Keywords
Biomarkers; GM-CSF antibodies; Tregs; Biologics; Pediatric IBD
Introduction
What is known
• The incidence of inflammatory bowel disease (IBD) in pediatric population has doubled in the past 2 decades.
• IBD is recognized as immune dysregulation that fails to maintain gut homeostasis between intraluminal intestinal microbiota, host inflammatory mediators and host regulatory immune response [1,2].
• Granulocyte-macrophage colony-stimulating factor (GMCSF) has an important role in the pathogenesis of intestinal immune and inflammatory processes and the regulation of gut homeostasis.
What is new
• Levels of GM-CSF antibodies and peripheral Tregs may have value in predicting the choice of therapy in pediatric IBD.
• A combination of biomarkers and their potential for incorporation in a predictive model may be more useful than any individual biomarker, to predict the need for early biologic therapy in pediatric IBD.
The incidence of inflammatory bowel disease (IBD) in pediatric population has doubled in the past 2 decades. With more intensive research delving into the pathogenesis of the disease process, IBD is increasingly being recognized as an immune dysregulation that fails to maintain gut homeostasis between intraluminal intestinal microbiota, host inflammatory mediators and the host regulatory immune response [1,2].
Granulocyte-macrophage colony-stimulating factor (GMCSF) has an important role in the pathogenesis of intestinal immune and inflammatory processes and the regulation of gut homeostasis [3-6]. Prior studies in a mouse model showed that GM-CSF-deficient mice developed more severe colitis in response to mucosal injury [2,3,7]. GM-CSF antibodies in high titers are associated with reduced intestinal GM-CSF bioactivity, reduced neutrophil bacterial killing, increased intestinal permeability, and more severe mucosal injury in IBD [5,8]. Recent studies have correlated higher GM-CSF antibody levels with stricturing disease or luminal narrowing as evidenced by magnetic resonance enterography [5,9,10].
The human immune system is pivotal in maintaining a dynamic balance between intestinal luminal microbiota and host protective immune responses by using complex regulatory mechanisms [11]. The balance between host and intestinal flora is maintained through the cooperation of various regulatory mechanisms that prevent the immune system from reacting toward harmless exogenous antigens present in the intestine. Among the cells mediating dominant tolerance, Foxp3+regulatory T cells (Tregs) play an essential role in intestinal homeostasis [11-14]. Foxp3+Tregs are reportedly reduced in peripheral blood in patients with active IBD when compared to patients in remission [11,15].
In most adult patients, generally steroids and aminosalicylates for induction of remission is successful, making the step-up approach a viable option to avoiding the cost and potential complications of biologics [16-22]. However, children with IBD often have a severe presentation and a predilection for colitis. It is also important to note that early mucosal healing is correlated with longer remission periods in IBD and reduced steroid dependence, thereby justifying use of biologics when steroids fail to induce mucosal healing at diagnosis [16,18,23,24].
Although follow-up endoscopy to verify mucosal healing is the strongest predictor of drug efficacy, it is expensive to perform repeated colonoscopies in children to determine response to therapy [21]. Additionally, fecal calprotectin, a marker of neutrophil infiltration, has been shown to be helpful in assessing disease activity but requires stool collection, a return visit to clinic, and mailing out the sample to a reference lab [25-28]. For these reasons, we proposed to determine the predictive value of 2 new non-invasive biomarkers in the blood that could be drawn at the time of examination.
We conducted a prospective translational study comparing GM-CSF antibodies and peripheral Tregs as biomarkers to predict response to first-line medications versus step-up to biologics for remission in pediatric IBD. Our primary aim was to measure these non-invasive biomarkers in active IBD and remission, to determine if a significant increase in GM-CSF antibodies and/or a decrease in peripheral Tregs predicted a need for step-up therapy to biologics in IBD. Our secondary aim was to assess the trend of these biomarkers in response to therapy and disease activity.
Methods
All pediatric patients with active IBD seen at the University of Texas Health Science Center at Houston, Pediatric GI clinic and Children’s Memorial Hermann Hospital in the Texas Medical Center were enrolled from November 2014 to March 2016. The study was approved by the Institutional Review Board (IRB # HSC-MS-14-0711) and the Institutional Biosafety Committee at the University of Texas Health Science Center at Houston.
Eligibility
We included all pediatric patients aged 4-18 years presenting with symptoms suggestive of IBD and elevation in blood inflammatory markers e.g. erythrocyte sedimentation rate (ESR), C-reactive protein (CRP). All patients were endoscopically diagnosed with Crohn’s Disease or Ulcerative Colitis with biopsy confirmation. We excluded those who had recently required steroids within the past 2 months, or any other form of immunosuppression. We also excluded patients with evidence of enteric infection or infestation detected by laboratory screening. Written parental/legal guardian informed consent and subject assent (for children 7 years of age or older) was obtained prior to study enrollment.
Activity index
Patients and caregivers were provided with a questionnaire to obtain a validated Activity Index (Figure 1).

Figure 1: Patients and caregivers provided with a questionnaire.
That objectively assesses their disease severity as mild or moderate-severe based on points allotted to severity of abdominal pain, frequency of bowel movements, blood in stools, night-time awakenings, limitation of daily activities, weight loss and constitutional symptoms of fever, joint pains, fatigue, skin rash or mouth sores. These questions allowed calculation of a PCDAI (Pediatric Crohn’s Disease Activity Index) or a PUCAI (Pediatric Ulcerative Colitis Activity Index) along the recent NASPGHAN guidelines [29]. The Activity Index was monitored after treatment to document clinical remission (Tables 1 and 2).
