Research Article - (2017) Volume 25, Issue 4
Abaya Mulugeta
Department of Epidemiology and Biostatistics, Faculty of Health Sciences, Jimma University, Ethiopia
Henok Assefa
Department of Epidemiology and Biostatistics, Faculty of Health Sciences, Jimma University, Ethiopia
Tsegaye Tewelde
Department of Epidemiology and Biostatistics, Faculty of Health Sciences, Jimma University, Ethiopia
Lamessa Dube*
Department of Epidemiology and Biostatistics, Faculty of Health Sciences, Jimma University, Ethiopia
Corresponding Author:
Lamessa Dube
Departments of Epidemiology and Biostatistics
Faculty of Health Sciences, Jimma University
Jimma, P.O. Box 378, Ethiopia
E-mail: dubelamessa@yahoo.com
Submitted date: July 05, 2017; Accepted date: July 28, 2017; Published date: August 04, 2017
Background: Human Immunodeficiency Virus (HIV) infection rapidly progresses in children. If not put on a treatment nearly half of them would be dead by the second year of infection. Highly Active Antiretroviral Treatment (HAART) consistently improved survival of children infected with the virus and contributed to prevent new infections. Baseline factors that envisage survival could permit for their possible amendment in order to improve pediatric HIV care and decrease HIV-related mortality among children on the treatment. However, there are inconsistencies of finding by fragmented studies about predictors of survival among children on HAART in Ethiopia. Resolving these inconsistencies of the predictors of survival among children after initiation of HAART is important to improve the effectiveness of HAART on child survival. Therefore, this study was conducted to identify the predictors of the survival among children after initiation of ART and also rectify the inconsistency via multihospital based and longer period of follow up.
Method: A retrospective study was conducted on 757 children who started antiretroviral therapy from January 1, 2008 to December 31, 2010 and followed up until December 31, 2015 at selected public hospitals in Addis Ababa. Cox regression model was fitted to identify independent predictors of survival.
Results: The median survival times after initiation of antiretroviral therapy for children who died and censored were 9 months and 72 months respectively. The overall mortality rate was 12.4 deaths per 1000 child-year. Advanced clinical stages, low hemoglobin level and CD4 cell count at initiation of the treatment were found to be significantly associated with risk of mortality among children on HAART.
Conclusion: The study demonstrated that survival rate among HAART children showed that country’s ART program has been successful in increasing longevity of life among HIV infected children. While advanced clinical stages, presence of anemia and low CD4 cell count demonstrated higher mortality among children. The study finding has important policy implications for HAART programme in the country. This claims for the need to identify and treat eligible HIV infected children at an early stage. Therefore, attention is needed to ensure an early diagnosis, enrollment into HAART and improving the quality of existing services like nutritional evaluation.
Keywords
HIV; Antiretroviral therapy; Children; Survival; Addis Ababa; Ethiopia
Introduction
Human immunodeficiency virus (HIV) is global public health challenge and main cause of morbidity and mortality sub- Saharan since the first case was reported in the early 1980’s. Recently, expanded access to Highly Active Antiretroviral Therapy (HAART) has led to dramatically reduction in HIV associated mortality and morbidity among children. HAART has radically reduced hospitalization rates and deaths associated with HIV infection. It results in noticeable survival benefits for HIV-infected children. Infants and young children have an exceptionally high risk of poor outcomes from HIV infection. Up to 52% and 75% of children die before the age of two and five years respectively in the absence of any intervention [1,2].
There are 25.8 million people living with HIV in Sub- Saharan Africa, comprising 70% of the global figure. New HIV infection declined by 41% since 2000, sub-Saharan Africa accounts for 66% of the global total of new HIV infections. In 2014, there were 190,000 new HIV infections among children, accounting for 86% of the newly infected children globally. As of December 2015, 49% of all children living with HIV were accessing HAART [3,4].
The survival benefit of HAART in reducing HIV related deaths has been well noticed in resource-rich settings. A recent systematic review reported 10-fold higher in mortality rate after HAART initiation among children in developing countries compared with developed. Identification of baseline factors that envisage mortality could permit for their possible amendment in order to improve pediatric HIV care and decrease HIV-related mortality among children on HAART. Studies showed that age, gender, World Health Organization (WHO) clinical staging, viral load, CD4 cell count, total lymphocyte count (TLC), body mass index (BMI), adherence and baseline hemoglobin level were the independent predictors of survival [5-8].
The HIV/AIDS epidemic has remained one of the important public health challenges in Ethiopia since it was first recognized in 1984. Currently, Ethiopia has a low rate of HIV endemicity, with a national prevalence of 1.1% [9]. Currently, 753,100 Ethiopians are living with HIV [10].
Since the introduction of free HAART services in the country in 2005 with a primary aim to reduce mortality among HIV infected patients, there have been improvements in service delivery and utilization via designing varies national guideline and policies. From an estimated of 765,500 people currently living with HIV, 542600 require HAART and 367,000 patients including 23,400 children under age of 15 are currently taking HAART. There are 1045 health facilities providing HIV care and treatment service [10].
Recognizing the need of HAART the government of Ethiopia issued the first HAART guideline in 2003, which was subsequently revised in 2005, 2008 and 2014 to facilitate rapid scale up of the service. According to the 2014 national guideline, which was mainly based on 2013 WHO guideline [11], HAART is recommended for all children <15 years with HIV infection regardless of CD4 count and WHO clinical stage. The HAART regimens for children were classified into three based on child age; <3 years, from 3 years to less than 10 and adolescent 10 to 19 years [10].
The mortality rates among children receiving HAART in Ethiopia ranges from 16.85 deaths per 1000 child-years to 40.0 deaths per 1000 child-years of follow-up period [12-16]. Predictors of mortality among children on HAART in the country were inconsistently reported by fragmented studies conducted in hospitals. WHO clinical stages were found statistically significant predictors of child survival in studies conducted at Adama [12], Bahirdar [13] and Mekele [16] hospitals, yet not statistically significant in studies conducted at Zewuditu [12] and Bahirdar [15] hospitals. Likewise, age was found as statistically significant predictor of survival in the study conducted in Bahirdar [13] and Mekele [16] hospitals, while it was not in the other studies [12,14,15]. CD4 cell count was found statistically significant predictor of child survival in four studies while not in the study conducted at Bahirdar [13].
Resolving these inconsistencies of the predictors of survival among children after initiation of HAART is important in designing a national and regional strategy to ultimately improve the effectiveness of HAART on child survival. Therefore, this study was conducted to confirm the predictors of the survival among children after initiation of ART and also resolve the inconsistency via multihospital based and longer period of follow up.
Methods
Study setting and period
The study was conducted in public hospitals in Addis Ababa, the capital city of Ethiopia. There are 10 public hospitals in the city. From 10 public hospitals in Addis Ababa, pediatric ART clinic existed only in 6 hospitals during January 1, 2008 to December 31, 2010. Out of these 6 hospitals, 4 were randomly selected and included, namely St. Paul referral Hospital and Millennium Medical College, St. Peter Referral Hospital, Zewuditu Memorial Hospital and Yekatit 12 Hospital. Medical records of HIV positive children who were enrolled on HAART in these hospitals are kept on-site in an electronic database.
Study design and participants
A retrospective cohort study was conducted among HIV positive children, under fifteen years old, who started HAART from 1st January 2008 to 31st December 2010. Each child was followed from the time of HAART initiation till death, loss to follow up or the end of study period, 31st December 2015. A child who was transferred out to other facilities during the study period or whose intake form was not complete was excluded from this study. All eligible children (n=757) during the specified time period were included to the study.
Measurements
The outcome variable was survival time or time to death of HIV-infected children after starting HAART. The survival time was measured using months from the time of HAART initiation to until the time of death, lost to follow up and the end of the study period. Survival times of children who were still alive as of 31st December 2015 or lost to follow up were considered censored times.
Independent variables were socio-economic and demographic characteristics (age, sex, educational status and employment status of the primary care giver of the child); baseline and follow-up clinical and immunologic information like presence of TB, TB treatment, opportunistic illness (OI), type of OI, developmental milestones, WHO clinical stages, prophylaxis use, nutritional status, immunization status, CD4 count, ART adherence, viral load and hemoglobin level.
