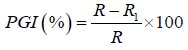Keywords
Moringa oleifera; Vernonia amygdalina; Aspergillus flavus; Botryodiplodia theobromae; Vigna unguiculata; Pathogenicity; Inhibition; Antifungal; Efficacy; Seeds
Introduction
Cowpea (Vigna unguicualata L. Walp) belongs to the family Leguminosae and to the genus Vigna. The crop originated from Africa and is commonly cultivated in the semi-arid and forest margin tropics and sub-tropics where it is well adapted and probably the most popular grain legume crop in Nigeria. Cowpeas are the second most important food grain legume crop in tropical Africa, the most important being Phaseolus vulgaris, the common bean. Nigeria, Niger, Burkina Faso, Uganda and Senegal grow cowpea for market, but they are widely grown as a subsistence crop for home use in nearly all African countries south of the Sahara. Cowpea is an important food for human beings due to their high amounts of the essential amino acid contained in its protein such as lysine and tryptophan. They also provide feed, forage, hay and silage for livestock and green manure and cover crop for maintaining the productivity of soils. Cowpeas are susceptible to serious damage by insect pest during storage. They are generally not as susceptible to epidemics of diseases as many other grain legumes. The most important diseases of cowpea are: brown blotch caused by Colletotrichum capsici, Cowpea wilt caused by Fusarium oxysporum, other pathogens of cowpea include; Aspergillus flavus, Aspergillus niger, Brotryodiplodia theobromae, Rhizopus spp., scab (sphuceloma spp.) and septotial leaf spot (Septoria spp.) [1]. The control of seed-borne pathogens through the application of chemicals for seed dressing has been effective in reducing seed-borne pathogens and even improved the germination ability of seeds [2]. However, there is fear about the safety of chemical residues and the possibility of human toxicity and environmental pollution [3]. Chemical pesticides are also non-biodegradable and extremely toxic [4,5]. On the other hand, pesticides of plant origin are specific biodegradable, cheap, readily available and environmentally safe than synthetic chemicals. Hence, plant extracts could be an alternative to toxic fungicides for controlling plant pathogens [6-8]. The research focused on the management of cowpea seed pathogens isolated from different locations in Dutsin-Ma local government area of Katsina State, Nigeria, using Moringa oleifera and Vernonia amygdalina leaf extracts.
Materials and Methods
The study area
The study area lies on latitude 12o 27’ 18’’ N and longitude 07o 29’ 29’’ E. The experiment was conducted at the Biological Science Laboratory, Federal University Dutsin-Ma, Katsina state, Nigeria.
Collection of rotted and healthy cowpea seeds
Cowpea (Vigna unguiculata) seeds showing various degrees of rot symptoms of fungi organisms were collected from three locations of Darawa, Dutsin-Ma and Makera. The fungi organisms in the cowpea seeds were isolated and subsequently identified in the Biological Science Laboratory, Federal University Dutsin-Ma, Katsina state, Nigeria. The healthy cowpea seeds were used for pathogenicity test of the isolated fungi organisms.
Preparation of potato dextrose agar (PDA)
Potato dextrose agar (PDA) was prepared by measuring 10 g PDA and dissolving into 250 ml of distilled water using measuring cylinder and was autoclaved at 121°C for 15 minutes, the media was allowed to cool at room temperature to 45°C. 0.16 g/L streptomycin sulphate powder was added to suppress bacterial contaminations [9,10]. 15 ml of the molten PDA was poured into sterile glass petri dishes of 9 mm diameter and were allowed to cool at room temperature before inoculation of the test fungi.
Isolation of test fungi organisms
Rotted and diseased cowpea seeds were collected and firstly sterilized by dipping completely in a concentration of 5% sodium hypochlorite solution for 1 min. The seeds were then removed and rinsed in four successive changes of sterile distilled water. 0.16 g of streptomycin was added to suppress the growth of bacteria. The medium was thereafter poured in 9 cm sterile glass petri dishes before inoculation of the rotted cowpea seeds.
Inoculation: The infected cowpea seeds were transferred onto solidified potato dextrose agar (PDA) medium in petri dishes. Up to four seeds of cowpea seed were placed on each PDA plates and incubated at ambient room temperature for 7 days. The plates were examined daily for the development of fungal growth.