| History (Recall, 1 week) |
| Abdominal Pain |
Score |
| |
0 - None |
5 = Mild: Brief,
does not
interfere with activities |
10 = Moderate/ Severe: Daily, longer lasting, affects activities, nocturnal |
|
| Patient Functioning, General Well-Being |
Score |
| |
0 = No limitation of activities, well |
5 = Occasional difficulty in maintaining age appropriate activities,
below par |
10 = Frequent limitation of activity, very poor |
|
| Stools (per day) |
Score |
| |
0=0-1 liquid stools, no blood |
5 = Up to 2 Semi formed with small blood, or 2-5 liquid |
10 = Gross bleeding,
≥ 6 liquid, or nocturnal diarrhea |
|
| Examination |
| Abdomen |
Score |
0 = No tenderness,
no mass |
5 = Tenderness,
or mass without
tenderness |
10 = Tenderness, involuntary guarding, definite moss |
|
| Perirectal Disease |
Score |
| |
0 = None, asymptomatic tags |
5 = 1-2 indolent fistula, scant drainage, no tenderness |
10 = Active fistula
drainage, tenderness,
or abscess |
|
| Weight |
Score |
| |
0 = Weight gain or voluntary weight stable/loss |
5 = Involuntary weight stable, weight loss 1%-9% |
10 = Weight loss
≥ 10% |
|
| Extra intestinal Manifestations |
Score |
| (Fever ≥ 38.5°C for 3 days over past week, definite arthritis, uveitis, E. nodosum, P. gangrenosum) |
|
| 0 = None |
5 = 1 |
10 = ≥ 2 |
|
| Total Score: |
|
Table 1: Activity index PCDAI (Abbreviated PCDAI)
| Item |
Points |
| 1.Abdominal pain |
|
| No pain |
0 |
| Pain can be ignored |
5 |
| Pain cannot be ignored |
10 |
| 2.Rectal bleeding |
|
| None |
0 |
| Small amount only, in < 50% of stools |
10 |
| Small amount with most stools |
20 |
| Large amount (> 50% of stool content) |
30 |
| 3.Stool consistency of most stools |
|
| Formed |
0 |
| Partially formed |
5 |
| Completely unformed |
10 |
| 4.Number of stools per 24 hours |
|
| 0-2 |
0 |
| 3-5 |
5 |
| 6-8 |
10 |
| > 8 |
15 |
| 5. Nocturnal stools (any episode causing wakening) |
|
| No |
0 |
| Yes |
10 |
| 6. Activity level |
|
| No limitation of activity |
0 |
| Occasional limitation of activity |
5 |
| Severe restricted activity |
10 |
| TOTAL MAXIMUM SCORE |
85 |
Monitoring Disease Activity in Paediatric IBD patients. A CME Monograph series by NASPGHAN & CDHNF.
Table 2: Activity index PUCAI (Abbreviated PUCAI)
Study design
Routine labs and stool studies including complete blood count with differential, electrolyte panel, ESR, CRP and fecal calprotectin were ordered at the initial visit as part of routine standard of care. Blood was collected for the research study on endoscopic and pathological confirmation of IBD. The blood was than analyzed for GM-CSF antibodies and peripheral Tregs by enzyme-linked immunosorbent assay (ELISA) and flow cytometry respectively.
Plasma was stored immediately at -80 degrees C until performing ELISA to detect the levels of GM-CSF auto antibodies. The specimens were shipped to Cincinnati Children’s Hospital Medical Center, Cincinnati, OH for ELISA testing to detect GM-CSF antibody levels.
For Flow cytometry, fresh blood samples in EDTA-treated tubes were centrifuged for 15 minutes at 350 g to separate plasma from blood cells. Peripheral blood mononuclear cells (PBMCs) were isolated from whole blood by using Ficoll-Paque (GE Healthcare), and immediately stained with T cell surface CD markers, CD4 (T helper) and CD25 (high expression in Treg population) and intracellular Foxp3+ (specific marker for functional Tregs) [30]. To ensure highly reliable results, all samples were processed on the same day of phlebotomy for surface and intracellular staining without freezing. The cells were then analyzed by flow cytometry (FACS Caliber; Becton Dickinson) and FlowJo software (TreeStar, Inc) [30-34].
Patients were subsequently followed prospectively to assess response to first-line medications (most often, mesalamine and/or steroids) and stratified into 2 groups: non-biologic (responding to first-line therapy) and biologic group. The latter included those treated with escalation of care to biologics: anti-TNF-alpha (Remicade or Humira). GM-CSF antibodies, peripheral Tregs and fecal calprotectin were monitored in both groups in remission, at the completion of induction phase. Differences in these biomarkers between the groups at diagnosis, and the change in these biomarkers after treatment were analyzed statistically at the end of the study. The study methods have been summarized (Figure 2).
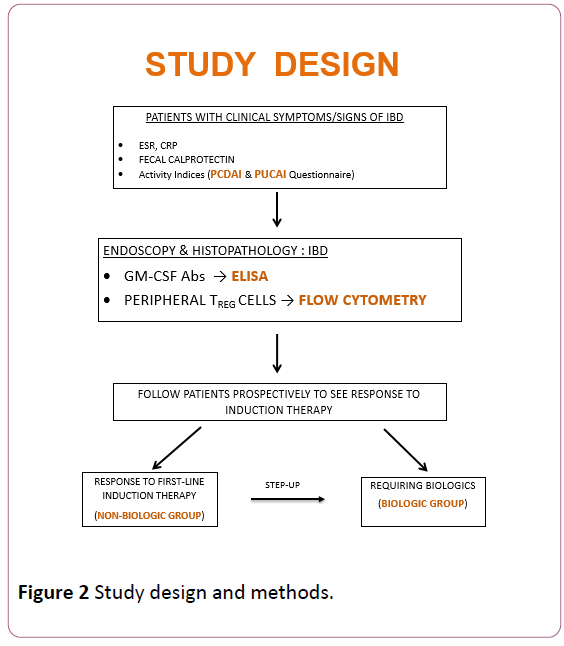
Figure 2: Study design and methods.
Statistical analysis
Wilcoxon Rank Sum tests were used to compare baseline GM-CSF antibodies and Tregs in the non-biologic vs. biologic groups. The percent decrease in GM-CSF antibodies and increase in Tregs in response to therapy was analyzed by the Wilcoxon paired signed-rank test. The Mann-Whitney test was used to compare the therapeutic change in biomarker levels between the biologic and non-biologic cohorts. Multivariate logistic regression analysis was done to see if any individual laboratory variable including the two study biomarkers or a combination of variables predicted a need for early biologic therapy. A two-sided p-value of 0.05 was considered significant.
Results
We enrolled 31 patients in the study, of whom 28 were diagnosed with IBD on endoscopy and biopsy confirmation. Of the 28 patients, 14 had CD (50%). Eleven out of 23 patients (48%) required biologics, while 52% patients responded to first-line medications. Of the 12 who responded to first line medications, 1 required only mesalamine for induction of remission while 3 required addition of a topical steroid.
