- (2013) Volume 14, Issue 6
Sirio Fiorino1*, Andrea Cuppini1, Gastone Castellani2, Maria Letizia Bacchi-Reggiani3 and Elio Jovine4
1Unit of Internal Medicine, Hospital of Budrio. Budrio, BO, Italy. Institutes of 2Fisics and 3Cardiology, “Alma Mater Studiorum” – University of Bologna, S. Orsola-Malpighi Hospital. Bologna, Italy.
4Unit of General Surgery, