Research Article - (2016) Volume 1, Issue 1
Harold I. Zeliger*
Zeliger Research and Consulting, 41 Wildwood Drive, Cape Elizabeth, Maine 04107
*Corresponding Author:
Harold I. Zeliger Zeliger
Research and Consulting, 41 Wildwood Drive, Cape Elizabeth, Maine 04107, USA
Tel: 207-747-4040
E-mail: hiz@zeliger.com
Received Date: October 27, 2015; Accepted Date: December 24, 2015; Published Date: December 29, 2015
Citation: Zeliger HI.Causes, Mechanisms and Prevention of Environmental Diseases.Dual Diagn Open Acc. 2015, 1:1. doi: 10.21767/2472-5048.100001
Copyright: © 2016 Zeliger HI.Causes. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
The onset of non-communicable disease has been attributed in large part to environmental exposure to toxic chemicals, including persistent organic pollutants, volatile and semi-volatile organic compounds and transition metals. This review summarizes recent research into the causes and mechanisms of environmental disease onset and examines the role of oxidative stress as well as steps that can be taken to predict and lower the incidences of environmental diseases.
Introduction
Environmental diseases (ENVDs) are non-communicable diseases that result when people are chronically exposed to toxic environmental chemicals. Other contributory causes of ENVDs include radiation, pathogens, allergens and psychological stress. These other causative agents, however, are minor compared with the chemical ones, which are the primary focus of this review. ENVDs are generally late-onset, appearing only after numerous toxic exposures. In recent years, however, the age of onset has been trending lower [1]. For all the ENVDs discussed, it is appreciated that genetic as well as environmental factors contribute to the onset of these diseases. It has been shown, however, that incidences of ENVDs are greatly enhanced by exposures to toxic environmental chemicals, and that more than one exogenous agent may trigger any given disease. The World Health Organization estimates that as much as 24% of global disease is caused by environmental exposures [2]. It has also been shown that 40% of cancers world wide can be prevented by lifestyle choices [3].
Hundreds of diseases fall under the definition just offered [4]. It is beyond the scope of this writing to address them all. The ENVDs discussed here are those that have been extensively studied and written on, thus providing a basis for reaching reasonable scientifically certain conclusions. Table 1 contains a list of the ENVDs addressed here and representative references for these [5-15]. The reader is referred to these citations for additional reference material.
| Disease | |
|---|---|
| Metabolic: | Type 2 diabetes (T2D) Metabolic syndrome (MET-S) |
| Cardiovascular: | myocardial infarction atherosclerosis hypertension coronary heart disease peripheral heart disease ischemic heart disease cardiac autonomic function |
| Neurodevelopmental: | autism spectrum disorder (ASD) attention deficit hyperactivity disorder (ADHD) |
| Neurodegenerative: | Alzheimer's disease (AD) Parkinson's disease (PD) Amyotrophic lateral sclerosis (ALS) |
| Neurologicalimpairments: | cognitive effects motor deficits sensory deficits peripheral nervous system effects |
| Immunological: | Allergic responses including chemical sensitivity systemic sensitization autoimmune diseases |
| Musculoskeletal: | Rheumatoid arthritis osteoporosis |
| Respiratory: | Asthma chronic obstructive pulmonary disease (COPD) |
| Cancer: | childhood leukemia other childhood cancers breast cancer prostate cancer kidney cancer numerous other cancers |
Table 1: Environmental diseases associated with chronic exposures to lipophilic exogenous toxicants and references for these [5-15]. References are to review publications. The reader is referred to the numerous references contained in these sources for primary material.
All of the diseases in Table 1 have reached epidemic and pandemic proportions in the past two generations. The dramatic increase of environmental disease prevalence with time can be seen from plots of disease percent increases versus time, from the 1950's to the present time. Such plots for numerous diseases produce hyperbolic curves, with examples being autism and autism spectrum disorders [16], type 2 diabetes [17], and obesity [18] in the United States. The slopes of these curves exactly correspond to those of plots for chemical production and use versus time, as illustrated by data for synthetic chemical production [17], and increased pesticide use versus time, much of which is dictated by the increased use of genetically engineered crops and global warming [19,20]. Figure 1, which shows the increase in incidence of autism in the United States from 1975 to the present, is representative of these relationships. Other disease increase rates that follow this curve include; childhood cancers, onset of dementia, other neurological diseases, breast, prostate and numerous other cancers as well both male and female infertility [4]. World wide energy production from combustion of fossil fuel use and its resultant air and water pollution increases also follow the slope of the curve in Figure 1 [21].
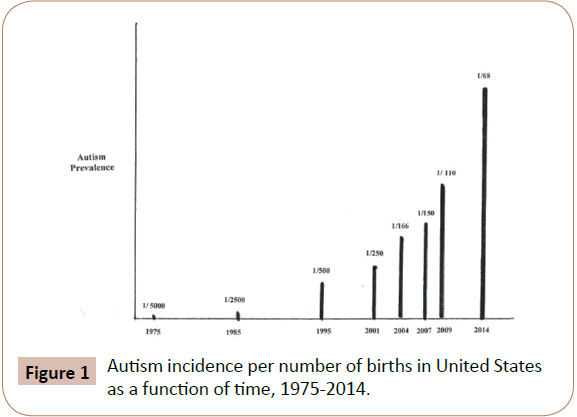
Figure 1: Autism incidence per number of births in United States as a function of time, 1975-2014.
The rapid increase in the incidence of ENVD occurred as a result of industrialization, changes in farming protocols and the increases in exposures to chemicals released into the environment as a result of these activities. Life style changes, including increased use of tobacco and the widespread introduction of processed foods also greatly contributed to the ENVD epidemics [4]. Before 1950, largely agrarian China was largely shielded from dramatic increases in non-communicable diseases. In recent decades, however, with rapid industrialization and resultant environmental impact, the Chinese people have experienced the same ENVD acceleration as those in the Western industrialized nations have, but over a much shorter period of time [22]. Environmental pollution, tobacco use and the obesity rate, for example, have dramatically increased [23]. Thus the Chinese "experiment" has served to highlight the cause and effect relationship between chemical exposure and environmental disease onset.