Baseline CD4 count was categorized as per WHO age appropriate classification to describe their immunologic level. Children under age 1 and had CD4 cell count <1500 cells/mm3; children aged 1 to below 3 years old and had CD4 cell count <750 cells/mm3; children aged 3 to below 5 and had CD4 cell count <350 cells/mm3; and children aged 5 to below 15 and had CD4 cell count <200 cells/mm3 were categorized as having CD4 cell count below threshold [17]. Hemoglobin level was retrieved from record to assess anemia; for age 6-59 months, <11 g/dl; 5 to 12 years, <11.5 g/dl; and 12 to 15 years, <12 g/dl were categorized as anemic otherwise not anemic [18]. Adherence to HAART was measured by the last adherence level recorded on the follow-up form and classified as good >95%, fair 85-94%, poor <85% based on percentage of drug dosage calculated from the total monthly doses of HAART drugs.
Developmental milestone status was obtained from intake form and classified as: delayed, if a child fails to attain milestones for age; regressed, if a child loses what has been attained for age, otherwise normal.
Nutritional status was categorized as under nutrition for under five children if weight for age of a child is below (-2SD) standard deviation score, the standard WHO weight for age z-scores. Body mass index (BMI) was calculated for 5 years old or above children and BMI less than 16 kg/m2 were categorized as under nutrition [19].
Data source
A secondary data from follow up medical records of children who were enrolled on HAART from January 1, 2008 to December 31, 2010 were obtained from the hospitals’ database, extracting to excel files. A total of 945 children started HAART in the 4 hospitals during this period. Incomplete data in the excel file were extracted from patients intake forms and follow up forms manually after identifying the records using their HAART identification number.
Statistical analysis
The data was exported to SPSS version 20 for data analysis. The children’s characteristics were described in terms of median value for continuous data and percentage for categorical data. Finally, Kaplan-Meier method was used to estimate survival probability after initiation of ART, and log rank test was used to compare survival curves of children between different categories of predictor variables at p-value (<0.05). Cox proportional-hazard regression was used to determine determinants of survival after ART initiation. In the bivariate model, covariates with p-values (<0.2) were selected to be included in the multivariate model. Interaction effects were assessed for the covariates included in the multivariate model along with their main effects. Backward selection was used to retain the significant variables in the final model. The final model was interpreted using adjusted Hazard Ratio (AHR) with 95% confidence interval and p-value<0.05 to measure the risk of death between different levels of independent variables. Model adequacy was assessed for the variables finally remained in the multivariate model using schoenfeld residuals plots and tests of proportional hazard by considering an alternative model with time dependent coefficients of the covariates.
Ethical consideration
Ethical clearance was obtained from Institutional review board (IRB) of Collage of Public Health and Medical sciences, Jima University. In addition, permission was sought from the hospitals Administrative. The retrieved data were kept strictly confidential and names of children or their parents were not included.
Results
Baseline characteristics
A total of 757 children were included in the analysis, of which, 371 (49.0%) were female. More than seven in ten of the participants were in the age category of 5-14 years. Children with CD4 counts below threshold level at baseline were 240 (32.2%). Three fifth of the participants were undernourished at baseline, 443 (58.8%).
Survival status of children
From the total children who initiated HAART during the study period, 51 (6.7%) of them died, 614 (81.1%) were alive and 92 (12.2%) were lost to follow up. The median follow up time was 68 months with intra quartile range (IQR) of 62.0 to 83.0 months. The cumulative survival probabilities of children after 3, 6, 12, 24, 36, 48 and 72 months were 0.975, 0.971, 0.958, 0.949, 0.94, 0.935 and 0.931, respectively.
Of the 51 deaths, 17 (33.3%) of them occurred within the first three months after initiation of HAART. The total cohort contributed 4112 child-years, resulting in the overall mortality rate of 12.4 deaths per 1000 child-years. The median survival time after initiation of HAART for children who died and censored were 9 months, 72 months, respectively.
Comparison of survival functions
Using Kaplan-Meir survival curve, survival experience of the children was assessed against different categories of the predictor variables. Among all the independent variables, only survival experience of different categories of Hemoglobin level, CD4 count and WHO clinical stages were found significant. Children with normal hemoglobin level have longer survival experience than those children with low hemoglobin level. This visually observed difference was also statistically significant (Log rank, p-value<0.001). Children with WHO clinical stages I & II survived significantly longer than those with stages III & IV and also children on WHO clinical stage III had longer survival than those on clinical stage IV after HAART initiation (Log rank, p-value<0.001). The observed difference of shorter survival of children with CD4 cell count below threshold compared with those children above the threshold was found to be statistically significant (Log rank, p-value<0.001) (Figure 1).