Identification of fungal growth: The mycelial growths of the fungal organisms that grew on PDA were used to sub-culture in order to obtain pure cultures. Sterilized inoculation needle was used to transfer the mycelial growth into sterile PDA plates. The inoculated plates were then incubated at ambient room temperature (30 ± 5°C) for 5 days. The pure isolates were kept in slants and stored for characterization and pathogenicity test. Macroscopic examination as well as microscopic and morphological characteristics and identification of fungi organisms were made and compared with other authorities [11].
Pathogenicity test
Healthy cowpea seeds were collected and thoroughly washed in 5% sodium hypochlorite solution and rinsed in sterile distilled water for three consecutive times [12]. About 20 ml of distilled water was mixed separately with 5 mm disc from 5 days old culture of A. niger, B. theobromae and A. flavus that were isolated from rotted cowpea seeds. The solutions made of the fungi organisms were spread on filter paper contained in petri dishes and the healthy cowpea seeds were inoculated on the fungi blot paper in the petri dishes. The same procedure was used for the control except that the blot paper was spread with distilled water instead of discs of fungi organisms and cowpea seeds were inoculated accordingly. The plates were incubated for 5 days to give enough time for maturity and growth of the fungus. When growth was fully established, the growth characteristics were compared with the original rotted cowpea seeds and examined for infection and disease development.
Preparation of plant extracts
Plants extracts were prepared according to the method of Gwa and Akombo with little modifications [13]. Extracts were obtained by addition of powder of 5 g, 10 g, and 15 g of each plant extract to 250 ml of sterile distilled water separately in 1000 ml Pyrex flask. These were left for 24 hours and subsequently filtered through four fold of sterile filter cloth. The filtrates obtained were used as the plant extracts in the experiment. About 5 ml of the extracts of plant at each level of concentration was mixed in sterile petri dishes containing 15 ml of PDA solution and allowed to solidify before inoculation of the pathogens.
Measurement of mycelial radial growth: The efficacies of the aqueous plant extracts and chemical fungicide were tested in vitro for their fungicidal activity against the cowpea (V. unguiculata) fungi pathogens caused by A. flavus, B. theobromae and A. niger. Three plates were treated with extract of each plant at each concentration. The control experiments had 5 ml of distilled water added to PDA in place of plant extracts respectively. The treatments and control were incubated for four days at ambient room temperature and measurement of growth as radius of a growing fungal colony were undertaken at intervals of one day for four days using a transparent ruler. The absence of growth in any of the plates was indication of the potency of the extract and the chemical fungicide against the test fungi. Fungitoxicity was determined as percent growth inhibition (PGI) according to the method described by Korsten and De Jager [14].
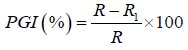
Where,
PGI=Percent Growth Inhibition
R=The distance (measured in mm) from the point of inoculation to the colony margin in control plate,
R1=The distance of fungal growth from the point of inoculation to the colony margin in treated plate.
Experimental design and data analysis
Data collected were subjected to Analysis of Variance (ANOVA) using GenStat Discovery Edition 12 for ANOVA and means separation, Minitab Release 17 for descriptive statistics and Graph Pad Prism 6 for trend graphs. Statistical F-tests were evaluated at P ≤ 0.05. Differences among treatment means for each measured parameter were separated using Fisher’s Least Significant Difference (FLSD).
Results
Isolation and identification of fungal organisms
The fungal organisms isolated and identified from the rotted cowpea seeds were Aspergillus niger, Aspergillus flavus, Botryodiplodia theobromae, Fusarium oxysporum and Colletotrichum spp. Results presented in Figure 1 show the culture characteristics of some of the isolated and identified fungi organisms.
Figure 1: Culture of (A) Aspergillus niger (B) Aspergillus flavus (C) Botryodiplodia theobromea and (D) Fusarium oxysporum.