Chemical Toxicity
Historical
Acute exposures to high concentrations of toxic chemicals have been known for a long time to induce predictable deleterious health effects, but low concentrations of such chemicals have historically been believed to be benign to humans, In fact, regulatory agencies have assigned acceptable levels of exposure, known as permissible exposure levels (PELs) time weighted averages for 8 hours of exposure (TWAs) and no observed effect concentration values (NOECs) at which exposures presumably have no toxic effects [24].
Toxicity of Chemical Mixtures
Toxic effects of mixtures of chemicals with similar modes of action have been believed to be additive, while the effects of exposures to mixtures of chemicals with dissimilar toxic effects have been assumed to be benign as long as the concentrations of each was below established toxic levels. Synergistic and potentiated effects of dissimilar toxic chemicals have also been known for a long time. A review of the toxicological literature pre-2003 revealed that exposures to chemical mixtures had at times resulted in low concentration level toxicity, unpredicted target organ attack or greater than anticipated toxicity than from the known toxicology of individual chemical species in the mixtures. The studies that reported these unusual effects from exposures to chemical mixtures conceded that unknown factors were responsible for the observed effects [23].
Toxicity of Mixtures of Lipophilic and Hydrophilic Chemicals
Thus, health effects resulting from environmental exposures to toxic chemicals below regulatory agency established limits remained unexplained until 2003, when for the first time; it was shown that exposures to mixtures of chemicals containing at least one lipophilic and one hydrophilic species produced acute and chronic toxic effects even at very low level concentrations. In all of the chemical mixture exposure cases cited in the pre- 2003 literature and those reported for the first time in 2003 that described unusual and unpredicted toxic effects, the chemicals comprising the mixtures contained at least one lipophilic and hydrophilic component. It was hypothesized that the lipophilic component, which can penetrate mucous membranes, promoted the permeability of the hydrophilic species which would otherwise not permeate the lipophilic membranes. This hypothesis was supported by evidence from the literature in which it is shown that lipophilic chemicals are routinely added to hydrophilic pharmaceuticals that alone would not penetrate tissue at rates sufficient for clinical usefulness [25].
It was reported in 2003 that exposures to mixtures of lipophiles and hydrophiles produced enhanced toxicities at higher concentrations and, surprisingly, that such mixtures targeted organs and systems not known to be affected by the individual species and that different mixtures attacked different body organs and systems with each mixture acting as a unique toxic agent [25]. Though the original study focused on respiratory, central nervous system, liver, gastrointestinal and cardiovascular system effects, subsequent studies revealed that virtually every organ and system in the body is affected by low level exposures to mixtures of lipophilic and hydrophilic chemicals. Numerous examples, with no dissenting citations, were found in the literature to support these findings [24].
Disease Clusters
Cancer Clusters
The study of disease clusters affecting non-related individuals offers the opportunity to relate toxic effects of environmental chemicals with the genetic components factored out. In 2004, it was reported that that previously unexplained cancer clusters could be attributed to chronic exposures to mixtures of lipophiles and hydrophiles. A study based on an analysis of 12 cancer clusters that had previously been reported in the literature, and were not predicted from a consideration of the etiologies of the individual chemicals of exposure, was undertaken. It was shown that in each instance the cluster that ensued was attributed to exposures to mixtures of lipophilic and hydrophilic environmental chemicals. The cancer clusters included childhood leukemia, prostate cancer, testicular cancer, brain cancer and intracranial neoplasms in the children of mothers and fathers who were exposed and colorectal cancer [26].
Sequential Absorption
Sequential Absorption of Lipophiles
It has been well established that high molecular weight lipophilic exogenous chemicals such as polychlorinated biphenyls (PCBs) and organochlorine pesticides (OCs) are retained in the body for as long as decades [27-30]. In 2012 it was reported that even very low molecular weight lipophilic hydrocarbons, such as propane and butane, are retained for relatively long periods of time (4-7 days) and that constant exposure to these, via inhalation of polluted air, for example, can establish steady states of these compounds in the body [9]. It was further reported in that study and elsewhere [24] that due to the retention of lipophilic species in the body, absorption of both lipophile and hydrophile need not occur simultaneously. Rather, the absorption may be sequential, with the more slowly eliminated lipophiles absorbed initially followed by the subsequent absorption of hydrophiles to form the toxic mixtures. It is to be noted that hydrophilic chemicals are either not absorbed or are rapidly purged from the body in the absence of lipophilic species in which they are soluble [24].
Delayed Onset of Disease
The studies just discussed demonstrated that sequential absorption can account for the delayed onset of toxic consequences and led to the study of such induction of environmental disease. The first evidence for this was came in 2012, when it was reported that statin use by postmenopausal women resulted in an elevated risk for type 2 diabetes (T2D) [31]. An analysis of the data in that study revealed that all of the Statins which were reported on were lipophilic species and that the only Statin in clinical use at that time which did not raise the incidence of T2D was a hydrophilic species. It was hypothesized that though the lipophilic Statins are not as long lived in the body as PCBs and OCs, they are taken on a regular basis and establish a steady state in the body thus enabling them to act in a similar manner to other, longer retained, lipophilic chemicals [32].
Diabetes
In a study reported on in 2013, the association of lipophilic chemical exposure as a cause of type 2 diabetes (T2D) was examined [33]. The results of that study revealed that exposures to numerous lipophilic chemicals can lead to the increased incidence of T2D and that the slow onset of disease is due, at lease in part, to the initial absorption and the establishment of a steady state of lipophiles in body serum, followed by the sequential absorption of the hydrophiles as these become available. The sequential absorption of hydrophilic chemicals can occur minutes to years after the initial absorption of lipophilic species [33]. In T2D, dose-response relationships between serum levels of lipophiles and prevalence of disease have been reported [34,35]. Dose-response relationships have also been reported for neurological disease, cardiovascular disease, respiratory disease and cancer [36-40].