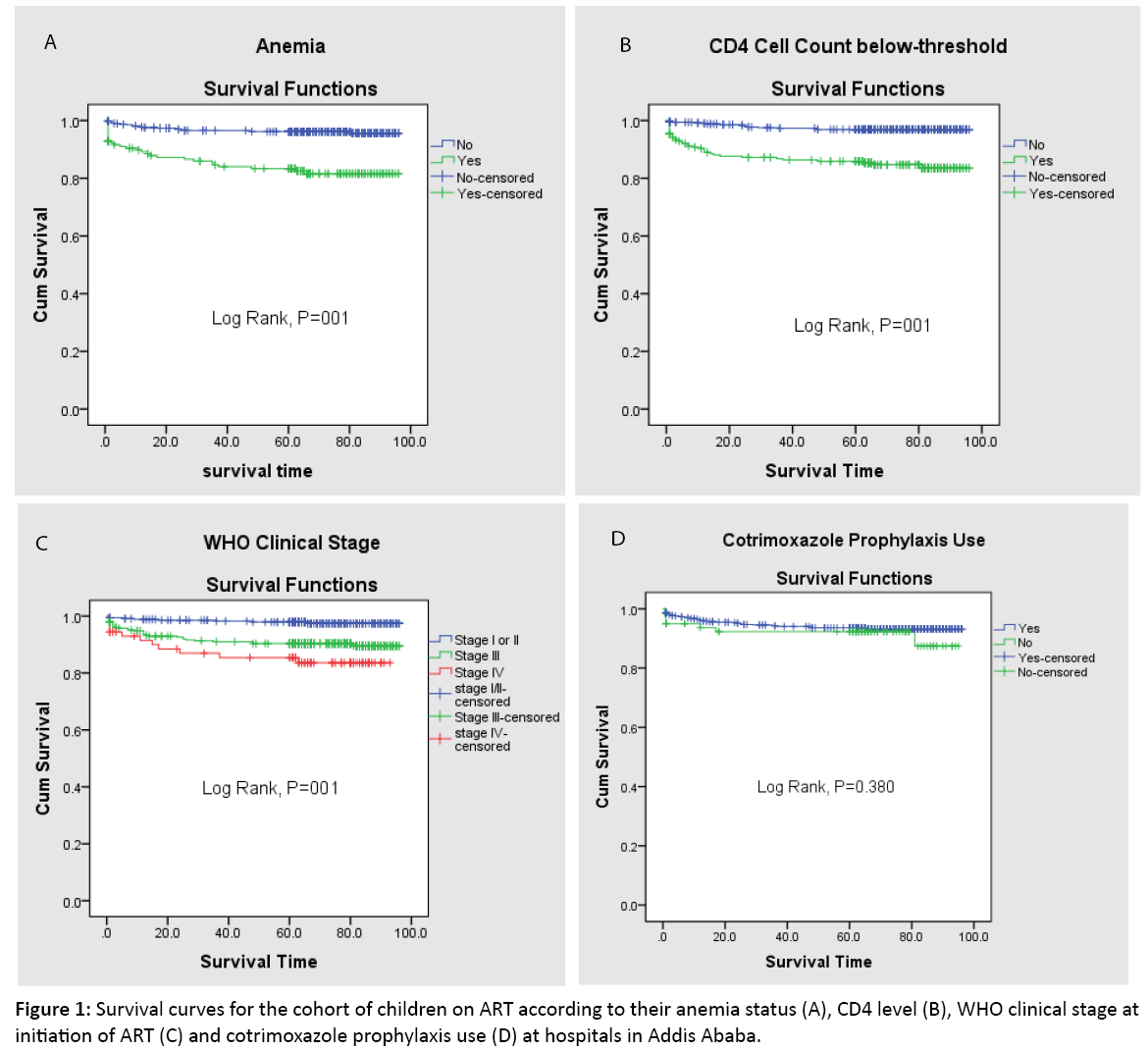
Figure 1: Survival curves for the cohort of children on ART according to their anemia status (A), CD4 level (B), WHO clinical stage at initiation of ART (C) and cotrimoxazole prophylaxis use (D) at hospitals in Addis Ababa.