Frequency of occurrence of fungal pathogens isolated from cowpea seeds at different locations
Results of the isolation of fungi organisms in Table 1 show the mean frequency of the fungi organisms in different locations. F. oxysporum occurred most in Darawa and Makera (1.66) but least in Dutsin-Ma (1.00). B. theobromae however showed more occurrences in Dutsin-Ma (3.66) compared with Darawa and Makera. The occurrence of A. flavus was highest in Darawa and Dutsin-Ma (3.00) and least in Makera (2.66). The frequency of occurrence of A. niger was high in all the locations but highest in Makera (3.67) and lowest in Darawa (3.00) and Dutsin-Ma (3.00) respectively. Colletotricum spp was only isolated in Dutsin-Ma (0.33) location. There were no significant differences (P ≤ 0.05) between locations for each of the fungal organisms isolated.
| Fungi |
Locations |
|
| Darawa |
Dutsin-Ma |
Makera |
P-Value |
| F. oxsporum |
1.66 ± 0.58 |
1.00 ± 0.57 |
1.66 ± 0.33 |
0.70ns |
| B. theobromae |
2.00 ± 0.00 |
3.66 ± 0.33 |
1.67 ± 1.20 |
0.19ns |
| A. flavus |
3.00 ± 0.57 |
3.00 ± 1.53 |
2.66 ± 0.66 |
0.96ns |
| C. gloesporiodes |
0.00 ± 0.00 |
0.33 ± 0.33 |
0.00 ± 0.00 |
0.42ns |
| A. niger |
3.00 ± 1.00 |
3.00 ± 1.00 |
3.67 ± 1.20 |
0.88ns |
| Colletotrichum sp. |
0.00 ± 0.00 |
0.33 ± 0.33 |
0.00 ± 0.00 |
0.42ns |
Means on the same row with ns are not statistically significant (P ≤ 0.05); ns=not significant
Table 1: Mean frequency of occurrence of fungal pathogens from cowpea seeds at different locations.
Number of fungal pathogens occurring at different locations
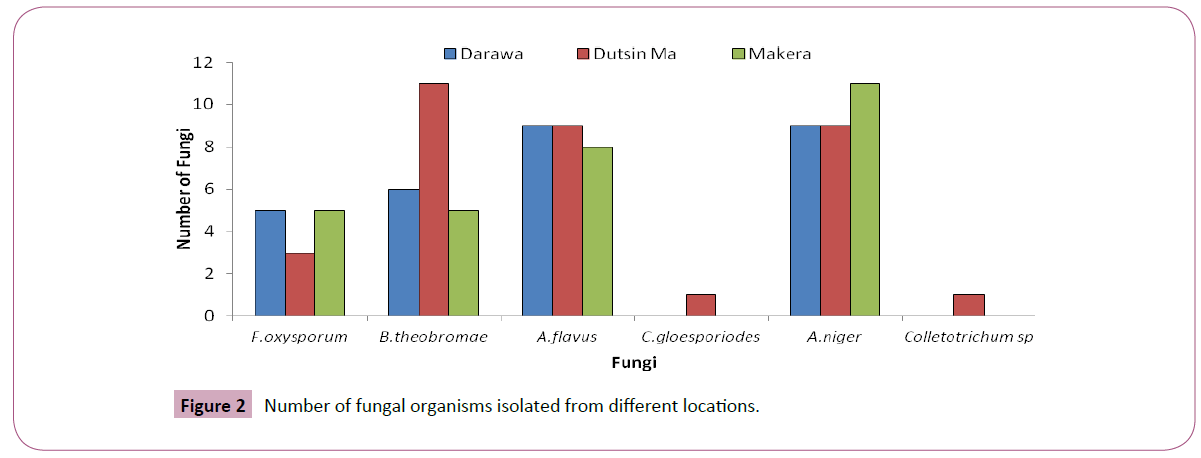
Result presented in Figure 2 shows the different number of fungal organisms isolated from different locations. The result showed that the sum total of the fungi organisms isolated in three different locations were 13 (F. oxysporum), 22 (B. theobromae), 26 (A. flavus), 29 (A. niger) and 1 (Colletotricum sp).
Figure 2: Number of fungal organisms isolated from different locations.
Pathogenicity test
Pathogenicity test revealed that healthy cowpea seeds showed symptoms of rot after 5 days of inoculation with fungi mycelial. Morphological and microscopic characteristics were compared with initial cultures after re-isolation and were found to be same. The control (healthy cowpea seeds inoculated without fungi mycelial) showed no symptoms of rot.
