Canverenler E1*, Buke B2,Akkaya H2, Akarsu S3, Ahmet IZ3 and Canverenler S4
1Department of Obstetrics and Gynecology, Sinop Ataturk State Hospital, Sinop, Turkey
2Department of Obstetrics and Gynecology, Kayseri Education and Research Hospital, Kayseri, Turkey
3Department of Obstetrics and Gynecology, Izmir University, Izmir
4Department of Radiology, Sinop Ataturk State Hospital, Sinop, Turkey
Received date: October 16, 2015; Accepted date: November 22, 2015; Published date: November 24, 2015
*Corresponding Author:
Canverenler E
Department of Obstetrics and Gynecology, Sinop Ataturk State Hospital, Sinop, Turkey
E-mail: canverenleremre@gmail.com
Copyright: © 2015 Canverenler E, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Ductus arteriosus aneurysm; Obstetrics; Fetal echocardiography
Introduction
Ductus arteriosus aneurysm (DAA) is characterized by a fusiform and saccular dilatation of the ductus arteriosus (DA) [1]. Although the true incidence of congenital DAA is still unknown, recent reports suggest it may be much more common than previously thought.
Although imaging of the ductus arteriosus (DA) by ultrasonography has been possible for many years, it is only since 1995 that DAA has been reported using fetal echocardiography. In 1995, Puder et al. described the first case of DAA detected at 39 weeks of gestation and it spontaneously closed during the neonatal period [2]. In the same year, Mielke et al. reported a case of an abnormally S-shaped kinking of the ductus arteriosus with increased systolic and diastolic Doppler flow velocities [3]. Since then, several case reports and a retrospective study have been reported.
DAA likely develops especially in the third trimester perhaps due to abnormal intimal cushion formation or elastin expression [4]. A late onset of prenatal development is perhaps due to an altered circulation, weakening of the wall of the ductus arteriosus or a combination of both [5]. Infants with large for their gestational age, poorly controlled diabetic mothers and mothers with blood group A also have a high risk of DAA [6]. In addition, it may be more commonly observed in patients with connective tissue disease. When combined with connective tissue disease, DAA has the highest risk of spontaneous rupture and surgical resection must be considered [7].
It can be potentially fatal because of its association with severe complications. Aneurysmal rupture, dissection, thromboembolism, compression of the adjacent structures, infection secondary to bronchial obstruction, left pulmonary artery stenosis and sudden death have all been reported in the postnatal period [7]. Although DAA can be associated with severe complications, the majority of affected fetuses have been asymptomatic at birth. On the other hand, although DAA usually resolves spontaneously after birth following closure of the DA, the association of chromosomal anomaly and connective tissue disease cannot be ignored [8].
We have prepared a case report on this important issue.
Case Report
A 25-year-old pregnant woman (gravida 2, para 1) have been referred to our hospital with the diagnosis of oligohydramnios. She did not have any medical or surgical condition and there were no obstetrical complications. Both nuchal translucency and quad tests were all within the normal limits. A fetal US examination was performed to check fetal growth and measure the amniotic fluid index. At that time, a fusiform dilatation of the DA was incidentally detected. Fetal echocardiography was performed with a Mindray DC-7 scanner equipped with transabdominal 4-8 MHz curvilinear probe and the DAA was confirmed on the three-vessel view of the fetal heart (Figure 1). Its diameter was 12 mm.
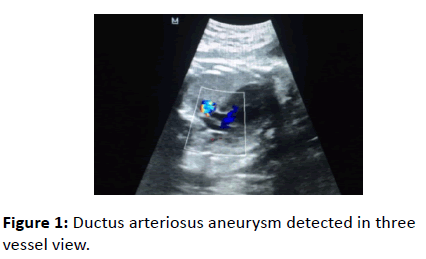
Figure 1: Ductus arteriosus aneurysm detected in three vessel view.
Since the original description of the three-vessel view, there have been other reports confirming its usefulness and expanding its diagnostic role in fetal echocardiography. With slight angulation of the transducer more anatomical details are demonstrated, including the transverse aorta, ductus, superior vena cava and trachea in the three vessels and trachea view [9,10].
The sagittal view of the ductal arch demonstrated a dilated DA at the distal end (Figure 2).
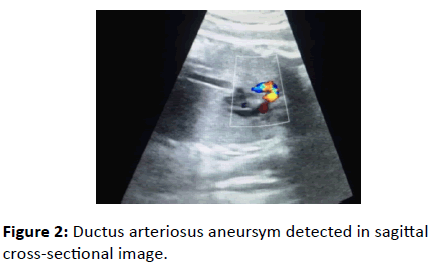
Figure 2: Ductus arteriosus aneursym detected in sagittal cross-sectional image.