The remaining 8 required oral prednisone for clinical remission, followed by primary maintenance with immunomodulators such as azathioprine. Of the 11 requiring biologics, 8 had Crohn’s disease (72%) while 75% of the nonbiologic group had ulcerative colitis. Figure 3 shows the flow diagram for study participants.
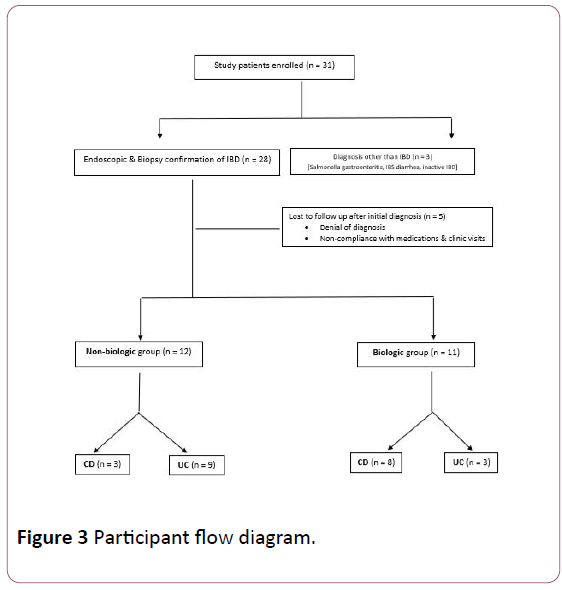
Figure 3: Participant flow diagram.
A total of 18 of the 28 patients completed the study, with both pre- and post-treatment samples. We were able to obtain baseline blood samples and to follow another 5 patients to assess response to first-line medications or step-up to biologics; however, these 5 failed to return for a posttreatment sample after remission.
Table 3 shows the demographic, clinical and laboratory variables in the non-biologic (first-line medications) and biologic groups. Study subjects’ ages ranged from 5 to 18 years; 61% were male and 39% female. Caucasians comprised 73% of the biologic group while Hispanics comprised 50% of the non-biologic group.
| Variable |
Non-biologic group (n=12) |
Biologic group (n=11) |
p-value |
| n |
% |
n |
% |
| Mean Age (years) |
14 |
- |
13 |
- |
0.852 |
| Gender |
Male |
5 |
41.7 |
8 |
72.7 |
0.214 |
| Female |
7 |
58.3 |
3 |
27.3 |
| Ethnicity |
Caucasian |
3 |
25 |
8 |
72.7 |
0.015 |
| African American |
2 |
16.7 |
3 |
27.3 |
| Hispanic |
6 |
50 |
0 |
0 |
| Asian |
1 |
8.3 |
0 |
0 |
| IBD subtype |
Crohn’s Disease |
3 |
25 |
8 |
72.7 |
0.039 |
| Ulcerative Colitis |
9 |
75 |
3 |
27.3 |
| Penetrating/Perianal Crohn’s |
0 |
0 |
5 |
45.5 |
0.014 |
| Improvement in Activity Index |
≥ 90% |
9 |
75 |
5 |
45.5 |
0.214 |
| <90% |
3 |
25 |
6 |
54.5 |
| ESR |
>20 mm/hr |
6 |
50 |
7 |
63.6 |
0.68 |
| ≤ 20 mm/hr |
6 |
50 |
4 |
36.4 |
| CRP |
>2.9 mg/L |
7 |
58.3 |
9 |
81.8 |
0.371 |
| ≤ 2.9 mg/L |
5 |
41.7 |
2 |
18.2 |
| Calprotectin |
≥ 1000 µg/g |
5 |
41.7 |
9 |
81.8 |
0.089 |
| <1000 µg/g |
7 |
58.3 |
2 |
18.2 |
| Hemoglobin |
<11 g/dL |
4 |
33.3 |
6 |
54.5 |
0.414 |
| ≥ 11 g/dL |
8 |
66.7 |
5 |
45.5 |
| Albumin |
<3.5 g/dL |
4 |
33.3 |
8 |
72.7 |
0.1 |
| ≥ 3.5 g/dL |
8 |
66.7 |
3 |
27.3 |
Table 3: Baseline demographic, clinical and laboratory characteristics of the non-biologic and biologic groups.
Of the patients in the biologic group, 46% had penetrating or perianal Crohn’s Disease. Forty-five % of the biologic group showed a ≥ 90% improvement in activity indices following treatment, compared to 75% in the non-biologic group (p=0.21). As seen in Table 3, there are no significant differences in laboratory parameters, including ESR, CRP or hemoglobin between the biologic and non-biologic cohorts.
Albumin < 3.5 was seen in 73% of the biologic group compared to 33% of the non-biologic group (p=0.10). Fecal calprotectin was also noted to be ≥ 1000 in 82% of the biologic group compared to 42% of the non-biologic group (p=0.08). Initial biomarker levels were compared between the biologic and non-biologic groups (Figure 4).
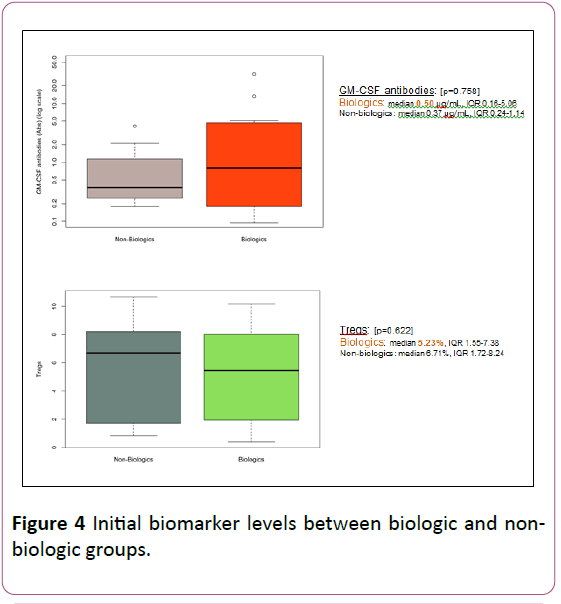
Figure 4: Initial biomarker levels between biologic and nonbiologic groups.
GM-CSF antibodies were numerically higher in biologics than non-biologics group, though not statistically significant (median 0.50 μg/mL, IQR 0.16-5.06 in biologics vs. median 0.37 μg/mL, IQR 0.24-1.14 in non-biologics; p=0.57).
Tregs, although numerically lower in the biologic group, were also not found to significantly differ, when comparing the two groups (median 5.23%, IQR 1.55-7.38 in the biologic group vs. median 6.71%, IQR 1.72-8.24 in non-biologic group; p=0.62).
GM-CSF antibodies were found to be higher in Crohn’s disease than ulcerative colitis, as seen in Figure 5. This approached statistical significance in our study (median 0.81 μg/mL, IQR 0.35-7.02 in CD vs. median 0.31 μg/mL, IQR 0.19-1.04 in UC; p=0.07). Tregs were lower in CD as compared to UC though not statistically significant (median 5.07%, IQR 1.55-8.29 in CD vs. median 6.31%, IQR 2.31-8.15 in UC; p=0.74).
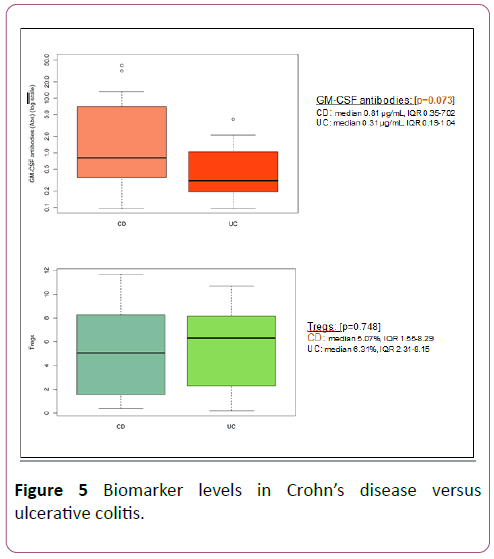
Figure 5: Biomarker levels in Crohn’s disease versus ulcerative colitis.
Following induction therapy, GM-CSF antibodies declined in 39% of all treated patients and specifically declined in 38% of the biologic group. The decline in GM-CSF was more in the biologics group although the median change was similar between the groups (median change 0.05 μg/mL, IQR-0.55-4.52 in biologics vs. median change 0.02 μg/mL, IQR-0.06-0.22 in non-biologics; p=0.78). Tregs increased following induction therapy in 67% of all treated patients and specifically in 75% of the biologic group.
The rise in Tregs was more in the biologics group although the median change was similar between the groups (median change 1.23%, IQR-1.95-3.61 in biologics vs. median change 0.87%, IQR-4.67 to 3.17 in non-biologics; p=0.65). The change in biomarker levels in response to therapy is shown in Figure 6.
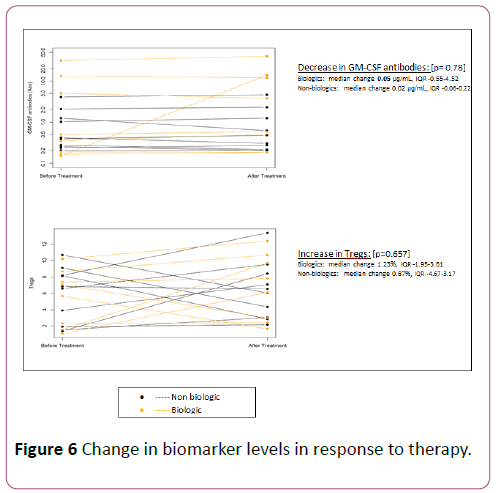
Figure 6: Change in biomarker levels in response to therapy.
Figure 7 shows a representative flow cytometry plot of Foxp3+Tregs before and after therapy.
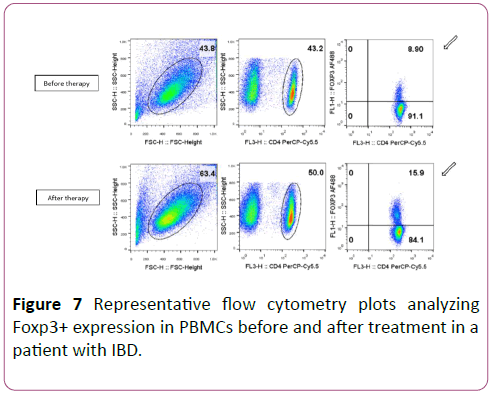
Figure 7: Representative flow cytometry plots analyzing Foxp3+ expression in PBMCs before and after treatment in a patient with IBD.
An ROC analysis was done for baseline GM-CSF antibodies and Tregs to predict need for biologics. GM-CSF antibodies ≥ 4.3 μg/mL showed the highest accuracy with 100% specificity but low sensitivity of 36.4% (AUC of 0.68) as seen in Figures 8 and 9.
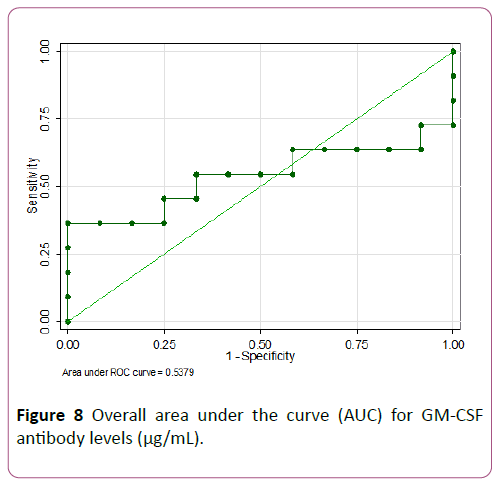
Figure 8: Overall area under the curve (AUC) for GM-CSF antibody levels (μg/mL).
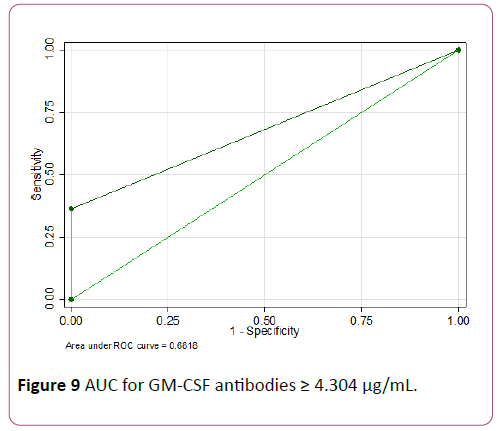
Figure 9: AUC for GM-CSF antibodies ≥ 4.304 μg/mL.