Determinants of child survival
In order to select the potential covariates for multivariable Cox regression model, variables associated at p-value<0.25 at bivariate regression were considered. Based on this criterion four variables namely WHO Clinical stage, presence of anemia, CD4 cell count below threshold and undernutrition were the candidates for the multivariable Cox regression analysis.
Interaction effects were checked along with the main effects and none of the interaction effects were found significant at p-value<0.05. WHO clinical stages III & IV, presence of anemia, and CD4 cell count below threshold level were identified as independent determinants for child survival in the final model. The results were interpreted as, at any given time during the study period, the hazard of death in children with WHO clinical stages III and IV were respectively 3 (AHR=3.4; 95% CI: 1.5, 7.4) and 5 (AHR=4.8; 95% CI: 1.9, 12.2) times higher than those children who had WHO clinical stages I and II at baseline. Similarly, the risk of death in children with low hemoglobin level (AHR=3.3; 95% CI: 1.9, 5.9) was 3 times higher than those who had normal hemoglobin level. Children who had CD4 cell count below threshold level (AHR= 3.4; 95% CI: 1.8, 6.5) were 3 times more likely to die when compared with those children above the threshold at baseline (Table 1). The schoenfeld residual analysis and tests of proportional hazard results showed the fulfillment of proportional hazard assumption for the covariates found significant predictors of child survival in the final model.
| Variables | Category | No (%) | P-value | CHR (95% CI) | AHR (95% CI) |
|---|---|---|---|---|---|
| Sex | Female | 371 (49.0) | 1.0 | ||
| Male | 386 (51.0) | 0.603 | 1.2 (0.7, 2.0) | ||
| Age category(years) | <1 | 8 (1.1) | 0.974 | 0.0 (0.0, 3.03 ) | |
| 1-5 | 212 (28.0) | 0.898 | 1.0 (0.5, 1.8) | ||
| ≥ 5 | 537 (70.9) | 1.0 | |||
| Cotrimoxazole use | Yes | 677 (89.4) | 1.0 | ||
| No | 80 (10.6) | 0.383 | 1.4 (0.6, 3.2) | ||
| Developmental milestone | Normal | 574 (83.3) | 1.0 | ||
| Delayed | 101 (14.7) | 0.313 | 1.5 (0.7, 3.0) | ||
| Regressed | 14 (2.0) | 0.865 | 1.2 (0.2, 8.7) | ||
| Immunization Status | Appropriate for age | 642 (92.5) | 1.0 | ||
| Not appropriate | 37 (5.3) | 0.318 | 1.7 (0.6, 4.7) | ||
| Not immunized at all | 15 (2.2) | 0.936 | 1.1 (0.1, 7.9) | ||
| Tuberculosis | No | 600 (79.4) | 1.0 | ||
| Yes | 156 (20.6) | 0.590 | 1.2 (0.6, 2.3) | ||
| ART Adherence status | Good (≥ 95%) | 690 (96.5) | 1.0 | ||
| Fair (85-94%) | 16 (2.2) | 0.970 | 0.0 (0.0, 2.2) | ||
| Poor (≤ 84) | 9 (1.3) | 0.371 | 2.5 (0.3, 18.2) | ||
| WHO Clinical Stage | Stages I & II | 346 (46.1) | 1.0 | 1.0 | |
| Stage III | 332 (44.3) | <0.01 | 4.3 (2.0, 9.4) | 3.4 (1.5, 7.4) | |
| Stage IV | 72 (9.6) | <0.001 | 7.2 (2.9, 18.0) | 4.8 (1.9, 12.2) | |
| Low hemoglobin level | No | 535 (76.1) | 1.0 | 1.0 | |
| Yes | 168 (23.9) | <0.001 | 4.9 (2.8, 8.5) | 3.3 (1.9, 5.9) | |
| Low CD4 count | No | 505 (67.8) | 1.0 | 1.0 | |
| Yes | 240 (32.2) | <0.001 | 5.4 (3.0, 9.9) | 3.4 (1.8, 6.5) | |
| Nutritional status | Normal | 310 (41.2) | 1.0 | ||
| Under Nutrition | 443 (58.8) | 0.140 | 1.6 (0.9, 2.8) | ||
| OI presence | No | 431 (57.0) | 1 | ||
| Yes | 325 (43.0) | 0.504 | 1.2 (0.7, 2.1) |
1. Developmental milestone status is delayed, if a child fails to attain milestones for age; regressed, if a child loses what has been attained for age, otherwise normal.