Cardiovascular and neurological diseases
In studies subsequent the diabetes study, it was reported that exposures to the identical lipophilic chemicals that were associated with increased incidence of T2D also triggered the onset of cardiovascular disease (CVD) [41], neurological disease; neurodevelopmental disease, including autism and autism spectrum disorders (ASD), as well as attention deficit hyperactivity disorder (ADHD); and neurodegenerative disease, including Alzheimer's disease (AD), Parkinson's disease (PD) and amyotrophic lateral sclerosis (ALS) [42]. Many other environmental diseases, that affect virtually all body systems, are also associated with exposure to and retention of exogenous lipophilic chemical species. These include: immunological [43], musculoskeletal [44,45] and respiratory diseases [46-48]; obesity [13] and numerous cancers [49-51]. Table 2 lists lipophilic exogenous chemicals, exposure to which has been shown to increase the incidences of these environmental diseases [13,33-51].
| Chemical |
|---|
| Persistent organic pollutants: polychlorinated biphenyls (PCBs) organochlorine pesticides (OCs) DDT DDE dieldrin dioxins furans hexachlorobenzenes (HCBs) polybrominated biphenyls (PBBs) polybrominateddiphenyl ethers (PBDEs) |
| Polynuclear aromatic hydrocarbons (PAHs) acenaphthene acenaphthylene anthracine benz[a]anthracine benzo[a]pyrene benzo[e]pyrene benzo[b]fluoranthrene benzo[g,h,i]perylene benzo[j]fluroanthene chrysene dibenzo[a,h]anthracene fluoanthene fluorine indeno[1,2,3-c,d]pyrene naphthalene |
| C-10 to C-20 alkanes |
| Perfluorinated compounds |
| Plastic formulation additives Phthalates diethylhexyl phthalate dibutyl phthalate di-n-pentyl phthalate dicyclohexylphthalate diallyl phthalate diissodecyl phthalate di-n-hexyl phthalate diisobutyl phthalate di-n-octyl phthalate diisononyl phthalate dihheptyl phthalate |
| bisphenol-A bisphenol-S |
| Phthalates |
| Low molecular weight hydrocarbons (LMWHCs) C-1 to C-8 alkanes benzene ethyl benzene styrene toluene xylenes |
| Halogenated alkanes and alkenes vinyl chloride trichloroethylene tetrachloroethylene methylene chloride |
Table 2: Lipophilic exogenous chemicals exposure to which has been shown to increase the incidence of numerous environmental diseases [13,33-51].
It has been reported that individuals morbid with one of the ENVDs in Table 1 also have high incidences of co-morbidities of other ENVDs [52]. In the United States alone, half of all adults have at least one environmental disease and more than a quarter of the adult population suffers from two or more co-morbid environmental diseases [53-55]. Accordingly, it is likely that an individual ill with one ENVD is likely to develop additional ENVD illness.
Co-morbidity of environmental diseases
All of the diseases listed in Table 1 are co-morbid with other environmental diseases that are known to be triggered by exposures to lipophilic chemicals [52]. Table 3 lists co-morbid disease pairs and references for each pair. Figure 2 [52] reprinted with permission) shows the co-morbidities of the 11 types of these diseases with each other. It is of note that of the 55 binary combinations possible, 45 (82%) % have been shown to be comorbid to date.
| Disease Pair |
|---|
| T2D - CVD |
| T2D - NRD |
| T2D - NDV |
| T2D - NDG |
| T2D - MSK |
| T2D - IMM |
| T2D - RES |
| T2D - CMS |
| T2D - OBS |
| T2D - CAN |
| CVD - NRD |
| CVD - NDV |
| CVD - NDG |
| CVD - MSK |
| CVD - IMM |
| CVD - RES |
| CVD - CMS |
| CVD - OBS |
| CVD - CAN |
| NRD - NDV |
| NRD - NDG |
| NRD - MSK |
| NRD - IMM |
| NRD - RES |
| NRD - CMS |
| NRD - OBS |
| NDV - RES |
| NDV - OBS |
| NDV - CAN |
| NDG - MSK |
| NDG - CAN |
| MSK - IMM |
| MSK - OBS |
| MSK - CAN |
| IMM - RES |
| IMM - CMS |
| RES - CMS |
| RES - OBS |
| RES - CAN |
| CMS - OBS |
| CMA - CAN |
| OBS - CAN |
Table 3: References for environmental disease co-morbidity pairs, for individual pair references see [52].
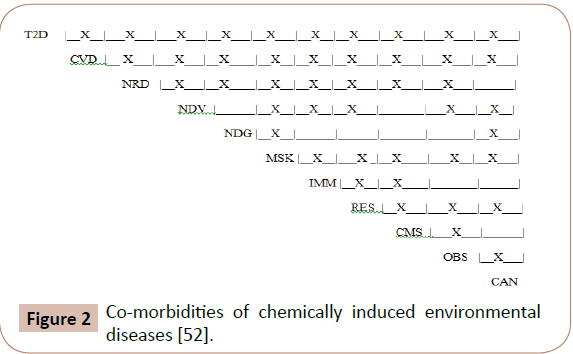
Figure 2: Co-morbidities of chemically induced environmental diseases [52].
Though different diseases involve attacks on widely disparate organs and systems, co-morbidity rates are high when individuals are exposed to environmental lipophilic toxins [9,52]. The onsets of co-morbid diseases do not follow set patterns. Published studies show that individuals with two co-morbid diseases, e.g., T2D and hypertension, are just as likely to become ill with one first as the other first [56]. The wide prevalence of co-morbid environmental diseases and the lack of a pattern of onset strongly suggest the common cause for these diseases that has been previously reported on [33,41,42] and that each lipophile: hydrophile pair acts as an independent toxicant [57].