Tregs ≤ 7.4% showed the highest accuracy with a sensitivity of 81.8% but lower specificity of 41.7% (AUC of 0.62) (Figures 10 and 11).
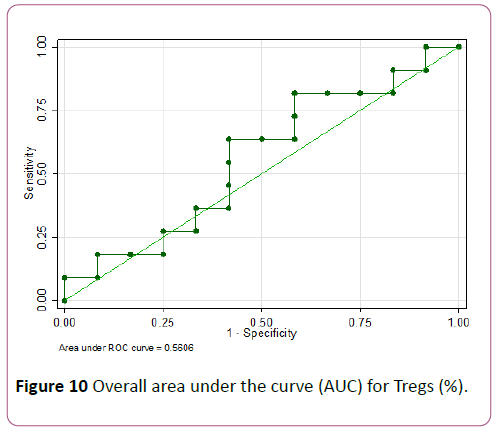
Figure 10: Overall area under the curve (AUC) for Tregs (%).
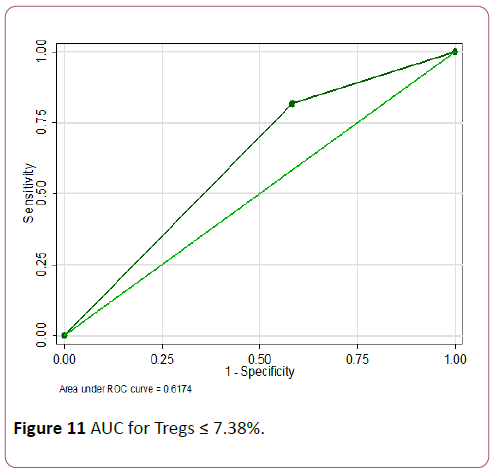
Figure 11: AUC for Tregs ≤ 7.38%.
Fecal calprotectin at diagnosis didn’t show any significant difference between the biologic and non-biologic groups (median 1280 μg/g, IQR 1072 to >2000 in biologics group vs. median 403.9 μg/g, IQR 215 to >2000 in non-biologics group; p=0.13). Calprotectin did not differ between the CD and UC groups (median 1224 μg/g, IQR 422 to >2000 in CD vs. median 949 μg/g, IQR 225 to >2000 in UC; p=0.25). Laboratory parameters routinely used in clinic were compared between biologic and non-biologic cohorts and in CD vs. UC.
ESR and CRP were significantly higher and albumin significantly lower in CD compared to UC (median ESR 32.5 mm/hr in CD vs. 8.5 mm/hr in UC; p=0.01, median CRP 23.2 mg/L in CD vs. 2.9 mg/L in UC; p=0.007 and median albumin 3 g/dL in CD vs. 4 g/dL in UC; p=0.020). No differences in these parameters were noted between the biologic and non-biologic groups. A multivariate logistic regression analysis did not show any individual biomarker to significantly predict a need for early biologic therapy.
Discussion
Inflammatory bowel disease results from an immune dysregulation involving Tregs, as well as a dysfunction of the innate immune system, which includes neutrophils and macrophages. GM-CSF is required for priming of monocyte/ macrophage and neutrophil antimicrobial functions. An altered bioactivity of GM-CSF, therefore, likely plays a role in the innate immune dysregulation in CD. This immune dysfunction leads to increased bowel permeability and progression of disease type from primarily inflammatory, to penetrating and stricturing disease later in the process [2,9]. The impaired intestinal barrier function and gut homeostasis is associated with reduced GM-CSF bioactivity and monocyte/ neutrophil function, resulting in high titers of endogenous anti-GM-CSF antibodies (GM-CSF Ab) with increasing gut permeability [5].
Foxp3+Tregs play an important role in maintenance of intestinal homeostasis through IL-10 and TGF-β1 – dependent mechanisms [35]. Tregs can produce IL-10 and TGF-β. IL-10 acts on innate immune cells to reduce inflammation, whereas TGF-β inhibits effector cells directly and promotes epithelial homeostasis. In addition, TGF-β can synergize with other cytokines to promote the differentiation of Th17 cells that promote IL-17 and IL-22, which also contributes to the barrier function [11,36]. Tregs are characterized by high levels of the transcription factor Foxp3, which is considered to confer their regulatory activity [37].
There is a proven underlying pathogenesis involving a complex interplay between host genes, microbial and food antigens and the genetically mediated immune response of the host to these antigens, which influences the severity of presentation in IBD. GM-CSF antibodies and Tregs are likely to play a central role in this pathogenesis. Current available blood markers like ESR and CRP and stool markers of inflammation such as fecal calprotectin are elevated in response to the inflammation associated with IBD [25]. GM-CSF antibodies and peripheral Tregs are an integral part of the immunopathogenesis of the disease process, hence are impacted earlier in the disease process and may have potential for more accuracy and reliability as predictive markers of response to therapy [8]. This was the basis for our hypothesis that increased GM-CSF antibodies and/or decreased Tregs may predict a need for earlier biologic therapy for improved mucosal healing in IBD.
This study suggests, though does not prove, that higher GMCSF antibodies may be associated with a need for biologic therapy. GM-CSF antibodies were also found to be higher in Crohn’s Disease compared to Ulcerative Colitis. Furthermore, antibody levels declined in response to therapy with a greater decline in the biologic group. Prior studies have shown a statistically significant difference in levels of GM-CSF antibodies when measured during active disease compared to values obtained at remission [2,5,8-10]. Nylund et al suggested that disruption of the intestinal barrier in IBD could lead to permeation of luminal antigens and increased bacterial translocation. This stimulates memory B cells to upregulate GM-CSF autoantibodies [2]. Dabritz, et al. reported a clear increase in GM-CSF antibodies in the months preceding a relapse where they studied patients longitudinally with serial measurements of GM-CSF antibody levels. They theorized that fluctuating antibody levels during remission versus relapse affect myeloid functions and result in an impaired modulation of gut homeostasis and gut mucosal barrier function [8].