Effect of Moringa oleifera and Vernonia amygdalina extracts on mycelial growth inhibition of A. flavus and B. theobromae
Results obtained in Table 2 shows that the different concentration of M. oleifera and V. amygdalina show variation in effect on the fungi organisms tested. Moringa showed mycelial inhibition of 3.75 cm at 15 g/250 ml better than at 10 g/250 ml and 5 g/250 ml and there was no significant difference (P ≤ 0.05) between the concentrations tested. However, there were significant differences among concentrations when bitter leaf extract was used for the control of A. flavus in vitro. The result also showed that there was a significant difference (P ≤ 0.05) in activities of M. oleifera and V. amygdalina extracts in controlling B. theobromae at different concentrations with the highest level of inhibition recorded at 15 g/250 ml, followed by 10 g/250 ml and least in 5 g/250 ml respectively.
| Pathogen/Plant extract |
Treatments and mycelial growth inhibition |
P-Value |
| 5 g/250 ml |
10 g/250 ml |
15 g/250 ml |
Control |
|
| A. flavus |
|
|
|
|
|
| M. oleifera |
5.37 ± 0.45 |
4.66 ± 0.47 |
3.75 ± 0.35 |
5.41 ± 0.95 |
0.18ns |
| V. amygdalina |
4.41 ± 0.36b |
3.41 ± 0.30bc |
2.70 ± 0.32c |
8.42 ± 1.00a |
0.00 |
| B. theobromae |
|
|
|
|
|
| M. oleifera |
4.12 ± 0.50b |
2.95 ± 0.37b |
2.04 ± 0.39b |
7.38 ± 1.77a |
0.00 |
| V. amygdalina |
5.66 ± 0.72b |
3.66 ± 0.52b |
2.33 ± 0.37b |
8.55 ± 2.47a |
0.00 |
Means on the same row with different superscript are statistically significant (P ≤ 0.05); ns=not significant
Table 2: In vitro mycelial growth inhibition of A. flavus and B. theobromae at different concentrations of plant extracts with control.
This work shows that the extract of V. amygdalina was more potent in growth inhibition of A. flavus compared with M. oleifera. On the other hand, the efficacy of Moringa oleifera was observed to inhibit the growth of B. theobromae more compared with V. amygdalina. Mean percentage growth inhibition of M. oleifera and V. amygdalina on B. theobromae at different concentrations showed a statistical significant (P ≤ 0.05). There was no significant difference (P ≤ 0.05) between concentrations and control (without Moringa extract) in controlling A. flavus when M. oleifera extract was used.