Obesity
Obesity as an environmental disease
Obesity is an environmental disease that is a predictor of human adipose tissue concentration of POPs [58]. This is consistent with the fact that obesity is usually associated with CVS, T2D and other diseases, as adipose tissue releases the lipophiles it holds to the blood stream. Obesity is itself caused by POPs, phthalates, bisphenol A, polynuclear aromatic hydrocarbons (PAHs) and other exogenous lipophiles [13,59-63]. Being obese and having high serum lipophiles contributes to the absorption of these exogenous chemicals [58,64].
Obesity-lipophile-disease triangle
An Obesity - Lipophile - Disease triangle has been described as follows [52]. Exogenous lipophiles cause environmental diseases and obesity [52,65-70]. Environmental diseases (T2D, for example) cause the absorption of exogenous lipophiles and cause obesity [66,68,71]. Obesity causes environmental diseases and the absorption of lipophilic chemicals [72,73]. The Obesity- Lipophile- Disease triangle [52] is shown in figure 3.

Figure 3: Obesity-lipophile-disease triangle [52].
Absorption of Metals and other hydrophilic chemicals
Metals
The causes for the onset of the diseases listed in Table 1 are not limited to the action of lipophilic chemicals alone. Exposures to heavy metals and metalloids, which are listed in Table 4, have also been shown to increase the incidence of type 2 diabetes, cardiovascular diseases, neurological diseases and other ENVDs [74-86].
Hydrophilic transport
As described above, prior to 2015, is known that the toxic action of hydrophiles was based upon their solution in lipophiles and transport by lipophiles across the body's lipophilic membranes. Lipophilic species were defined as those with octanol: water partition coefficients (Kow) of 2.00 or greater and hydrophilic species were defined as those with Kow values of less than 2.00 [24,25]. It was empirically determined that optimal transport of hydrophiles by lipophiles across lipophilic membranes, and hence toxic effects of mixtures, were greatest when the Kow differences between hydrophiles and lipophiles was between 2.00 and 3.00 [25,57], and it was hypothesized that when Know differences were 5.00 or more, the solubility of hydrophile in lipophile was so low that no combined effect was observable [25]. Thus it was believed that metal ions, which are essentially insoluble in hydrocarbons, would act independently from hydrocarbons if exposure to both occurred simultaneously. As will be seen below, it has now been shown that metals are indeed transported across the body's lipophilic membranes by hydrocarbons when the hydrocarbons are planar aromatic species [87].
Metal transport
Some transition metals, iron and zinc, e.g., are required for body homeostasis. Yet, the onset of T2D, cardiovascular diseases, neurological diseases, neurodevelopmental diseases, neurodegenerative diseases, cancers and other ENVDs has been shown to be related to the action of elevated levels of Iron III ions (Fe+3) [73,88-94]. Until recently, it was mechanistically troubling as to how elevated levels of these metals permeate lipophilic cell membranes to enter body tissues. Recently, a physiochemical mechanism for ENVD onset based on the transport of metal ions through cell membranes via pi-bonding of metal ions to planar aromatic hydrocarbons was presented [87].
Cell Membranes
Membrane structure
Cell membranes are amphiphilic structures composed primarily of phospholipid bilayers with embedded proteins. The bilayers contain hydrophilic heads and hydrophobic (lipophilic) tails which contain embedded proteins. This arrangement allows the membrane to be selectively permeable to metal ions and other polar molecules, at the head, and through ion channels as well as the passive diffusion of lipophilic species through the tail. The lipid bilayer contains acyl groups that vary with cell type and include saturated and unsaturated fatty acid chains, with the two layers bonded by covalent disulfide bridges and van der Waals forces [95]. Two types of proteins are embedded in the lipid bilayer. The first, integral membrane proteins, interact primarily in the hydrophobic tails of cell membranes via nonpolar bonding. The second, peripheral proteins, interact primarily in the hydrophilic heads of cell membranes, via electrostatic and hydrogen bonds [96-99]. This variability allows particular cell membranes to selectively permit entry and egress of molecular species essential to their functioning. Thousands of different, unique membrane phospholipid structures have been identified. Each is characterized by the length and degree of saturation of its acyl chain, and all vary in membrane thickness, membrane fluidity, curvature and surface charge [98-100]. The differences in chemical composition and physical structure account for varying permeability’s of membranes.
Membrane permeability by lipophiles
Cell membranes are permeable to lipophilic exogenous chemicals. Such permeable lipophiles include but are not limited to: volatile and semi-volatile aliphatic hydrocarbons, single ring and polynuclear aromatic hydrocarbons (PAHs); phthalates; bisphenol-A; persistent organic pollutants such as polychlorinated biphenyls (PCBs), organochlorine pesticides (OCs) and polybrominated diphenyl ethers (PBDEs) fire retardants used in clothing; as well as food preservatives and disinfectants such as butylated hydroxytoluene (BHT), butylated hydroxyanisole (BHA), triclosan and parabens [24,25].
Effects of membrane permeation by lipophiles
Exogenous lipophilic chemicals (low molecular weight hydrocarbons, PAHs, pesticides and PCBs permeate through all cell membrane structures found in the human body, including the blood brain barrier (BBB) [101-106]. These chemicals penetrate into the hydrophobic tails of phospholipid cell membranes and accumulate there, leading to changes in membrane structure [107-109]. Such changes include: swelling of the membrane lipid bilayer, and thereby an increase in membrane surface area; and increase in membrane fluidity; inhibition of primary ion pumps; and an increase in proton and metal ion permeability [102-105]. These changes also bring about inhibition of embedded enzyme activity and the accumulation of exogenous lipophiles in the interior of the affected membranes [100,110].