On the spectral Doppler analysis, both the systolic and the diastolic flow velocities of the DAA were within the normal limits.
Turbulent flow in the DAA was detected when using color Doppler (Figure 3). There were not any other intracardiac abnormality.
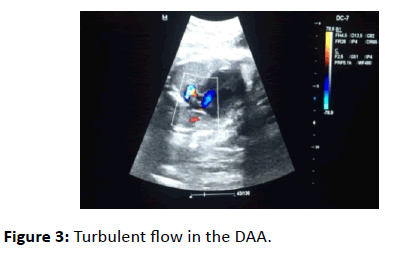
Figure 3: Turbulent flow in the DAA.
Cesarean section has been decided because of fetal distress findings in non-stress test. The asymptomatic male newborn weighted 3300 g with Apgar scores of 8 and 9 at 1 and 5 minutes, respectively. Clinical examination was normal. No other cardiac abnormalities were detected. The DAA was confirmed by postnatal echocardiography within 24 hours after birth showed a PDA with a particularly tortuous course, and a restrictive pulmonary artery end was noted with partial involution of the aneurysmal DA. Flow was left to right. Three days after birth the mother and baby were discharged in a healthy way.
Discussion
Congenital ductus arteriosus aneurysm has been considered a rare cardiovascular lesion. Although the incidence of neonatal DAA was previously reported to be 0.8%, the true incidence is unclear, depending on the criteria of diagnosis [11]. In a previous study, there were three cases of congenital DAA in a review of 200 consecutive third-trimester US examinations (1.5%) [12]. In another longitudinal series that included 509 fetuses and more that 3000 US examinations, the prevalence of congenital DAA was 2.2% [13].
DAA has been uniformally identified in the third trimester and has not been detected earlier in gestation. This suggests a late onset of prenatal development may be the result of abnormal intimal cushion formation or defective elastin in ductus arteriosus [5]. DAA may be observed in the patients with connective tissue diseases such as Marfan, Ehler-Danlos and Larsen syndromes [14]. Previous studies showed the association of trisomy 21, 13 or Smith-Lemli-Opitz syndrome with DAA [15]. High risk factors associated with DAA include newborns with large for gestational age, maternal DM and mothers with blood group A [6].
Three-vessel view is helpful on diagnosis of DAA [16]. In our case, the DAA was confirmed by the three-vessel view on the fetal echocardiography, although it was incidentally detected during routine US evaluation of the fetal growth and the amniotic fluid index.
Although the majority of affected neonates are clinically asymptomatic, several reports demonstrated potential serious complications including spontaneous rupture, thromboembolism, erosion into airways, infection, and compression of surrounding vessels, airways and nerves [17]. Lund et al. cite a 31% complication rate in their earlier review of 65 cases of DAA previously diagnosed in children under 2 months of age, although it is rarely fatal [18]. It may also be associated with chromosomal anomalies and, more importantly, connective tissue disorders which may be progressive and place the infant at higher risk of spontaneous aneurysm rupture [19].
To avoid such lethal complications, surgical intervention may be considered if
1. The ductus arteriosus with DAA remains patent beyond the neonatal period,
2. The DAA is associated with connective tissue disease,
3. There is evidence of thrombus extension into other vessels or thrombo embolism or
4. There is significant compression of adjacent structures [20].
Although it does not appear to warrant intervention in the majority of cases, it would be wise to document complete closure of the aneurysmal duct prior to neonatal discharge. Like most cases, the case presented here was asymptomatic, closed spontaneously and did not require treatment.
In addition to surgical considerations, close follow-up of the neonate is important because the majority of cases may spontaneously resolve after a period of time. The use of indomethacin has been reported to close DAA successfully in one case report; however the definitive effect of it on DAA is uncertain [21].
In summary, increased use of fetal echocardiography has detected more cases of congenital DAA. The majority of affected neonates are clinically asymptomatic and tend to progressively diminish in size of DAA and spontaneous closure, especially in cases of smaller DAA [22]. However, serious complications can occur during waiting periods indicating surgical intervention, especially in cases of persisting DAA beyond the neonatal period or associated connective tissue diseases. Finally, indomethacin treatment may be used to successfully close DAA in premature infants even though its definitive effect on DAA is still uncertain [20].
References
- Tseng JJ, Jan SL (2005) Fetal echocardiographic diagnosis of isolated ductus arteriosus aneurysm: a longitudinal study from 32 weeks of gestation to term. Ultrasound Obstet Gynecol 26: 50-56.
