Research Article - (2018) Volume 4, Issue 2
Kodolov VI1,2* and Trineeva VV1,3
1Basic Research - High Educational Centre of Chemical Physics and Mesoscopy, Ural Division of Russian Academy of Sciences, Izhevsk, Udmurt Republic, Russia
2M.T. Kalashnikov Izhevsk State Technical University, Studencheskaya, Izhevsk, Russia
3Institute of Mechanics, Ural Division, Russian Academy of Sciences, Izhevsk, Russia
*Corresponding Author:
Kodolov VI
Basic Research - High Educational Centre of Chemical Physics and Mesoscopy
Ural Division of Russian Academy of Sciences
Izhevsk, Udmurt Republic, Russia
Tel: +34-915 622 900
Fax: 3412-50-79-59
E-mail: vkodol.av@mail.ru
Received Date: June 04, 2018; Accepted Date: July 04, 2018; Published Date: July 11, 2018
Citation: Kodolov VI, Trineeva VV (2018) Chemical Mesoscopics as New Trend in Chemical Sciences. Polym Sci Vol.4 No.2:10
DOI: 10.4172/2471-9935.100038
The definition and theoretical fundamentals of new scientific trend – chemical mesoscopics are considered on the example of nanostructure new class – metal/carbon nanocomposites. The theoretical and experimental methods for the processes direction prognosis as well as for the chemical systems reactivity estimation are discussed. The hypothesis of mesoscopic metal containing cluster creation at mechanic-chemical formation of metal/ carbon nanocomposites is proposed. The electron structures of carbon shells for metal obtained clusters are determined. The metal nature influence of metallic phase cluster on the carbon shell is shown. The changes of media electron structure are possible under the nanocomposites minute quantities influence that it is confirmed by the X-ray photoelectron spectroscopic investigations and also is explained by the Chemical Mesoscopics principles. It’s shown that the orientation processes, in which mesoscopic particles (nanocomposites) participate, lead to the changes of sub molecular structures of polymeric compositions.
Keywords
Chemical mesoscopics; Self organization; Mechanism of RedOx; Synthesis within nanoreactors; Charge quantization; Mechanism of nanostructures
Introduction
At present we observe the creation of new synthetic science on the base of synergetic, fractal theory, chemistry within nanoreactors and mesoscopic physics. Everyone from these scientific trends consider the objects of nano sized level (0.1- 1000 nm) and investigates near phenomena. These phenomena take place at definite conditions (size of phase coherence must be less than 1000 nm).
From the comparison of the notions and fundamentals for above named scientific trends it's possible to speak about the creation of new scientific trend or new science – the chemical mesoscopics.
Thus, the definition of chemical mesoscopics is possible as:
Chemical mesoscopics is the science which study the reactivity of nano (meso) particles and also nanosize systems from their formation moment to the active behavior in media and materials [1]. Theoretical fundamentals of this new science give the explanations for peculiarities of processes within nanoreactors and also the influence of nanostructures minute quantities on media and compositions.
Below the some fundamentals of this scientific trend are considered on the example of metal/carbon nanocomposites with its application for the modification of polymeric compositions.
Conditions for Processes in Chemical Mesoscopics
The restrictions for processes defined by the limits of Chemical Mesoscopics are concluded in following:
• The mesoscopic particle which may be presented as big molecule or the linked molecules group is found in the active interaction with medium.
• In this case the size of phase coherency is located in limits up to 1000 nm
• Then the phenomena such as interference, spectrum quantization and charge quantization are appeared.
• In other words, there is the source of quant radiation which activate the certain functional (active) groups in medium is formed when nanosized particles and also confined space take place.