Metal Toxicity
Metal absorption
As discussed above, Fe+3 is vital for cell homeostasis and is absorbed naturally via the polar heads and ion channels in healthy cells. Other vital transition metal ions are similarly absorbed. There are, however, two other routes by which Fe+3 and other transition metal ions may be absorbed through cell membranes. Firstly, cells with badly compromised lipid bilayers may cease to be barriers to hydrophilic species and permit direct passage of metal ions into the interior of cells [93,111]. Lipid bilayers can be compromised by the action of exogenous lipophiles or by surfactants that have polar ends which can bond to metal ions and non-polar ends which act as lipophiles and permeate cell membranes resulting in the transport of metal ions into cell interiors [115-117]. Secondly, cells that do not have previously damaged lipid bilayers may also be permeated by metal ions via pibonding of metals with planar aromatic molecular structures and co-passage of such metal/hydrocarbon complexes through the lipid bilayer. Pi-bonding of metal ions to aromatic ring structures is well established and the passage of metal-PAH complexes through lipophilic cell membranes has been previously reported [93]. Support for this hypothesis comes from the known toxic effects of mixtures of metals and aromatic hydrocarbons [118] as well as from consideration of the absorption of metal ions on the planar geometric shapes of PAHs, PCBs, phthalates, bisphenol-A, and mononuclear aromatics just described [93,114]. Metal ion/arene bonds arise from metal ion/pi-electron complexes which, due to the lipophilicity of the hydrocarbon component of the complex can permeate through the membrane lipid bilayer in a manner analogous to the permeation of lipophile/soluble hydrophile mixtures, as described above [24,25]. Following the penetration of the complex through the lipid bilayer, the complex dissociates, with the metal ions dissolving into the hydrophilic parts of the cell [93]. Both absorption routes lead to concentrations of metal ions that are toxic to cells.
Toxic consequences of metal absorption
Trivalent iron (Fe+3) can activate non-enzymatic blood coagulation resulting in the formation of parafibrin via a hydroxyl radical-catalyzed reaction that generates reactive oxygen species (ROS), which, in turn initiates oxidative stress [79,88,89]. Ferric ions cause the concomitant generation of hydroxyl radicals and polymerization of purified fibrinogen (FBG) [88]. In addition to iron, other transition metals have also been reported to generate hydroxyl radicals and thereby induce disease in humans. These metals, which include; silver, copper, vanadium, zinc, cobalt, mercury, lead, cadmium, arsenic, manganese and chromium are listed in Table 4 [74-78,80,89]. As is discussed below, production of ROS/RNS is the trigger of environmental disease.
Sequential absorption of metals
Essential metals are continually absorbed and desorbed into and out of healthy cells. Yet, as is the case with iron, excessive concentrations of these can lead to toxic consequences [88,89,90]. Non-essential metals such as cadmium, mercury, lead and chromium are toxic at very low concentration levels and have long half-lives in the human body with that of cadmium being on the order of 20-30 years. These transition metals are nonbiodegradable and can accumulate in the body with time [119]. The association between the sequential exposure to exogenous lipophilic chemicals and the onset of diabetes, cardiovascular disease, neurological disease, cancer and other diseases was discussed above [33,41,42]. Exposures to metals (including arsenic, cadmium, mercury lead, nickel, manganese, copper, zinc and chromium) are also known to induce all these diseases via the generation of ROS initiated oxidative stress [92-94,120-123]. Exposures to metals need not occur in one event and sequential exposures can, over time, generate toxic levels in the body [87]. It has also been reported that toxicity need not be due to a single metal ion species, but that total metal ion load may responsible for toxic effects [87]. This effect is complementary to the sequential absorption of lipophilic species and the reliance on total lipophilic load as indicative of prediction of permeability through lipophilic barriers and the onset of type 2 diabetes, cardiovascular disease, neurological diseases and co-morbidities [33,41,42,55]. Also, sequential absorption and accumulation in the body of both lipophiles and metal ions means that the body can be exposed to very low levels of both toxins and these toxins can accumulate in the body over time until critical levels are reached.
Total lipophilic and metal loads
It was also reported that both the lipophiles and the transition metal ions must be present in sufficiently high concentrations to induce disease onset [87]. At times, high concentrations of metal ions alone may be present and at times high concentrations of lipophiles alone may be present. Since all environmental exposures inevitably include absorption of metals and lipophilic chemicals, it is difficult to ascribe the onset of disease to metal or lipophile alone. Each alone, however, may not induce disease since the body continually metabolizes and/or eliminates both lipophiles and/or metals. Only when both species are present at critical levels, often involving sequential absorption, is disease triggered. It was additionally reported that total lipophilic load and total transition metal ion load, irrespective of the chemical nature of the individual species, are critical; since all lipophiles can permeate cell membrane lipophilic bilayers and numerous transition metal ions can pi-bond to a wide spectrum of planar aromatic hydrocarbons. Support for this dual chemical hypothesis comes from a consideration of the published health effects of both lipophilic chemicals and heavy metals. Both species have been shown to cause diabetes, cardiovascular disease, neurological disease (including neurological impairments, neurodevelopmental disorders and neurodegenerative diseases), immunological disease, obesity, cancer and act as endocrine disruptors. It is to be noted that although numerous studies have been published on the effects of a wide spectrum of individual lipophilic chemicals and heavy metals, only recently have the effects of mixtures of lipophiles and heavy metals been reported on [87]. Yet, it is essentially impossible for a human being living anywhere on the globe today not to be impacted by both lipophiles and transition metals at all times [124,125].
Body Protective Defenses
Metabolism and elimination
As discussed above, the human body naturally metabolizes and excretes exogenous toxic chemicals. Volatile and semi-volatile organic compounds are readily metabolized and eliminated. Though persistent organic pollutants, such as PCBs, PBBs, PBDEs, dioxins and furans are retained in adipose tissue for decades [27- 30], they slowly but continually partition into the blood stream from whence they can be absorbed by polyunsaturated oils, such as olive oil, and excreted [126-129]. Metals are also eliminated naturally via the action of natural chelating agents contained in spices, tea and other foods, for example [130-131]. In addition, the body has the ability to adapt to and repair damage caused by toxic chemicals [10]. These factors delay the onset of environmental disease. The liver and kidneys play vital roles in metabolism and elimination of toxic chemicals. A thorough discussion of these is, however, beyond the scope of this paper. The reader is referred to the literature for a fuller treatment of this subject.