2. Low Hemoglobin level is <11 g/dl for age 6-59 months; <11.5 g/dl for 5 to 12 years; and <12 g/dl for 12 to 15 years for 3 to below 5; and <200 cells/mm3 for children aged 5 to below 15.
3. Low CD4 Count is <1500 cells/mm3 for under 1year age; <750 cells/mm3 for 1 to less than 3 years old; <350 cells/mm3 for 3 to below 5; and <200 cells/mm3 for children aged 5 to below 15.
4. Nutritional status is under nutrition if weight for age of a child is below (-2SD) standard deviation Z-score, the standard WHO weight for age z scores for under 5 children and body mass index less than 16 kg/m2 for above 5 years old children.
Table 1: Bivariable and multivariable Cox regression analysis for socio-demographic, clinical and immunologic determinants of survival among children started ART at public Hospitals between, January 1, 2008 and December 31, 2010 in Addis Ababa, Ethiopia (n=757).
Discussion
The mortality rate in this study was 12.4 deaths per 1000 child-years and the predictors of mortality were clinical stages, hemoglobin level and CD4 cell count. Compared to the rates of 4.0 and 16.85 per 100 child-years reported in Ethiopia [15-16], the mortality rate observed in this study was low. Similarly, it is lower than the rates reported by the study done in South Africa and Kenya [20,21]. The reasons for lower mortality rate in this study could be related to difference study setting and period. Comparing with studies conducted in Ethiopia, the low mortality rate observed in this study could be for the reason that as this study was conducted in Addis Ababa hospitals, there might be better quality of HAART services and improved awareness of patients’ family to comply with advises of the counselors. It is the result expected at this time as the government of Ethiopia has been making a great effort in reducing the prevalence of death due to AIDS. The low mortality observed could also be because lack or incompleteness of data on some patients which resulted in under estimation of number of death.
About one-third of deaths occurred within the first 3 months of HAART initiation. Studies done in Ethiopia and abroad consistently reported that majority of death occurs during the first few months of HAART initiation. Study conducted in South Africa reported 64% of death occurred within three months of HAART initiation [20]. Similarly in Nigeria, 81.3% and 84.4% of deaths occurred in the first 6 months and with 1 year respectively [22]. The study conducted in Adama [12] and Zewuditu Memorial [14] hospitals reported majority of death within the first sixth months of starting HAART. The study conducted in Mekele showed high mortality in the first 12 months after HAART initiation [16].
Children who initiated HAART at WHO clinical stage (III/IV) were associated with the reduced survival. The result of this study is in line with other studies conducted in Adama, Felegehiwot and Mekele hospitals [12,13,16]. Children with low hemoglobin level have higher risk of death than those with normal hemoglobin level. This finding is consistent with the findings of studies conducted in hospitals of Ethiopia and Kenya [12-14,16,21].
CD4 cell count below threshold was the determinant factor found significant for child mortality in this study. It is in line with findings of studies conducted in Adama, Felegehiwot, Zewuditu and Mekele hospitals [12-14,16]. Cotrimoxazole prophylaxis utilization was one of the variables this study was aimed at to assess its effect on child survival after HAART initiation and yet it was found insignificant.
Finally, this study is limited in that it does not take in to account some predictors of child survival including viral load, type of opportunistic infections, care givers educational and employment status, as the presence of these variables could have given a good insight about the determinants of child survival after initiation of HAART.
Conclusion
In general, survival after HAART initiation among children infected with HIV was high. The estimate of the survival rate among HAART Children suggests that country’s ART program has been successful in increasing longevity of life of HIV infected children. The study also demonstrated higher mortality among children with advanced clinical stages, presence of anemia, and having lower CD4 cell count. The study findings have important policy implications for ART programme in the country. This claims for the need to identify and treat eligible HIV infected children at an early stage. Therefore, attention is needed to ensure early diagnosis and enrollment into ART. This mortality and its determinants need to be addressed by assessing patients for those risk factors and improving the quality of existing services like nutritional evaluation. Health worker need to give more emphasis during clinical care for patients like advanced WHO HIV disease stage and those who has low CD4 count.