The aforesaid restrictions are at the metal/carbon nanocomposites obtaining. Let us consider the obtaining of Copper/and Nickel/Carbon nanocomposites from metal oxides and polyvinyl alcohol (PVA).The mechanochemical obtaining of metal/carbon nanocomposites are considered in detail in papers [1-5]. According to this paper it’s possible to conclude that the redox process of nanocomposites obtaining takes place already at the joint grinding PVA and metal oxide, and the further heating (no more 400° C) of reactive mass (xerogel) only secure the process result [1,6]. Metals are reduced with the change of system electron structure. This is confirmed by spectroscopic and diffracted investigations. Chemical Mesoscopics explains these processes by the nanoreactor formation, in which the metal oxide cluster formed is coordinated with hydroxyl groups of PVA and promotes to its separation as well as the Hydrogen separation from polymeric chain. Then in this processes the electron flow from nanoreactor walls across the cluster appeared that leads to the change of metal oxidation state. As the result of RedOx processes, the metal clusters are formed within shells which contain fragments of poly acetylene and carbine chains. Those shells actively interact with d metals and initiates to d electrons transition of metals on more high energetic levels that promote the magnetic characteristics increasing.
Below the diffract gram of nanocomposites (NC) obtained are given for the confirmation of metal phase formation (Figures 1, 2, Tables 1 and 2) [6].
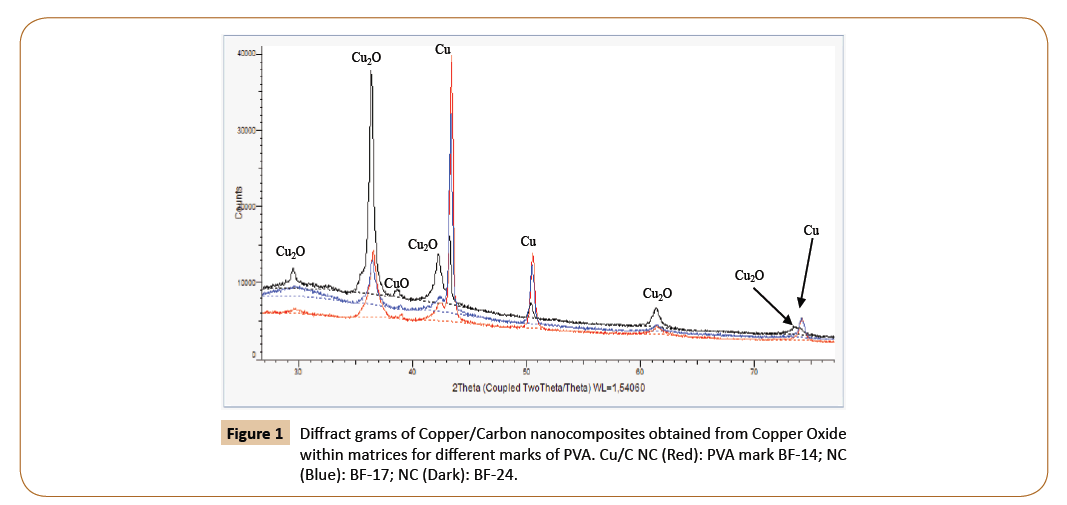
Figure 1: Diffract grams of Copper/Carbon nanocomposites obtained from Copper Oxide within matrices for different marks of PVA. Cu/C NC (Red): PVA mark BF-14; NC (Blue): BF-17; NC (Dark): BF-24.