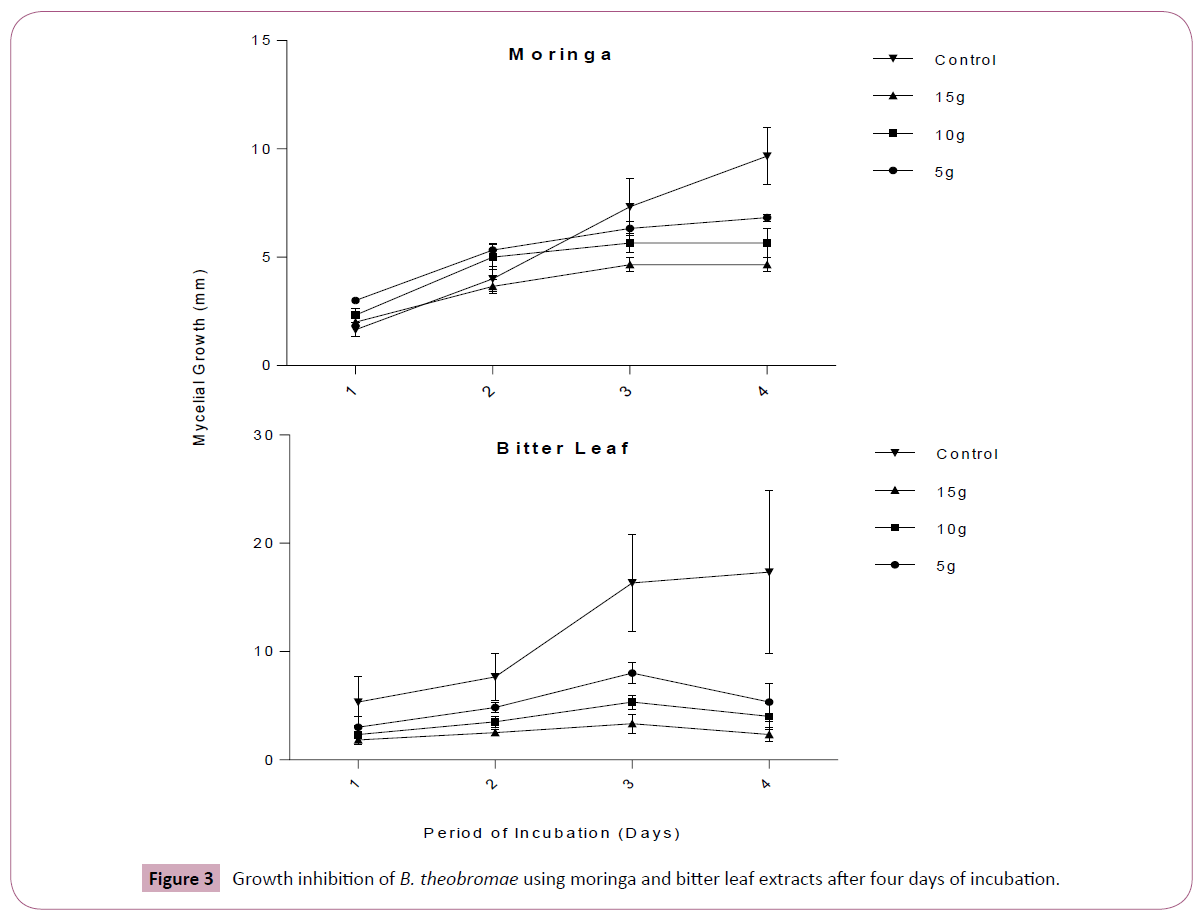
The results in Table 3 compare the inhibition of A. flavus and B. theobromae using crude extracts of M. oleifera, V. amygdalina at each concentration. The result revealed that there was no significant difference (P ≤ 0.05) between Moringa and bitter leaf extracts at 5 g/250 ml in controlling A. flavus. However, there were significant differences (P ≤ 0.05) in the activities of the extracts at 10 g/250 ml, 15 g/250 ml and control in inhibiting the mycelia growth of A. flavus. When M. oleifera and V. amygdalina were compared at each level of concentration as well as the control. There was no significant difference (P ≤ 0.05) in efficacy of the extracts in inhibiting the mycelia growth of B. theobromae. Figure 3 shows the inhibitory effect of plant extracts on mycelia growth of B. theobromae for the period of four days.
| Treatment |
Plant extract and growth inhibition |
Df |
T-Value |
P-value |
| M. oleifera |
V. amygdalina |
| A. flavus |
|
|
|
|
|
| 5 g |
5.37 ± 0.45 |
4.41 ± 0.36 |
21 |
1.63 |
0.11 |
| 10 g |
4.66 ± 0.47 |
3.41 ± 0.30 |
18 |
2.24 |
0.03* |
| 15 g |
3.75 ± 0.35 |
2.70 ± 0.32 |
21 |
2.19 |
0.04* |
| Control |
5.41 ± 0.96 |
8.42 ± 1.00 |
21 |
-2.16 |
0.04* |
| B. theobromae |
|
|
|
|
|
| 5 g |
4.12 ± 0.50 |
5.66 ± 0.73 |
19 |
-1.73 |
0.10 |
| 10 g |
2.96 ± 0.37 |
3.66 ± 0.52 |
19 |
-1.10 |
0.28 |
| 15 g |
2.04 ± 0.39 |
2.33 ± 0.37 |
21 |
-0.54 |
0.59 |
| Control |
7.38 ± 1.80 |
8.55 ± 2.50 |
19 |
-1.60 |
0.12 |
* indicates statistical significance at 0.05%
Table 3: In vitro comparative growth inhibition of A. flavus and B. theobromae using M. oleifera and V. amygdalina at different concentrations.
Figure 3: Growth inhibition of B. theobromae using moringa and bitter leaf extracts after four days of incubation.
Discussion
Fungal pathogens are commonly associated with rot of cowpea seeds similar to the work of Nyaka that isolated seven pathogenic fungi organisms responsible for cowpea seed rot disease. These pathogenic fungi included Colletotrichum sp., Fusarium sp., Pestalotia sp., Geotrichum sp., A. flavus, A. niger and B. theobromae. Magdalena et al. observed damages caused by Colletotrichum sp. on cowpea similar to the work of Akinbode et al. who earlier observed the growth of Colletotrichum sp. on cowpea seeds.