Oxidative Stress
Oxidative stress can be induced in numerous ways. These include; exposure to exogenous toxic chemicals, including low molecular weight hydrocarbons, polynuclear aromatic hydrocarbons, chloro- and bromo- water disinfection by-products, phthalates, bis-phenol A, heavy metal ions; pharmaceuticals; exposure to radiation; as a result of environmental disease or infectious disease illness; psychological stress; sensory offensive agents; ingestion of certain foods and food additives; and obesity [6-8,25,55,87].
Chemical causes of Oxidative Stress
It is widely accepted that in vivo damage of biological molecules is initiated by reactive oxygen species (ROS) as well as by reactive nitrogen species (RNS) via oxidative stress (OS). It is believed and that elevated OS is responsible for a wide spectrum of diseases via molecular level toxic effects include lipid peroxidation, DNA attack, adduction, enzyme inhibition, oxidative attack on the central nervous system and cell signalizing, all of which have been linked to ENVDs including neurodegenerative diseases (Alzheimer's disease, Parkinson's disease, Amyotrophic Lateral Sclerosis), cancer, cardiovascular disease, diabetes, and others [132-135]. Electron transfer (ET) and the resultant formation of ROS can proceed via the reactivity of the metabolites of lipophilic molecules or via the action of metal ions to produce hydroxyl radicals [134,136].
Benzene serves as an example of a lipophilic molecule that undergoes a series of chemical reactions in the body to ultimately produce ROS. Benzene is metabolized to phenol, which is converted to catechol which is in turn oxidized to quinone [134]. Benzo[a]pyrene (BaP), a polynuclear aromatic hydrocarbon found in petroleum products and tobacco smoke and a known carcionogen, is another such example. It is the diol-epoxide of BaP, rather than BaP itself, that is the actual tumerogen [25].
Iron serves as an example of metal ion reactivity producing hydroxyl radicals. Molecular oxygen in the body can undergo a single electron reduction to form the superoxide radical anion, which in turn reacts with 2 protons to produce hydrogen peroxide (H2O2). H2O2 then reacts with divalent iron (Fe+2) to produce trivalent iron (Fe+3) and hydroxyl radical (OH*) via the Fenton reaction. Fe+3 may be converted back to Fe+2 via reaction with superoxide radical anion via the Haber-Weiss reaction. The net result is reaction of superoxide with hydrogen peroxide to produce hydroxyl radical [134].
All of the diseases listed in Table 1 as well as genitourinary, skin, hematological diseases and endocrine disorders including male and female infertility [137-149] have been associated with the generation of ROS. In addition, OS has also been shown to induce epigenetic effects [150-154].
The chemicals known to induce ROS are those listed in Tables 2 and 4. These include lipophilic species [155-160] and transition metal ions [161-164]. In addition, it has been reported that other chemical species, fructose for example, also induce oxidative stress [165].
| Arsenic |
| Cadmium |
| Chromium |
| Cobalt |
| Copper |
| Iron |
| Lead |
| Manganese |
| Mercury |
| Nickel |
| Silver |
| Tungsten |
| Vanadium |
| Zinc |
Table 4 Transition metal ions known to induce environmental disease [74-81].
Radiation
The parts of the electromagnetic spectrum (EMR) that induce oxidative stress include; ionizing, ultraviolet (UV), microwave and radiofrequency frequencies. It is well known that ionizing radiation causes oxidative stress [150]. UV radiation [166-168], microwave radiation [169-171] and radiofrequency radiation [172- 175] have also been associated with OS.
Biological stimuli
Biological stimuli that induce OS include; endogenous enzymes [176,177] and phagocytes [178,179], plant and animal allergens [180-183], and pathogens including bacteria, viruses and fungi [184-189].
Psychological stress
Chronic psychological stress (CPS) results in a prolonged state of oxidative stress, impairs the body's immune system response to anti-inflammatory signals [190] and results in the elevated secretion of glucocorticoids, a biomarker for which is cortisol [191,192]. CPS has been associated with increased risk of many diseases. These include; cardiovascular disease, diabetes, respiratory infections, autoimmune diseases, neurological diseases, including depression and psychiatric disorders, musculoskeletal disease, gastrointestinal and endocrine disorders, obesity and accelerated aging [192-199].
Aging
A discussion of OS as it relates to ENVDs requires attention to the part played by aging in the production of ROS. A widely held theory holds that, as part of aging, OS within mitochondria damages the mitochondria, which in turn leads to the production of increased quantities of ROS which cause further damage. Once it starts, this cycle leads to further damage and corresponding aging [200]. Oxidative stress has also been associated with the acceleration of telomere shortening and accelerated aging [201]. It is beyond the scope of this paper to fully explore the subject of aging. Numerous papers have been written on this subject, with the following representative of these [202-203].
Combined effects
The combination of CPS and stress induced by exogenous chemical exposure has been shown to produce additive OS effects [204-206]. These points out the need to consider all sources of stress - chemical, radiation, biological, aging as well as the social and psychological environment when evaluating total oxidative stress.
Environmental Disease Prevention
Absorption and ROS/RNS formation
As discussed above, two distinct events must occur for exogenous environmental chemicals to trigger disease: absorption and ROS/ RNS production. Prevention of ENVD prevalence can be reduced by limiting these events. That said, preventing chemically induced environmental disease, completely, is impossible, given the omnipresence of toxic chemicals everywhere on the globe. There are, however, steps that can be taken to significantly reduce the incidences of ENVDs. Proactive measures to prevent disease have been elucidated and are reviewed here. It is to be noted, however, that the preventative steps that are presented here cannot always be accomplished.
Individual actions
Exposure limiting steps that can be undertaken include those the individual can act on and those that society must take. One can control one's own lifestyle by addressing diet; immediate environment (home and work); avoiding the use of toxic chemical-containing products, avoiding the use of pesticides, avoiding the use of tobacco and tobacco smoke and secondary exposure to smoke, avoiding foods and personal care products that contain phthalates, bisphenol A and lipophilic preservatives such as triclosan, butylated hydroxy anisole (BHA), butylated hydroxy toluene (BHT) and parabens and limiting, to the extent that is medically advisable, the use of lipophilic pharmaceuticals. The individual can also choose to restrict the use of plastics containing phthalates and bisphenol A.