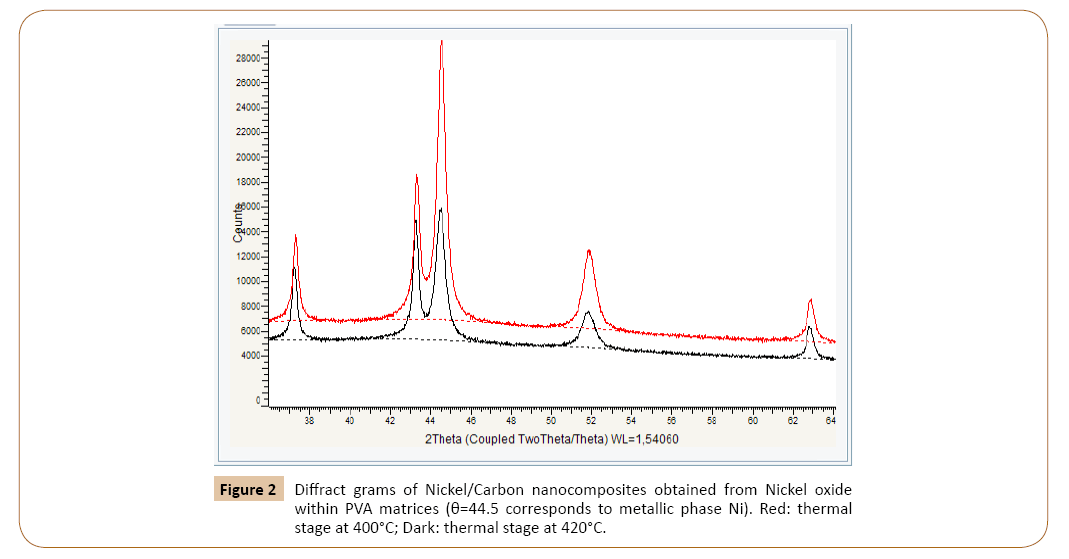
Figure 2: Diffract grams of Nickel/Carbon nanocomposites obtained from Nickel oxide within PVA matrices (θ=44.5 corresponds to metallic phase Ni). Red: thermal stage at 400°C; Dark: thermal stage at 420°C.
Table 1: The influence of process temperature on the quantity metallic Nickel in nanocomposite.
| The curve color on diffract grams of Nickel/Carbon nanocomposite | I(NiO)/I(Ni) | The maximal temperature (°?) |
|---|---|---|
| Dark | 0.94 | 420 |
| Red | 0.62 | 400 |
Table 2: The influence of PVA marks on process completion.
| Marks of PVA | The cuC line color on diffract grams of Copper/Carbon nanocomposite | I1350/I1580 |
|---|---|---|
| PVA mark BF-17 | blue | 0.77 |
| PVA mark BF-14 | red | 0.82 |
| PVA mark BF-24 | dark | 0.72 |
As the addition to Figures 1 and 2, the Tables 1 and 2 are presented.
The First table illustrates the influence of process temperature on the RedOx process quality (perfect). According to data of diffract grams (Figure 2) the temperature increasing upper 400°C leads to the decreasing of the metallic Nickel at the obtaining of Nickel/Carbon nanocomposite. This fact can be explained by the disturbance of metal coordination with the nanoreactors walls.
The Table 2 shows the dependence of process result from the nature of polymeric matrix or marks of PVA.
The comparison of these diffract grams shows that the RedOx processes of obtaining of Copper/Carbon nanocomposites proceeds more perfect then analogous processes for Nickel/ carbon nanocomposites obtaining.
Figures 1 and 2 show that the Copper Oxide completely participates in reduction process. At the same time the Nickel Oxide is reduced only on 65% (at 400°C). This fact is confirmed by x-ray photoelectron spectroscopy investigations. In C1s spectra there are lines correspondent to the C1s energy for CH groups. Wonderfully, but according to C1s spectra for Nickel/Carbon nanocomposite the content of CH groups equaled to 35%.
It is necessary the absence of progressive and rotatory motions and the decreasing of vibration mode possibilities for the creation of processes realization conditions according to principles of Chemical Mesoscopics. When these conditions are secured, the transport “quantized electrons” takes place across the nano(meso)particles. In order to this process promotes to proceeding of RedOx processes. These conditions may be possible only at the correspondent sizes and forms of nanoreactors and also at the interactions of nanoreactors walls with clusters created within these nanoreactors.
What are Nanoreactors and How the Correspondent Processes Realize within Nanoreactors?