Jurickova, et al. showed a significant correlation and inverse relation between GM-CSF antibodies and bacterial killing; the latter decreasing as serum GM-CSF autoantibodies increase [5]. This is seen progressively with a rise in GM-CSF antibodies from 1 to 10 μg/mL and studies in adults have shown levels >5 μg/mL to be the cut-point where GM-CSF antibodies start to inhibit neutrophil anti-bacterial function [8]. Gathungu, et al. showed an association of GM-CSF antibodies ≥ 4 μg/mL with aggressive ileal disease [10]. An ROC analysis in our study showed that GM-CSF antibodies ≥ 4.3 μg/mL had the highest accuracy of predicting need for biologics.
There were 5 patients in our study who had GM-CSF antibody levels >5 μg/mL. Of these, 4 patients had radiological evidence of luminal narrowing, intestinal strictures or microperforation with intra-abdominal abscess and all of them had Crohn’s disease. Dykes et al showed similar results with GMCSF antibodies ≥ 1.6 μg/mL associated significantly with luminal narrowing and CD patients being 5 times as likely to have a stricture on CTE or MRE [9]. Han et al also found elevated GM-CSF antibodies to be associated with a 2-fold higher rate of stricturing/penetrating disease behavior [7].
One patient had more than a 90-fold increase in GM-CSF antibody levels following therapy and clinical remission. This followed a prolonged course of refractory IBD that finally responded to biologics. The only plausible explanation seemed to be an early indication of relapse in this patient who shortly thereafter, went on to receive a higher dose of biologics with increased frequency of administration. This phenomenon was also noted by Dabritz et al in their study on GM-CSF antibody levels in patients with relapse, where they explain an elevated serum GM-CSF antibody titer in patients in clinical remission to represent an early stage of increased intestinal permeability and bacterial translocation [8]. Thus increasing GM-CSF antibody levels in a patient in clinical remission, may herald a secondary loss of response with a subsequent relapse, especially in CD that involves transmural inflammation.
Our study also suggested that lower Tregs may be associated with a need for biologic therapy. There was an increase in Tregs after treatment in 75% of the biologic group. Although not statistically significant, this increase was greater after biologic therapy, similar to prior studies [32,38]. Wang, et al. also reported higher Tregs in inactive versus active CD and UC, however this did not reach statistical significance [15]. Tregs were also found to trend lower in patients with Crohn’s disease, comparable to prior studies [39,40]. An ROC analysis in our study showed Tregs ≤ 7.4% to have the highest accuracy to predict a need for biologics. Saruta et al reported peripheral Tregs ranging from 5%-8% in active versus 9%-12% in inactive disease [41]. Li, et al. reported Tregs ranging from 3%-6% in active IBD that increased to 8%-16% following anti-TNFα therapy [38].
Tregs are anti-inflammatory and are proposed to mediate many of the beneficial effects on gut inflammation [31,32,35,42]. The severity of disease activity in IBD is inversely correlated with the frequency of peripheral Tregs [15,41,43,44]. This contraction of the peripheral pool of Tregs is postulated to be due to an insufficient counter-regulation to inflammation, rather than their flowing out from the circulation and accumulating in inflamed intestinal tissue [40]. There is, in turn, a significant increase in frequency of peripheral Tregs in response to treatment with biologics [39]. The increased apoptotic activity of Tregs in active IBD declines with response to anti-TNF therapy, and their numbers increase. Six out of 18 patients in our study did not have an increase in Tregs after treatment. Lack of improvement in Tregs after treatment has been attributed to alternate immune pathways in the intestinal mucosa, not involving Foxp3+Tregs during remission, in prior studies [39].
Fecal calprotectin did not significantly differ in the biologic and non-biologic cohorts or in CD versus UC in this study. Calprotectin, a gut inflammatory marker released from neutrophils at the site of intestinal inflammation, has been invaluable as a screening tool preceding invasive endoscopy. However, Henderson, et al. who studied calprotectin extensively in the pediatric population found that the level was not influenced by disease type (CD vs. UC) or location [28]. Previous studies have also shown that calprotectin has poor correlation with the clinical activity indices although better correlation with endoscopic indices of severity [26,45].
PCDAI and PUCAI were used in all our patients throughout the study to obtain an objective assessment of clinical severity and response to therapy. Forty-five% of the biologic group showed a ≥ 90% improvement in activity indices following treatment, compared to 75% in the non-biologic group. This is probably a reflection of the severity of disease in the biologic group that was less responsive to therapy, compared to the milder spectrum of disease in the non-biologic group. Clinical remission and improvement in activity indices was noted to be significant in the biologic cohort only in UC patients (p=0.04). CD patients did not show a significant improvement, indicating that severe CD may require earlier institution of anti-TNF therapy to bring about a significant clinical improvement. Several prior studies have shown short-term response rate with biologics in CD and failure rates ranging from 18 to 46% at 1 and 5 year follow up [16]. Although several factors may cause loss of response including host and drug pharmacokinetics and the development of antibodies to infliximab, the decreased response of CD to anti-TNF agents may reflect the underlying severe transmural inflammation, as well as the delay in starting biologic therapy that results after numerous unsuccessful attempts to treat flares with steroids. This delays mucosal healing further, which may contribute to lesser efficacy of biologics than their potential, in the long run.
Our study showed that higher levels of GM-CSF antibodies and lower levels of peripheral Tregs were associated with a greater need for biologics reflecting disease severity in pediatric IBD. Thus these biomarkers help guide treatment choices from the variety of treatment options available, based on individual disease activity. However, the lack of statistical significance may be attributed to the small sample size in this study. Despite a small sample size, the trends of these novel biomarkers in our study of pediatric patients paralleled those in adult IBD studies with statistical significance. It is also important to note that most prior studies investigating these blood biomarkers were larger studies in adult patients, who may have had more severe inflammation and an advanced disease process, allowing for a greater difference in biomarker levels between cohorts. The biologic cohort also had a larger proportion of Caucasians while there were a larger number of Hispanics in the non-biologic cohort. The effect of this demographic disparity on biomarker levels in this study, however, is unknown, despite studies reporting higher serological markers like p-ANCA in the Hispanic population compared to Caucasians [46].
Although sample size was a limitation of this study, we suggest it had a few strengths. Unlike prior studies comparing mostly between active and inactive IBD or remission and relapse, this study was, to our knowledge, the first to compare groups based on the type of therapy needed, reflecting disease severity. We aimed to evaluate the ability of these biomarkers to guide therapy decisions in our IBD patients. Designed as a prospective study, we were able to collect all clinical variables for an accurate scoring of the activity indices, to objectively assess disease severity and monitor patients for remission. Longer follow-up periods would add more value to assess loss of response and clinical relapse.