Societal actions
Steps that society can take include; educational programs and regulatory control of tobacco use, pesticides, persistent organic pollutants, plastics that exude phthalates and BPA and foods and personal care products that contain toxic components such as preservatives and solvents [25,207,208]. The development of green, non-polluting energy can greatly reduce toxic exposures by eliminating the release of lipophilic hydrocarbons, mercury, cadmium and other metals as well as by preventing global warming. Warmer environmental temperatures enhance the volatilization of POPs, pesticides and other organics into the air, require increased use of pesticides in farming, increase the rates of chemical reactions that lead to higher levels of ozone and secondary air pollutants and increase the risk of wildfires which spew large quantities of polluting species into the air [209,210].
Limitations to prevention
The measures to prevent ENVDs just discussed cannot always be implemented. Ignorance, socioeconomic status, lifestyle, peer pressure and economic interests of chemical manufacturers and polluters act counteractively to prevent the limiting of chemical exposures and ROS and RNS formation. There are also conflicting situations where well meaning people on both sides of an issue can reasonably disagree. Two examples of such situations serve to illustrate this point. DDT and its metabolite DDE are persistent organic pollutants that are causative of the ENVDs in Table 1. DDT, however, is still being used in parts of the world to control mosquitoes that carry malaria and other infectious diseases [211,212]. Water is routinely disinfected with chlorine and thus spares millions of people from exposures to water borne pathogens. Yet, the chlorinated and brominated hydrocarbon, haloketone and haloacetic acid byproducts of chlorination are human toxins [213,214]. These examples illustrate the often difficult choices that need to be made with regard to the use and subsequent human exposures to toxic chemicals.
Mediterranean diet
Reduction of ROS and RNS formation in the body can be readily accomplished through diet adherence to the Mediterranean diet, so named because it is the diet adhered to by populations bordering the Mediterranean Sea. This diet is rich in antioxidant phytochemicals, including phenolics, alkaloids, nitrogencontaining compounds, organosulfur compounds, phytosterols and carotenoids. More than 5,000 dietary phytochemicals have been identified in fruits, vegetables, whole grains, legumes, red wine, nuts and vegetable oils. An excellent review of the sources and benefits of dietary phytochemicals is presented by Liu [215]. In addition to the consumption of foods containing phytochemicals, the Mediterranean diet includes an increase in fish consumption, rich in antioxidant omega fatty acids, and a reduction of red meat consumption, a major source of serum triglycerides in which exogenous lipophiles accumulate.
Olive oil, and particularly extra virgin olive oil (EVOO), which is rich in phenolics, is the most representative food of the Mediterranean diet. Tree nuts are another major component of the diet. Both have been shown to reduce inflammation, oxidative stress and lower the risk of developing many ENVDs. These diseases include; Alzheimer's disease, Parkinson's disease, hypertension, diabetes, coronary heart disease and other cardiovascular diseases, obesity various cancers, anxiety and other central nervous system disorders [215-236].
Other preventive measures
The long onset times for ENVDs present opportunities to detoxify and otherwise protect the body of absorbed lipophiles and metals. Sauna has been demonstrated to reduce body levels of POPs and other lipophilic species and to lead to a relief of disease symptoms [237-239]. Detoxification by isolating patients in non-polluting environmental chambers along with diet control has been shown to result in reduced blood levels of volatile organic hydrocarbons as well as in the relief of disease symptoms [10,240]. Heavy metals can be removed via chelation by dietary components. Vitamins B1, B6, C and E have been shown to chelate heavy metals, as have garlic, ginger, onion, green tea, curry, tomatoes and other foods [241]. Selenium is an essential metal in the human body. When ingested in its selenite oxidation state, it has been shown to protect against metal induced neurotoxicity, renal toxicity and various cancers [242-244].
Lipophilic toxicity
Aromatic versus aliphatic compounds
The discussion presented here points out the need to avoid exposures to aromatic hydrocarbon-based lipophiles. It is well established that aromatic hydrocarbons are more toxic than aliphatic hydrocarbons of equal carbon number [245-247]. This may be seen from a comparison of blood: air partition coefficient (PC) and permissible exposure level (PEL) data. A higher PC indicates a greater propensity to absorb into blood and a lower PEL is indicative of greater toxicity. Table 5 demonstrates this by comparing PC and PEL data for C6-C8 hydrocarbons [87], reprinted with permission]. As can be seen from the data, aromatic hydrocarbons are more readily absorbed into blood and are more toxic than aliphatic hydrocarbons of equal carbon numbers. In addition, aromatic species, unlike aliphatic ones, can pi-bond with transition metals and thus facilitate their absorption.
| Carbon number | Compound | PC | PEL |
|---|---|---|---|
| 6 | benzene | 7.8 | 1 |
| cyclohexane | 1.3 | 300 | |
| 7 | toluene | 15.6 | 300 |
| n-heptane | 1.9 | 500 | |
| 8 | p-xylene | 28.4 | 100 |
| n-octane | 3.1 | 500 |
Table 5 Blood: air partition coefficient (PC) and permissible exposure level (PEL) data (given in parts per million) for C6-C8 hydrocarbons. The aromatic compound is listed first in each carbon number pair [87].
It is has been hypothesized [87] that the greater toxicity of the aromatic compounds is due to two factors. Firstly, their higher solubility in blood, from which these compounds can partition into membrane lipid bilayers. Secondly, the transport of transition metal ions dissolved in blood into cell structures via metal: pi-bonding with the planar aromatic species, as described above. Transition metal ions are always present in blood. Iron and some other transition metal ions, as also described above, are essential and naturally absorbed by the body form healthy foods. Non-essential transition metal ions enter the blood stream via inhalation, ingestion and dermal absorption. Accordingly, when aromatic hydrocarbons are absorbed into the blood, they can complex with the transition metals via pi-bonds and the complexed species absorbed through cell membranes, as described above, with resultant higher toxicity than is observed for aliphatic compounds which do not complex with transition metals.