About the nanoreactors formation in media
What are nanoreactors? Let us give the definition for this notion.
Nanoreactors are space limited part in which reactants chemical particles are orientated about each other for the formation of transitional state before nanoproduct creation [1,6].
It's known [7] the following classification of nanoreactors:
One-dimensional nanoreactors: In the clearance between probe and surface, Crystal canals, Complexes, Crystal solvates, Macromolecules, Micelles, Vesicles, Pores.
Two-dimensional nanoreactors: The double electrical layer, Monomolecular layers on surface, Membranes, Interface layers (boundaries), Adsorption layers.
Cluster nanoreactors (Metal clusters): Thus, the great variety of nanoreactors, formed at different conditions, takes place. Then it’s necessary to note that the formation of correspondent activated complex, which protects the process direction, is relieved within nanoreactors. The charge quantization into medium promotes the process evolution during the certain time interval. Also it’s noted that the formation of nanostructures within nanoreactors are determined by the nature of reactants, which participate in synthesis, and by the energetic and geometric characteristics of nanoreactors.
The analysis of modern scientific data and our experiments shows that the following peculiarities of nanostructures formation within nanoreactors may be noted:
• The principal peculiarity - the decreasing of collateral parallel processes and the process direction to special product side.
• The low energetic expenses and the high rates of processes.
• The dependence of obtained nanostructures properties from the energetic and geometric characteristics of nanoreactors.
What Basic Parameters and Equations Can Be Used for the Nanostructures Formation within Nanoreactors and What Matrices are Possible?
Firstly, the Coordination Number of Element or Elements group necessary to take into account for the definition of process direction. In this case it’s possible the application of multi plate theory.
Secondly, the chemical potential difference is basic factor for the self-organization process start.
Thirdly, the growth and form for nanostructures can be determined with the using of Kolmogorov-Avrami equations.
As the bright example of the Coordination Number influence on nanostructures formation can be present [8] by the Nan rotors formation based on Zinc Oxide systems (ZnO)m with the addition of Indio Oxide (In2O3). The Zinc Coordination number equals to 4. Therefore the ribbon formation can be equated explained. The In Coordination Number equals to 6.Then the possibility of six rabbets contained at rotor formation takes place when it is included in the chain of ZnO system (Figure 3).

Figure 3: Nan rotor with 6 rabbets on base of ZnO and additive In2O3
However let us return to metal/carbon nanocomposites in which metals can be Copper or Nickel. The Coordination Number of Copper may be 2 or 4 in the dependence of Metal Oxidation State (for +1-2; for +2-4). At the same time, the Nickel Coordination Number may be 4 or 6 in the dependence of Ligand nature. The formation of carbon shell from PVA chains starts off coordination process of four chain fragments with the metal containing cluster. When the dehydration and the dehydrogenization proceed, the connected fragments of polyacetylene and carbine are appeared. The fragments formed are coordinated on the metal. The electron structure of carbon fibers formed is investigated by x-ray photoelectron spectroscopy [9].
According to C1s spectra three types of satellites (sp, sp2, sp3 hybridization) with different relation between them are found. These satellites also take place in C1s spectrum. The intensities relation as Isp2/Isp3 for Cu/C nanocomposite corresponds to 1,7, and this parameter for Ni/C nanocomposite equals to 1. It’s known [8] that the satellites intensities relation also equals to 1. Really, according to PEM data the Nickel/Carbon nanocomposite contains tubular nanostructures. The development of the nanostructures self-organization process is estimated by means of the equation based on Kolmogorov-Avrami equations [10]. In this equation the RedOx potentials are used because of the metal reduction occur in process of Metal/Carbon nanocomposite formation:
W=1-k exp[-τnexp(zFΔφ/RT)], (1)
where k: Proportionality coefficient considering the temperature factor, τ: Duration of process, n: Fractal dimension, z: Number of electrons participating in the process, Δφ: Difference of potentials on the border “nanoreactor wall reaction mixture”, F: Faraday number, R: Universal gas constant, T: Temperature. The calculations are made when the process duration changing takes with a half-hour interval. It was accepted for calculations that n equals 2 (two-dimensional growth), potential of Redox process during the metal reduction equals 0,34V, temperature equals 473K, Faraday number corresponds to 26.81 A hr/mol, universal gas constant (R) equals 2.31 W.hr/mol?degree. Calculation results take in Table 3.
