin cancer due to photosensitization and photodynamic damage. [11]
Pollution caused by dye effluents is mainly due to durability of dye in wastewater, colour fastness, stability and resistance of dyes to degradation. Many of the dyes when released into the natural reservoir are toxic for the natural fauna and flora.
Several methods are used to treat coloured effluents to achieve decolourization. These include physicochemical methods such as filtration, coagulation, use of activated carbon and chemical flocculation. [12,13] These methods are effective but they are expensive and involve the formation of a concentrated sludge that creates a secondary disposal problem which requires safe disposal. [14-18]Many techniques like electrochemical coagulation, reverse osmosis, nano filtration, adsorption using activated materials etc., are used for the removal of dye from waste water. [19]
Many microorganisms belonging to different taxonomic group of bacteria, fungi, actinomycetes and algae have been reported for their ability to decolourize azo dyes.[20-22] Many bacteria are capable to degrade azo reactive dyes aerobically and anaerobically. [23,24]Biodegradation systems of colour removal through the use of bacteria have been shown to be highly effective. The lower cost of treatment and amenability to scale up are among the merits of biological methods. [25]
The objective of the present study was to examine the decolourization of the azo dye Reactive Orange 16 by Enterococcus faecalis YZ 66 under static conditions and in the determination of the effect of the dye and the degraded metabolites on the growth of crop plants.
Microorganism and culture conditions
Dye industry effluent contaminated soil, sewage, dung and dye waste were subjected for acclimatization to C.I. Reactive orange 16, an azo dye in the basal nutrient medium, nutrient broth. The most promising bacterial isolate was used for further dye degradation studies. The culture was identified as Enterococcus faecalis YZ 66. Pure culture was maintained on the nutrient agar slants. Composition of nutrient broth and agar used for decolourization is (g/L) Peptic digest of animal tissue 5, NaCl 5, Beef extract 1.5, Yeast extract 1.5 and pH 7.4±0.2
Dyestuff and chemicals
All chemicals used were of the highest purity and of analytical grade. Nutrient broth dehydrated was purchased from Hi-Media, Mumbai, India. The textile dye Reactive orange 16 (RO 16) was obtained from Spectrum dyes, Surat, India
Identification of the culture
A preliminary identification of the culture was based upon biochemical tests. 16sr-RNA sequencing of the isolated organism was done in GeneOmbio Technologies Pvt. Ltd., Pune, India. Phylogenetic tree was constructed by the Neighbour –Joining method.
Decolourization experiments
Enterococcus faecalis YZ 66 was grown for 24 hrs at 37°C on nutrient agar.10% inoculum of O.D600 1.0 [26] was used throughout the study. The isolate was inoculated in nutrient broth to study the decolourization performance of the culture. The dye was filter sterilized by using 0.2 μm filter and added after sterilization of medium throughout the study. The dye (50mg/L) was added immediately and incubated at static condition at 37°C. The aliquot (3ml) of culture media was withdrawn at different time intervals and centrifuged at 6000g for 20 min. Decolourization was monitored by measuring the absorbance of the culture at λmax of the dye at 495 nm and change in pH was also recorded.
Decolourization at different dye concentration
In order to examine the effect of initial dye concentration on the decolourization in static condition 50-500mg/L of RO 16 was added to the nutrient broth inoculated with 10% Enterococcus faecalis YZ 66 of O.D600 1.0 and incubated at 37°C under static condition. The % decolourization was measured after 13 hours. All decolourization experiments were performed in triplicates. Abiotic controls (without microorganism) were always included. The % decolourization and average decolourization rate was measured.
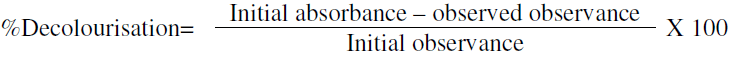
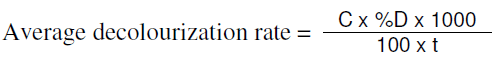
Where C = initial concentration of dye mg/L
% D = Dye decolourization % after time t. [27]
Effect of pH on dye decolourization
Nutrient broth of different pH 3, 4, 5, 6, 7, and 8 was inoculated with 10% inoculum and incubated at 37°C under static condition. The dye concentration was 50mg/L. All decolourization experiments were performed in triplicates. Abiotic controls (without microorganism) were always included. The % decolourization was measured.