The result obtained showed that all the leaf extracts at their different level of concentrations tested were effective in inhibiting the growth of seed-borne fungi pathogens of cowpea, however, the rate of growth was influenced by the type of the extracts used and the concentration. This result is in agreement with Domenico that described the antifungal activity of some plant extracts on the development of Fusarium oxysporum f. sp. lycopersici and selected the best extracts to be tested as phytofungicide to control crop diseases, with the ultimate goal of developing a green alternative to synthetic fungicides. Using the conidia germination assay, of the 24 plant extracts tested, 15 reduced conidia germination and 6 completely inhibited conidia germination. Extracts of Rivina humulis, Brassica carinata, Brunfelsia calyicina, Salvia guaranitica and Punica granatum showed good antifungal activity.
Gwa and Akombo observed that Piper nigrum, Zingiber officinale, A. indica, C. papaya and N. tabacum have significant effect (P ≤ 0.05) on mycelial growth of A. flavus in vitro. The authors showed that period of incubation and concentration influenced the efficacy of the extracts on mycelial growth of A. flavus in vitro. Sangoyomi et al. demonstrated the fungitoxic effect of extracts obtained from Allium sativum, Ocimum gratissimum, Cassia alata, Azadiracta indica, and Hibiscus rosasinensis. They showed that the extracts were able to inhibit mycelial growth and reduce production of conidia in the four major fungi associated with yam rot during storage. M. oleifera and V. amygdalina extracts when used at low concentrations were found to be effective against the growth of fungi organisms. There was a general increase in percentage growth inhibition with an increase in extract concentration. This finding agree with the work of Ghangaonkar, who observed that extracts of Polyalthia longifolia, Annonas quamosa and Tridax procumbens were found to be inhibitory for the growth of Aspergillus flavus, Aspergillus niger, Fusarium oxysporum and the mycelial growth of Cladosporium allii on cowpea seed. Cook and Baker revealed that the antifungal activities of the V. amygdalina inhibited the growth of B. theobromae and A. flavus. Sobowale et al. showed that M. oleifera retarded the maximum growth of B. theobromae 41% followed by Aspergillus flavus 29%, Fusarium oxysporum 28% and less effective against A. niger 11%. Polyalthia longifolia extract inhibited the fungi, Penicillium digitatum 50% followed by Botrytis cinera 45% over control [15-25].
Conclusion
Aspergillus niger, Aspergillus flavus, Botryodiplodia theobromae, Fusarium oxysporum and Colletotrichum sp are responsible for rot of cowpea seeds in Dutsin-Ma local government area of Katsina State. This reduced the seed quality and seed health of cowpea resulting in poor viability of seeds annually. Efforts should be made to increase the seed quality by using extracts of plant origin to control fungal pathogens of cowpea. The crude extracts of M. oleifera and V. amygdalina have the potency of controlling fungal pathogens of cowpea. Extract of V. amygdalina is the most effective extract in controlling A. flavus at all level of concentrations compared with M. oleifera. Both extracts at all level showed high efficiency in controlling B. theobromae in vitro. It is therefore concluded that both extracts could be formulated at the respective levels and used in the management of fungi pathogens of cowpea seeds.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Funding Acknowledgement
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
References
- Gali K, Ramakrishnan G, Kothai R, Jaykar B (2011) In vitro anti-cancer activity of methanolic extract of leaves of Argemone mexicana Linn. International Journal of Pharmaceutical Technology Research 3: 1329-1333.
- Okoro CK (2005) Antibacterial and antifungal activity of pomegranate on cowpea. African Journal of Environmental Agriculture and Food Chemistry 10: 1958-1969.
- Okigbo RN (2005) Biological control of post-harvest fungal rot of yam (Dioscorea spp.) with Bacillus subtilis. Mycopathologia 159: 307-314.
- Lakshmeesha TR, Sateesh MK, Vedashree S, Sofi MS, Umesha S (2013) Efficacy of botanicals on soybean seed-borne Fusarium equiseti. VCFL Sciences 3: 10-16.
- Gwa VI, Nwankiti AO (2017) Efficacy of some plant extracts in vitro control of collectotrichum species, causal agent of yam (Doscorea rotundata Poir) tuber rot. Asia Journal of Plant Science and Research 7: 8-16.