Table 3: The calculations results of process duration for Copper/Carbon nanocomposite obtaining.
| Duration (hrs) | 0:50 | 1:00 | 1:50 | 2:00 | 2:50 |
| Content of product (%) | 22.5 | 63.8 | 89.4 | 98.3 | 99.8 |
The calculation results practically coincide with experimental data. The comparison of experimental results obtained at metal/carbon nanocomposites synthesis with calculated results obtained by means of modified Kolmogorov-Avrami equations is discussed in full in paper [11].
The coordination processes lead to the changes of metal electron structure with unpaired electrons formation. That is accompanied by the metal atomic magnetic moment increasing, and also the appearance of unpaired electrons on the carbon shell surface (Table 4).
Table 4: Experimental EPR data and atomic magnetic moments (μB) for Copper and Nickel/Carbon nanocomposites [10].
| Type of metal/carbon nanocomposite | g-factor | Number of unpaired electrons, spin/g | Atomic magnetic moment, μB Metal / carbon nanocomposite/ Massive sample |
|---|---|---|---|
| Copper/Carbon nanocomposite | 2.0036 | 1.2 × 1014 | 1.3/ - |
| Nickel/Carbon nanocomposite | 2.0036 | 1022 | 1.8/0.5 |
Thus, the Metal/Carbon nanocomposites, obtained within PVA nanoreactors from Metal Oxides, represent active nanoparticles,
which can be source of electron quants for media and compositions. According to Chemical Mesoscopics principles the minute quantities of these nanoparticles (with size less 100 nm) can essence improve the materials properties.
The Influence of Nanostructures on Media and Compositions
When the additive sizes are decreased to nanometer sizes the phenomena of mesoscopic particles are appeared. According to studies [12-14] the phenomena such as interference, spectrum quantization, charge quantization occur when the mesoparticles have the limitation in motions or in the energetic possibilities realization. In this case the nano mesoparticles can only vibrate, and also the electron transport is possible across them.
The media (or polymeric compositions) properties changes under the mesoparticles influence can be achieved at equal distribution of these particles in composition volume and at its coagulation absence. Last is possible at the following conditions:
• Certain polarity and dielectric constant of medium;
• Minute concentration of mesoparticles;
• Ultrasound action on the correspondent suspension for the proportional distribution of mesoparticles.
The assignment of active nanostructures (mesoparticles) during the compositions modification is concluded in the activation of matrices self-organization in needful direction. For the realization of this goal the determination of organized phase part is necessary. The Jonson-Mel-Avrami-Kolmogorov equation is applied for the organized phase determination:
W=1-exp(-kτn), (2)
Where W: The part of organized phase, k: The parameter defined the rate of organized phase growth, τ: The duration of organized phase growth, n: The fractal dimension.
The consideration of metal/carbon nanocomposites reactivity is interesting in processes of interaction with different substances and media.
The presence of double bonds and unpaired electrons promotes to the interactions of Phosphorus, Silicon and Sulphur substances with nanocomposites. As the result of this interaction the reduction of correspondent elements takes place accompanied by the growth of metal atomic magnetic moments for nanocomposites and the increasing of their polarization action on media. For instance, after the silicon introduction into Copper/ Carbon nanocomposite its properties are changed:
• Copper atomic magnetic moment grows to 3 μB.
• Antioxidant activity increases in 11 times.
• Dynamic viscosity of liquid glass, which contains 0,001% of Cu/C nanocomposite with Silicon, is increased on 21%.