Effect of temperature on dye decolourization
Nutrient broth of pH 7 was inoculated with 10 % inoculum and filter sterilized dye at 50 mg/L was added after sterilization. The broth was incubated at 280C, 370C, 400C, 450C and 500C. The experiment was carried out in triplicate. Abiotic controls (without microorganism) were always included. The % decolourization was measured
Effect of carbon and nitrogen sources
To study the effect of carbon and nitrogen sources on decolourization of RO 16 , semi synthetic medium having following composition was used (g/L)- ( NH4)2SO4 0.28, NH4Cl 0.23, KH2PO4 0.067, MgSO4.7 H2O 0.04, CaCl2.2 H2O 0.022, FeCl3.6 H2O 0.005, yeast extract 0.2, NaCl 0.15, NaHCO3 1.0 and 1 ml/ L of trace element solution containing (g/L) ZnSO4. 7 H2O 0.01, MnCl2.4 H2O 0.1, CuSO4 .5 H2O 0.392, COCl2. 6 H2O 0.248, NaB4O7.7 H2O 0.177 and NiCl2 .6 H2O 0.02 with different carbon and nitrogen sources ( 1% each) such as glucose, sucrose, lactose and starch, yeast extract, peptone, malt extract, meat extract and urea respectively. [27]50 mg/L of the dye concentration was used. Filter sterilized dye was added after sterilization of the medium and after inoculation of the isolate.
Analytical procedure
The metabolites produced during the biodegradation of RO 16 at 3.5 hrs i.e. after decolourization of the medium were extracted twice with equal volume of dichloromethane (DCM). The DCM extracts were pooled and evaporated at 40°C in a rotary evaporator and then transferred to a test tube. [28]The extracted residue was dissolved in small volume of HPLC grade methanol and the same sample was used for analysis. During UV visible spectral analysis, changes in absorption spectrum in the decolourized medium (400-800nm) were recorded in comparison with the results from the control runs. [27] HPLC analysis was performed in an isocratic system Shimadzu (SCL 10 AVP) equipped with dual absorbance detector using C 18 column with HPLC grade methanol as mobile phase at the flow rate of 1.0 ml / min for 10 min at 422 nm. The mobile phase used for TLC was composed of methanol, ethyl acetate, n-propanol, water and acetic acid (1:2:3:1:0.2 v/v) and silica gel plates ‘Merck’ was used for separation. TLC plate was developed using iodine chamber. [27,29]
Toxicity study
Phytotoxicity tests were carried out in order to assess the toxicity of RO 16 and metabolites formed after decolourization of RO 16. Phytotoxicity tests were carried out at a final concentration of 400 ppm on two kinds of seeds. One from grains Sorghum vulgare (monocot) and second from pulses Phaseolus mungo (dicot), commonly cultivated. Phytotoxicity was conducted at room temperature (10 seeds of each) by watering separately 5ml sample of control RO 16 and its degradation products per day. Control set was carried out using distilled water at the same time. Germination % was recorded after 7 days.[29]
Statistical analysis
Data was analyzed by one way analysis of variance (ANOVA) with Turkey- Kramer multiple comparison test. Readings were considered significant when P was ≤0.05.
Isolation and identification of dye decolourizing bacteria
Isolation of bacteria from soil sample was carried out by the enrichment technique using nutrient broth and RO 16 as source of carbon and nitrogen that has rapid decolourization capacity. Decolourization occurred only when a carbon and nitrogen sources were available for growth, isolated bacteria was Gram positive facultative anaerobic motile coccus. An identification of the culture was based on biochemical tests and 16sr-RNA analysis done by geneOmbio Technologies, Pune. The nucleotide alignment of this strain showed it was most phylogenetically similar to the Enterococcus faecalis YZ 66. ( Gene bank Accession number JN 129260)
The isolate showed ability to decolourize 97.24% of RO 16 within 3.5 hrs at a dye concentration 50 mg/L. the absorbance peaks in the visible region disappeared indicating complete decolourization.[12]. In the UV spectra the peak at 495nm was replaced by new peak at 302 nm.(Figure 2)
Decolourization with respect to time showed at 3.5 hrs 96.56 % decolourization. (Figure 3) There was no abiotic loss of RO 16 within 24 hrs incubation indicating that the decolourization of RO 16 was due to biological mechanism rather than adsorption. To confirm whether this decolourization was due to the bacterial action or variation in pH, change in pH was recorded in the range of 7.4±0.2.
Effect of physiochemical conditions on the decolourization performance
The effect of various physiochemical conditions such as pH, temperature, dye concentration, effect of carbon and nitrogen sources on decolourization of RO 16 by the isolate were studied in detail. All parameters were studied at 370C under static condition. 10% inoculum with O.D600 1.0 was used at a dye concentration 50 mg/L.
Effect of pH
Bacterial cultures generally exhibit maximum decolourization at pH values near 7.0, our culture exhibited decolourization activity in the range of pH 5-8. At pH 3 and 4 decolourization observed was 11.276 % and 2.9 % respectively. Maximum 96.56 % being at pH6.(Figure 4) Enterococcus faecalis grows very well in the pH range of 5-9. E.coli and Pseudomonas luteola both exhibited best decolourization at pH 7.0 with constant decolourization rate up to pH 9.5 [30] Klebsiella pneumoniae RS-13 completely degraded methyl red in the pH range from 6.0 to 8.0.[30]
Effect of Temperature
Various microorganisms showed their survival at various temperatures ranging from 28°C to 50°C.[26] The isolate showed complete decolourization at 27°C, 37°C and 40°C but rapid decolourization was observed at 400C. (Figure 5) This may be owing to a greater production of enzymes and optimal growth conditions of the isolate for its dye decolourizing ability. The decolourization at this optimum temperature may be owing to higher respiration and substrate metabolism. This also demonstrates that decolourization of the dye was through microbial reaction which relies on optimal temperature and not by adsorption.[26] At 45°C and 50°C there was no decolourization. Hence pH and temperature optimum for RO 16 was found to be pH 5 and 40°C respectively
Effect of initial dye concentration
Actual concentrations of reactive dyes in dye house effluent have been reported to range from 60-250mg/L[30] Figure 6 showed decolourizing ability of our culture increased with increase in dye concentration from 50-400mg/L. The activity was lower at dye concentration 500mg/L. It has been proposed that dye concentration can influence the efficiency of microbial decolourization through a combination of factors including the toxicity imposed by dye at higher concentration. [30,31].