Conclusion
This prospective translational study of novel biomarkers in pediatric IBD, although limited by a small sample size, underscores the hypothesis that blood biomarkers like GM-CSF antibodies and Tregs, that play an important role in the underlying immuno-pathogenesis of IBD, may have value in guiding IBD therapy based on disease severity. GM-CSF antibodies ≥ 4.3 μg/mL and Tregs ≤ 7.4% may be used in combination to improve the predictive value of these biomarkers. The greater sensitivity of Treg cells and higher specificity of GM-CSF antibodies may be better combined in a predictive model using both these biomarkers to reflect disease severity. They may also help to identify CD from UC, complementing the serology tests used for evaluation of indeterminate colitis which is very common in the pediatric population. Although the findings in this study of new noninvasive biomarkers were not statistically significant, it does not preclude this work from serving as a pilot study, with promise for larger studies with more robust ROC analyses to improve the predictive accuracy of these non-invasive biomarkers. A combination of such validated biomarkers may help develop a predictive model, to guide management strategies and identify the most effective treatment in personalized treatment regimens. Our pilot study may serve as the basis for future studies, with combinations of these same or similar novel biomarkers involved in pivotal inflammatory pathways in IBD, to help develop a commercially marketable panel of biomarkers that would be more readily available in pediatric hospital and clinic settings. Guided by these biomarkers, we may be able to provide appropriate first-line therapy to those expected to respond, avoiding the potential adverse effects and higher cost of biologics. Conversely, institution of early anti-TNF therapy where indicated, may induce early remission especially in our CD patients, to reduce steroid-resistance and promote mucosal healing. This will ultimately improve quality of life scores and reduce hospitalizations and healthcare cost.
References
- Michail S, Durbin M, Turner D, Griffiths AM, Mack DR, et al. (2012) Alterations in the gut microbiome of children with severe ulcerative colitis. Inflamm Bowel Dis 18: 1799-1808.
- Nylund CM, D'Mello S, Kim MO, Bonkowski E, Däbritz J, et al. (2011) Granulocyte macrophage-colony-stimulating factor autoantibodies and increased intestinal permeability in Crohn disease. J Pediatr Gastroenterol Nutr 52: 542-548.
- Dabritz J (2014) Granulocyte macrophage colony-stimulating factor and the intestinal innate immune cell homeostasis in Crohn's disease. Am J Physiol Gastrointest Liver Physiol 306: G455-G465.
- Egea L, Hirata Y, Kagnoff MF (2010) GM-CSF: A role in immune and inflammatory reactions in the intestine. Expert Rev Gastroenterol Hepatol 4: 723-731.
- Jurickova I, Collins MH, Chalk C, Seese A, Bezold R, et al. (2013) Paediatric Crohn disease patients with structuring behaviour exhibit ileal granulocyte-macrophage colony-stimulating factor (GM-CSF) autoantibody production and reduced neutrophil bacterial killing and GM-CSF bioactivity. Clin Exp Immunol 172: 455-465.
- Xu Y, Hunt NH, Bao S (2008) The role of granulocyte macrophage-colony-stimulating factor in acute intestinal inflammation. Cell Res 18: 1220-1229.
- Han X, Uchida K, Jurickova I, Koch D, Willson T, et al. (2009) Granulocyte-macrophage colony-stimulating factor autoantibodies in murine ileitis and progressive ileal Crohn's disease. Gastroenterol 136: 1261-1263.
- Dabritz J, Bonkowski E, Chalk C, Trapnell BC, Langhorst J, et al. (2013) Granulocyte macrophage colony-stimulating factor auto-antibodies and disease relapse in inflammatory bowel disease. Am J Gastroenterol 108: 1901-1910.
- Dykes DM, Towbin AJ, Bonkowski E, Chalk C, Bezold R, et al. (2013) Increased prevalence of luminal narrowing and stricturing identified by enterography in pediatric Crohn's disease patients with elevated granulocyte-macrophage colony stimulating factor autoantibodies. Inflamm Bowel Dis 19: 2146-2154.
- Gathungu G, Kim MO, Ferguson JP, Sharma Y, Zhang W, et al. (2013) Granulocyte-macrophage colony-stimulating factor autoantibodies: a marker of aggressive Crohn's disease. Inflamm Bowel Dis 19: 1671-1680.
- Izcue A, Coombes JL, Powrie F (2009) Regulatory lymphocytes and intestinal inflammation. Annu Rev Immunol 27: 313-338.
- Barnes MJ, Powrie F (2009) Regulatory T cells reinforce intestinal homeostasis. Immunity 31: 401-411.
- Boehm F, Martin M, Kesselring R, Schiechl G, Geissler EK, et al. (2012) Deletion of Foxp3+ regulatory T cells in genetically targeted mice supports development of intestinal inflammation. BMC Gastroenterol 12: 97.
- Izcue A, Coombes JL, Powrie F (2006) Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol Rev 212: 256-271.
- Wang Y, Liu XP, Zhao ZB, Chen JH, Yu CG (2011) Expression of CD4+ forkhead box P3 (FOXP3)+ regulatory T cells in inflammatory bowel disease. J Dig Dis 12: 286-294.
- Assa A, Hartman C, Weiss B, Broide E, Rosenbach Y, et al. (2013) Long-term outcome of tumor necrosis factor alpha antagonist's treatment in pediatric Crohn's disease. J Crohns Colitis 7: 369-376.
- Bar-Meir S (2009) Mild to moderate Crohn's disease: Still room for step-up therapies? Dig Dis 27: 347-350.
- Lakatos PL, Lovasz BD, David G, Pandur T, Erdelyi Z, et al. (2013) The risk of lymphoma and immunomodulators in patients with inflammatory bowel diseases: Results from a population-based cohort in Eastern Europe. J Crohns Colitis 7: 385-391.
- Pittet V, Froehlich F, Maillard MH, Mottet C, Gonvers JJ, et al. (2013) When do we dare to stop biological or immunomodulatory therapy for Crohn's disease? Results of a multidisciplinary European expert panel. J Crohns Colitis 7: 820-826.