Predicting environmental disease onset
Symptoms
ENVDs are, to a great extent, silent illnesses, in that in many instances, they are devoid of symptoms in their early stages which would alert the individual and/or the physician of the need to take action. As was seen above, dose response relationships between lipophilic body loads as well as between transition metal body load are associated with ENVD onset. For example, serum levels of POPs, including PCBs and OCs, associated with T2D have demonstrated a dose-response relationship between serum concentrations of these and the onset of disease [34,35,248,249]. Safe serum levels of these toxins, however, are yet to be established. Adipose tissue POPs concentrations have also been associated with the onset of T2D in a dose-response relationship [250]. These data, however, do not predict at what levels these pollutants in blood and adipose tissue are safe. These data also do not take into account the blood concentrations of other exogenous lipophilies nor transition metal concentrations. As was seen above, the chemicals associated with the onset of ENVDs are ROS/RON producing species, as are radiation, pathogens, aging and psychological stress. Accordingly, it is hypothesized here that total OS in the body is a better predictor of the potential for the onset of ENVDs than concentrations of individual chemical species alone and that biomarkers for total ROS are expected to better predict a person's likelihood of the onset of illness.
Biomarkers
Numerous biomarkers have been identified as indicators of exogenous toxic chemical-induced oxidative stress and have been related to the onset of cardiovascular, respiratory, autoimmune, musculoskeletal, liver, kidney and gastrointestinal diseases, as well as to diabetes, obesity and nervous system disorders [160,251-264]. Table 6 lists some of these biomarkers. Research to identify a single or combination of several biomarkers that would accurately predict increased probabilities of disease onset is ongoing in molecular epidemiology, a discipline devoted to the application of biomarkers to epidemiological studies [254].
| Homocystine |
| Total glutatione |
| Oxidized low density protein |
| Conjugated dienes |
| White blood cell count |
| Cortisol |
| C-reactive protein |
| Fibrinogen |
| Malondialdehyde |
| Diminished glomerular filtration rate |
| Increased ceatinine |
| Elevated fasting glucose |
| Low LDL cholesterol |
| High LDL cholesterol |
| Urinary aromatic hydrocarbon metabolites |
| Urinary phthalate metabolites |
| Bisphenol A metabolites |
Table 6. Partial list of biomarkers those are indicative of oxidative stress [251-262].
Future Challenges
New disease causative agents
New chemical causes of oxidative stress are constantly arising. Examples are: rare earth exposure as a result of the widespread use of these elements in electronic components [265]; and nanoparticle exposures as new uses for these are found on an almost daily basis [266,267].
Pharmaceuticals
The dramatic increase in the use of pharmaceuticals, in no small part, due to the exponential growth of ENVD prevalence, in itself contributes to the increase of the incidence of ENVDs. Two examples illustrate this point. Statins are widely prescribed to protect against cardiovascular disease. However, the most commonly prescribed Statins are themselves lipophiles which have been associated with the increased onset of type 2 diabetes [268,269]. Second-generation antipsychotic drugs are widely prescribed for children with attention deficit hyperactivity disorder (ADHD). These lipophilic drugs have been associated with an increased incidence of metabolic syndrome in children who have taken Quetiapine and Risperidone [270].
Global warming
No discussion of OS would be complete without a consideration of the effects of global warming on it. It is beyond the scope of this review to thoroughly examine this topic. Following, however, are some of the OS implications of global warming, which is a major contributor to OS for several reasons. First: elevated temperatures increase the solubility of PAHs and other lipophiles in water and also increase the vaporization of these species and hence result in higher air concentrations [271]. Second: elevated environmental temperatures accelerate the absorption of toxicants through respiratory, cutaneous and gastrointestinal routes and hence increase toxicity [210,271-273]. Third; elevated temperatures increase thermal stress, impair heat stress response and delay unfolded protein recovery [274-276]. Finally, warmer temperatures increase PAH-containing smoke from wildfires, airborne pollen and mycotoxin-producing molds, all of which stimulate OS [209,277].
Summary
The onset of environmental disease requires the penetration of toxicants through lipophilic cell membranes. Exogenous lipophilic chemicals not only accomplish this step, but also facilitate absorption of hydrophiles which serve as solvents and carriers. Lipophiles which have flat aromatic structures with pi-electron clouds also act as carriers for transition metal ions which pi-bond to these species. Disease results from OS due to the action of ROS/ RNS formed via the action of absorbed hydrophiles, hydrophilic metabolites of lipophiles and/or metals. Late onset of ENVDs is due to the slow deterioration of the body's immune system with age and the need for a buildup of sufficient lipophilic, hydrophilic and/or metal ion loads to overwhelm the body's ability to metabolize and eliminate such exogenous chemicals and to repair cellular damage. Absorption of lipophiles and metals need not be one-time events. These can be absorbed sequentially over time to build critical load levels. Sequential absorption points out the need to severely limit exposures to exogenous toxicants and for the drastic lowering of permissible levels of exposure to these chemicals. Exposures to aromatic hydrocarbons, which can undergo metal to pi-cloud bonding, and hence are more toxic than aliphatics hydrocarbons, should be severely restricted.
OS, which is the ultimate cause of ENVD, results not only from toxic chemical exposure, but also of the action of pathogens, exposure to radiation, aging and psychological stress. These causes are additive and when addressing OS, the total of all of these causes require consideration.
Globally, human exposures to exogenous chemicals are growing at exponential rates. This is manifest by not only increased prevalence of ENVDs, but also by increasingly lower ages of onset for type 2diabetes, obesity and cardiovascular diseases among others.
There are steps that can be taken to lower the incidence of ENVDs, but complete prevention of ENVDs is impossible, given the wide spread distribution of the causative chemicals and genetic predispositions. Biomarkers that determine total OS in the body would provide predictive indicators of increased vulnerability to the onset of ENVD and enable individuals to act to reduce total OS and hence disease.