Thus, the isolate which could decolourize dye much above the reported dye concentration in wastewater, can be successfully employed for treatment of dye bearing industrial wastewater.
Effect of carbon and nitrogen sources
While trying to enhance decolourization performance of RO 16, extra carbon and nitrogen sources was supplied in semi synthetic medium. There was no decolourization in the presence of synthetic media. Percentage decolourization was maximum with purified substrate meat extract 96.34 % while little less decolourization with other supplements of carbon and nitrogen source within 24 hours.(Figure 7) Similar results were also reported by bacterial consortium RVM for decolourization of reactive violet 5. [27,32]The culture showed 93.57 % decolourization in the presence of glucose and maximum decolourization was reported in presence of lactose (96.816%). In addition, supplying urea as a nitrogen source exhibited less decolourizing ability. Similar results were reported by Sartale et al. 2009. In contrast, addition of carbon sources seemed to be effective to promote the decolourization probably due to the preference of the cells in assimilating the added carbon sources over using the dye compound as the carbon source.
Decolourization of mixture of dyes
Synthetic dyes of different structure are often used in the different industries and therefore the effluents from industry are markedly variable in composition. [27] We evaluated the decolourization ability of mixture of various industrial dyes such as RR 195, Direct Red 81, Acid Blue 113, Reactive orange 16 and Reactive yellow 145 at a concentration of 50mg/L in batch culture at 370C under static condition. The rate of decolourization was very fast and colour removal was 80.58% within 2 hrs. Isolated Aeromonas hydrophila showed significant decolourization performance foe mixture of dyes within 2 days. [33] Thus, potential of the isolate in concern with diversity of dyes and their non specificity could be useful for the treatment of the dye bearing wastewaters containing mixture of dyes.
Analysis of metabolites resulting from decolourization and biodegradation of RO 16 by Enterococcus faecalis YZ 66
To understand the possible mechanism of the dye decolourization, we also analyzed the products of biotransformation of RR195 by UV visible spectral analysis, TLC, HPLC.
UV visible scan (400-800nm) of the culture supernatant withdrawn at different time intervals indicated the decolourization and decrease in dye concentration from batch culture. Peak obtained at 422 nm decreased at complete decolourization. The absorbance peak in the visible region disappeared indicating complete decolourization. In the UV spectra, the peak at 495 nm was replaced by new peak at 302 nm.
TLC analysis showed the appearance of one spot in the sample containing the extracted metabolites of completely decolourized medium with Rf value 0.85 wheres Rf value of RO 16 was noted as 0.92 confirming the biodegradation of RO 16 by Enterococcus faecalis YZ 66.
HPLC elution profile showed prominent peak at retention time at 3.13 min when products were separated from the sample obtained after decolourization, compared to control peaks with retention time at 1.65 and 1.94 min. The analysis showed the presence of new peak with disappearance of the peak at 1.65 and 1.94 min which was of RO 16 confirming the degradation of RO16 by Enterococcus faecalis YZ 66(Figure 8)
Phytotoxcity study
Despite the fact, untreated dyeing effluent may cause the serious environmental and health hazards. They are being disposed off in the water bodies and this water can be used for irrigation purpose. Thus it was found necessary to access the phytotoxicity of the dye before and after degradation. The relative sensitivies ion terms of % germination towards the dye RO 16 and its degradation products in relation to wheat (Sorghum vulgare) and mung (Phaseolus mungo) seeds were presented in the Figure 9.
There is no significant difference in the % germination in case of wheat irrigated with the dye but in case of metabolite irrigated wheat it was significantly increased (p ≤ 0.05) as compared to control wheat.
Toxic RO 16 completely biodegraded by Enterococcus faecalis YZ 66 was able to degrade toxic reactive orange 16.It effectively decolourized under static condition various azo dyes, which are commonly used in the industries. Enhanced decolourization was observed in presence of meat extract as additional nitrogen and all tested additional carbon sources. UV visible analysis, TLC, HPLC analysis of extracted products confirmed the biodegradation of RO 16. The strain also showed colour removal of mixture of dyes indicating the biodegradation of complex dyes and applicability of the strain to decolorize and degrade wide variety of industrial dye and dye mixture. Hence this indigenous bacterial strain could be utilized for treatment of dye containing wastewater.