- Singh ND, Sharma AK, Dwivedi P, Kumar M, Patil RD, et al. (2011) Immunosuppressive effect of combined citrinin and endosulfan toxicity in pregnant wistar rats. Vet Arhiv 81: 751-763.
- Bhagwat MK, Datar AG (2014) Antifungal activity of herbal extracts against plant pathogenic fungi. Arch Phytopathology Plant Protect 47: 959-965.
- Gwa VI, Ekefan EJ, Nwankiti AO (2017) Antifungal potency of some plant extracts in the control of white yam (Dioscorea rotundata poir) tuber rot. Adv Biotech & Micro 7: 555703.
- Brooks GF, Carroll KC, Butel JS, Morse SA (2004) Jawetz, Melnick & Adelberg’s Medical Microbiology. 23rd edn. McGraw Hill Companies Inc., Singapore.
- Burgess LW, Knight TE, Tesoriero L, Phan HT (2008) Diagnostic Manual for Plant Diseases in Vietnam. ACIAR Monograph No. 129, Australian Centre for International Agricultural Research, Canberra.
- Navi SS, Bandyopadhyay R, Hall AJ, Bramel-Cox PJ (1999) A Pictorial Guide for the Identification of Mold Fungi on Sorghum Grain. Information Bulletin no. 59, International Crops Research Institute for the Semi-Arid Tropics, Andhra Pradesh, India.
- Ritchie B (1991) Practical Techniques in Plant Pathology CAB. Wallingford, UK.
- Gwa VI, Akombo RA (2016) Studies on the antimicrobial potency of five crude plant extracts and chemical fungicide in the in vitro of Aspergillus flavus causal agent of white yam (Dioscorea rotundata) tuber rot. Journal of Plant Science and Agriculture Research 1: 1-8.
- Korsten L, De Jager EE (1995) Mode of action of Bacillus subtilis for control of avocado post-harvest pathogens. South African Avocado Growers Association Yearbook 18: 124-130.
- Siva N, Ganesan S, Banumathy N, Muthuchelian (2008) Antifungal effect of leaf extract of some medicinal plant against Fusarium oxysporum causing wilt disease of Solanum melogena L. Ethnobotanical Leaflets 12: 156-163.
- Nongalleima K, Dey A, Deb L, Singh CB, Thongam B, et al. (2012) Endophytic fungus isolated from Zingiberzer umbet (L.) sympton inhibits free radicals and cyclo-oxygenase activity. Int J Pharm Tech Res 5: 301-307.
- Anis Z, Sulaiman O, Hashim R, Mehdi SH, Ghalib RM (2012) Radical scavenging activity, total phenol content and antifungal activity of Cinnamomum iners Wood. Iranica J Energy & Environ 3: 74-78.
- Sangoyomi JY, Otoide JE, Ijadunola JA, Aladejimokun AO (2010) Efficacy of antimicrobial effect of Venonia amygdalina and Tridax procumbens in in vitro control of cowpea (Vigna unguiculata) post-harvest fruit rot. Report and Opinion 3: 120-123.
- Ghangaonkar NM (2007) Efficacy of plant extracts on the post-harvest fungal pathogens of onion bulbs. Bioinfolet 4: 291-294.
- El-Khateeb AY, Elsherbiny EA, Tadros LK, Ali SM, Hamed HB (2013) Phytochemical analysis and antifungal activity of fruit leaves extracts on the mycelial growth of fungal plant pathogens. Journal Plant Pathology & Microbiology 4: 199.
- Rhouma A, Ben Daoud H, Ghanmi S, Ben Salah H, Romdhane M,et al. (2009). Antimicrobial activities of leaf extracts of Pistacia and Schinus species against some plant pathogenic fungi and bacteria. Journal of Plant Pathology 91: 339-345.
- Cochran GW, Cox GM (1992) Experimental Designs. 2nd edn. John Wiley and Sons, United States.
- Onyeke P, Ugwuoke C (2011) Effect of inorganic fertilizer on cowpea production and yield. African Journal of Agricultural Science 60: 846-849.
- Rachie P, Vanderlinde R, Dutra S, Marcon A (2005) Control of pest and diseases on cowpea seed. Text Book of Agricultural Science. pp: 13.
- Avita EJ (2013) Phytochemical screening and bioactivity assay of selected South Indian phytolaccaceae. J Nat Life Sci 1: 26-30.