These experimental results show the possibility of nanocomposites interaction with active media and substances. The illustrations of that possibility realization can be the investigations of nanocomposites fine dispersed suspensions.
The quantum charge wave expansion leads to the functional groups polarization (dipole moments) change as well as the extinction increasing. Last bring growth of peaks intensities in IR spectra. The individual peaks growth effects in IR spectra are observed at the introduction of nanocomposites minute quantities.
In some papers [15-17] the positive results on materials properties improvement are presented when the minute quantities of metal/carbon nanocomposites are introduced in these materials. In paper [17] the hypothesis about nanostructures influence transmission on macromolecules of polymeric matrices is proposed. This hypothesis is complied with Chemical Mesoscopics principles which consider quantum effects at the certain conditions of nano mesoparticle existence. In this case the peaks intensity growth in IR spectra is observed when the quantity of introduced nanocomposite is decreased. This fact is complied with fundamental principle, when there is excess of vibration above rotation motion. The nanocomposite action efficacy estimation in fine dispersed suspension on medium is carried out by means of the relation comparison of peak intensity to its half width.
This relation reflects according to Herzberg-Kondrat’ev equation excess of vibration above rotation motion:
ν=ω + B[J’(J’ + 1) - J(J + 1)] (3)
where ν: The vibration frequency of correspondent group, ω: The value of vibration motion lot, B: The value of rotation motion lot, J’ and J: The rotation quantum numbers. If this relation is increased, then the rotation motion lot will be less in system.
The second significant factor for active interaction characteristics into media appear the change of special model films electron structure. These films are obtained on the base of linear functional polymers such as poly vinyl alcohol (PVA), poly methyl met acryl ate (PMMA), polycarbonate (PC). The polarity of polymers increases in row from PVA to PC. The “stamp” of nanocomposite (NC) is characterized by sp, sp2, sp3 hybridization satellites and it’s appeared in C1s spectra of polymeric films at concentration intervals of NC in following order: for PVA at 10-3% NC; for PMMA-interval 10-2-10-4% NC; for PC-interval 10-2-10-5% NC. Besides it is noted [8] that the changes for relations Isp2/Isp3 take place. In these experiments the illustration of bond between electron structure and sub molecular structure of polymeric substances is presented. The analysis of adduced examples gives the basis for hypothesis about the propagation electron quants as direct flow or diffuse radiation in the dependence on the nanocomposite concentration and on the nature of NC, and also medium nature (magnetic and electric characteristics).
It's interesting to observe the direction of carbon fibers in comparison with the direction of sub molecular structures orientation in the nanostructured polycarbonate surface layers.
Thus, it's possible that the wave which initiates self-organization process in polymeric composition is expensed from these fibers associated with metal cluster. The last leads to the correspondent orientation of sub molecular structures in nanostructured composite surface layers.
The self-organization mechanism for polymeric compositions modified by the metal/carbon nanocomposite minute quantities is concluded in the conditions creation for composition polarization, which bring the great change of electron and sub molecular structures of materials. Certainly, these structures changes influence on the modified materials properties. The sufficient large quantity of experimental works carried out on the modification of different materials with the using of metal/carbon nanocomposites. These experiments confirm the improvement of materials properties on account of their modification by the minute (less 0.01%) quantities of metal/carbon nanocomposites.
Below the examples of results for modification with using of above considered suspensions or nanocomposites concentrates will be presented.
For example, it’s noted the decreasing of thermal conductivity in 1.5 times for polycarbonate, modified by 0.01% of Copper/ Carbon nanocomposite as well as the increasing the light radiation transmission in the field 400-500 nm at the decreasing in the field 560-760 nm.
The following example, it’s shown that polyvinyl chloride film, modified by 8.10-4%, has electrostatic properties.
Other instance, the increasing of adhesive durability more than 50 % is defined for glues nanostructured by 1-3.10-4% of Copper or Nickel/Carbon nanocomposites.