- Siu BL, Kovalchin JP, Kearney DL, Fraser CD, Fenrich AL (2001) Aneurysmal dilatation of the ductus arteriosus in a neonate. Pediatr Cardiol 22: 403-405.
- Mielke G, Peukert U, Krapp M, Schneider-Pungs J, Gembruch U (1995) Fetal and transient neonatal right heart dilatation with severe tricuspid valve insufficiency in association with abnormally S-shaped kinking of the ductus arteriosus. Ultrasound Obstet Gynecol 5: 338–341.
- Puder KS, Sherer DM, Ross RD, Silva ML, King ME, et al. (1995) Prenatal ultrasonographic diagnosis of ductus arteriosus aneurysm with spontaneous neonatal closure. Ultrasound Obstet Gynecol 5: 342-345.
- De DH, Pezzella AT (1995) Aneurysm following ligation of patent ductus arteriosus. Tex Heart Inst J 22: 324-326.
- Egami J, Tada Y, Takagi A, Sato O, Idezuki Y (1992) False aneurysm as a late complication of division of a patent ductus arteriosus. Ann Thorac Surg 53: 901-902.
- Jan SL, Hwang B, Fu YC, Chai JW, Chi CS (2002) Isolated neonatal ductus arteriosus aneurysm. J Am Coll Cardiol 39: 342-347.
- Acherman RJ, Siassi B, Wells W, Goodwin M, DeVore G, et al. (1998) Aneurysm of the ductus arteriosus: a congenital lesion. Am J Perinatol 15: 653-659.
- Yagel S, Cohen SM, Achiron R (2001) Examination of the fetal heart by five short-axis views: a proposed screening method for comprehensive cardiac evaluation. Ultrasound Obstet Gynecol 17: 367-369.
- Yoo SJ, Min JY, Lee YH, Roman K, Jaeggi E, et al. (2003) Fetal sonographic diagnosis of aortic arch anomalies. Ultrasound Obstet Gynecol 22: 535-546.
- Lund JT, Hansen D, Brocks V, Jensen MB, Jacobsen JR (1992) Aneurysm of the ductus arteriosus in the neonate: three case reports with a review of the literature. Pediatr Cardiol 13: 222-226.
- Maisel P, Brenner J (1999) Spontaneous closure and thrombosis of a ductal aneurysm in a neonate. Cardiol Young 9: 503-505.
- Tan TH, Wong KY, Heng JT (2000) Echocardiographic features and management of neonatal ductal aneurysm. Ann Acad Med Singapore 29: 783-788.
- Dyamenahalli U, Smallhorn JF, Geva T, Fouron JC, Cairns P, et al. (2000) Isolated ductus arteriosus aneurysm in the fetus and infant: a multi-institutional experience. J Am Coll Cardiol 36: 262-269.
- Leal SD, Cavallé-Garrido T, Ryan G, Farine D, Heilbut M, et al. (1997) Isolated ductal closure in utero diagnosed by fetal echocardiography. Am J Perinatol 14: 205-210.
- Silver MM, Freedom RM, Silver MD, Olley PM (1981) The morphology of the human newborn ductus arteriosus: a reappraisal of its structure and closure with special reference to prostaglandin E1 therapy. Hum Pathol 12: 1123-1136.
- Better DJ, Timchak DM, Allan LD (1997) Prenatal diagnosis of aneurysm of the arterial duct: postnatal management and literature review. Cardiol Young 7:160.
- Lund JT, Hansen D, Brocks V, Jensen MB, Jacobsen JR (1992) Aneurysm of the ductus arteriosus in the neonate: three case reports with a review of the literature. Pediatr Cardiol 13: 222-226.
- Jackson CM, Sandor GG, Lim K, Duncan WJ, Potts JE (2005) Diagnosis of fetal ductus arteriosus aneurysm: importance of the three-vessel view. Ultrasound Obstet Gynecol 26: 57-62.
- Tan J, Silverman NH, Hoffman JI, Villegas M, Schmidt KG (1992) Cardiac dimensions determined by cross-sectional echocardiography in the normal human fetus from 18 weeks to term. Am J Cardiol 70: 1459-1467.
- Hornberger LK (2002) Congenital ductus arteriosus aneurysm. J Am Coll Cardiol 39: 348-350.
- Dyamenahalli U, Smallhorn JF, Geva T, Fouron JC, Cairns P, et al. (2000) Isolated ductus arteriosus aneurysm in the fetus and infant: a multi-institutional experience. J Am Coll Cardiol 36: 262-269.