- Stallmach A, Hagel S, Bruns T (2010) Adverse effects of biologics used for treating IBD. Best Pract Res Clin Gastroenterol 24: 167-182.
- Turner D, Mack D, Leleiko N, Walters TD, Uusoue K, et al. (2010) Severe pediatric ulcerative colitis: a prospective multicenter study of outcomes and predictors of response. Gastroenterol 138: 2282-2291.
- Michetti P, Stelle M, Juillerat P, Gassull M, Heil F, J, et al. (2009) Appropriateness of therapy for active Crohn's disease: Results of a multidisciplinary international expert panel-EPACT II. J Crohns Colitis 3: 232-240.
- Baert F, Moortgat L, Van AG, Caenepeel P, Vergauwe P, et al. (2010) Mucosal healing predicts sustained clinical remission in patients with early-stage Crohn's disease. Gastroenterology 138: 463-468.
- Lichtenstein GR, Feagan BG, Cohen RD, Salzberg BA, Diamond RH, et al. (2014) Drug therapies and the risk of malignancy in Crohn's disease: results from the TREAT Registry. Am J Gastroenterol 109: 212-223.
- Lin JF, Chen JM, Zuo JH, Yu A, Xiao ZJ, et al. (2014) Meta-analysis: Fecal calprotectin for assessment of inflammatory bowel disease activity. Inflamm Bowel Dis 20: 1407-1415.
- Canani RB, Terrin G, Rapacciuolo L, Miele E, Siani MC, et al. (2008) Faecal calprotectin as reliable non-invasive marker to assess the severity of mucosal inflammation in children with inflammatory bowel disease. Dig Liver Dis 40: 547-553.
- Fagerberg UL, Loof L, Lindholm J, Hansson LO, Finkel Y (2007) Fecal calprotectin: A quantitative marker of colonic inflammation in children with inflammatory bowel disease. J Pediatr Gastroenterol Nutr 45: 414-420.
- Henderson P, Casey A, Lawrence SJ, Kennedy NA, Kingstone K, et al. (2012) The diagnostic accuracy of fecal calprotectin during the investigation of suspected pediatric inflammatory bowel disease. Am J Gastroenterol 107: 941-949.
- Bousvaros A, Turner D, Vitito L (2009) Monitoring disease activity in pediatric IBD patients: A case-based monograph focusing on pediatric IBD. NASPGHAN and CDHNF 2009.
- Mangalat N, Liu Y, Fatheree NY, Ferris MJ, Van Arsdall MR, et al. (2012) Safety and tolerability of Lactobacillus reuteri DSM 17938 and effects on biomarkers in healthy adults: Results from a randomized masked trial. PLoSONE 7: e43910.
- Liu Y, Tran DQ, Fatheree NY, Marc Rhoads J (2014) Lactobacillus reuteri DSM 17938 differentially modulates effector memory T cells and Foxp3+ regulatory T cells in a mouse model of necrotizing enterocolitis. Am J Physiol Gastrointest Liver Physiol 307: G177-G186.
- O'Garra A, Vieira P (2004) Regulatory T cells and mechanisms of immune system control. Nat Med 10: 801-805.
- Ross EM, Bourges D, Hogan TV, Gleeson PA, Driel IR (2014) Helios defines T cells being driven to tolerance in the periphery and thymus. Eur J Immunol 44: 2048-2058.
- Thornton AM, Korty PE, Tran DQ, Wohlfert EA, Murray PE, et al. (2010) Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol 184: 3433-3441.
- Tran DQ, Ramsey H, Shevach EM (2007) Induction of FOXP3 expression in naive human CD4+FOXP3 T cells by T-cell receptor stimulation is transforming growth factor-beta dependent but does not confer a regulatory phenotype. Blood 110: 2983-2990.
- Eastaff-Leung N, Mabarrack N, Barbour A, Cummins A, Barry S (2010) Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. J Clin Immunol 30: 80-89.
- Hori S, Nomura T, Sakaguchi S (2003) Control of regulatory T cell development by the transcription factor Foxp3. Science 299: 1057-1061.
- Li Z, Arijs I, De HG, Vermeire S, Noman M, et al. (2010) Reciprocal changes of Foxp3 expression in blood and intestinal mucosa in IBD patients responding to infliximab. Inflamm Bowel Dis 16: 1299-1310.
- Guidi L, Felice C, Procoli A, Bonanno G, Martinelli E, et al. (2013) FOXP3(+) T regulatory cell modifications in inflammatory bowel disease patients treated with anti-TNFalpha agents. Biomed Res Int 2013: 286368.
- Maul J, Loddenkemper C, Mundt P, Berg E, Giese T, et al. (2005) Peripheral and intestinal regulatory CD4+ CD25(high) T cells in inflammatory bowel disease. Gastroenterol 128: 1868-1878.
- Saruta M, Yu QT, Fleshner PR, Mantel PY, Schmidt-Weber CB, et al. (2007) Characterization of FOXP3+CD4+ regulatory T cells in Crohn's disease. Clin Immunol 125: 281-290.
- Liu Y, Fatheree NY, Dingle BM, Tran DQ, Rhoads JM (2013) Lactobacillus reuteri DSM 17938 changes the frequency of Foxp3+ regulatory T cells in the intestine and mesenteric lymph node in experimental necrotizing enterocolitis. PLoS ONE 8: e56547.
- Himmel ME, Hardenberg G, Piccirillo CA, Steiner TS, Levings MK (2008) The role of T-regulatory cells and Toll-like receptors in the pathogenesis of human inflammatory bowel disease. Immunol 125: 145-153.
- Yu QT, Saruta M, Avanesyan A, Fleshner PR, Banham AH, et al. (2007) Expression and functional characterization of FOXP3+ CD4+ regulatory T cells in ulcerative colitis. Inflamm Bowel Dis 13: 191-199.
- Belei O, Simedrea I, Ilie R, Daescu C, Tamara M, et al. (2011) Correlations of fecal calprotectin with clinical and endoscopic scores in inflammatory bowel diseases in children. Jurnalul Pediatrului 14: 18-25.
- Torres E, Cruz A, Monagas M, Bernal M, Correa Y, et al. (2012) Inflammatory bowel disease in Hispanics: The University of Puerto Rico IBD Registry. Inter J Inflam 1-5.