For the current conductive glues and pastes is observed the increasing of adhesion durability and the decreasing of electric resistance (Table 5) [15-17].
Table 5: The comparison of electric resistance for modified glues and pastes and their modified analogous.
| Parameters | The current conductive paste | The current conductive glue |
|---|---|---|
| Specific volume resistance for non-modified sample, Om?cm | 2,4 × 10-4 | 3,6 × 10-4 |
| Specific volume resistance for modified sample, Om?cm | 2,2 × 10-5 | 3,3 × 10-5 |
The introduction of metal/carbon nanocomposites in quantity equaled to 0.005% in epoxy resins leads to the increasing of thermo stability on 75°-100°.
For reinforced glass plastics the introduction of 0.02% Copper/ Carbon nanocomposite leads to the increasing of durability on 32.2%.
There are many examples of the materials properties improvement owing to the using of the metal/carbon nanocomposite minute (or super small) quantity.
Perspectives of Chemical Mesoscopics Development
One from fundamental points of new scientific trend development appears the creation of mathematical prognostic program, in which there is place for the theory of formation and application of nanoreactors which promote to processes proceeding of useful nanostructures or nanosystems obtaining. In this theory it’s necessary to determine the correspondent conditions for the formation of nanoreactors with the same geometric and energetic characteristics.
Certainly, theoretical apparatus for kinetics of processes within nanoreactor requires development, as well as the determination of peculiarities in the dependence on nanoreactor type. The separate very important problem is the mechanism of the quant radiation formation. In other words, what conditions are necessary for the creation of the charge (electron) quantization, and also the interference of quant waves with the formation of new oscillators which will promote the wave propagation into media.
With the development of different theories the correspondent trends in technological fields take place, especially technologies of nanostructures (nanosystems) producing as well as the manufactory of nanostructured materials, obtained owing to the modification of compositions by nanoproducts including metal/ carbon nanocomposites.
The proposed technologies on the basis of chemical mechanics, electrochemistry and chemistry on surface or processes on the phase boundaries are needed in the development of theoretical apparatus for results prognosis [18,19]. At the same time it’s necessary to develop experimental methods of processes control, and also the estimation of results obtained during the processes of nanoproducts obtaining.
The development of this new scientific trend will be more full at universal studies of nanostructures (nanosystems) properties such as catalytic and stimulator of processes, synergetic, quantum nano generator, etc. Simultaneously it’s necessary to increase the quantity of various (on content, structures and properties) nanoproducts including fine dispersed suspensions and nanostructures concentrators.
Conclusion
The unification of four scientific trends such as synergetic, fractal theory, chemistry within nanoreactors, mesoscopic physics, is proposed on the base of its comparison on investigated objects and observed phenomena. In this case the new science could be named as Chemical Mesoscopics. Some peculiarities and regularities of this new scientific trend on the instance of creation, characteristics investigations and application of nanostructure new class such as metal/carbon nanocomposites are discussed. On the base of discussion the following it may conclude that Chemical Mesoscopics considers the nano or mesoparticles reactivity, and also the processes with these particles participation.
The development of Chemical Mesoscopics supposes the theory development for mechanisms of nanosystems formation as well as mechanisms of synergetic and self-organization in media and compositions.
Certainly, the new scientific development will be hard up the creation of new methods for the processes investigations, and also for nanostructures properties studies. The extensive applications of new science results will be expected in practice. Thus, inasmuch as chemical mesoscopics is devoted to the nanoparticles reactivity and is based on ideas about wave nature of world, including charge quantization, spectrum quantization, interference and annihilation, then the possibilities are opened for the board of processes and the production of materials and substances with defined characteristics. In this trend the positions of multiples theory, and also the explanations from point of view of chemical mesoscopics for the processes within nanoreactors as well as positive effects at materials modification by means of minute quantities of active nanostructures are confirmed.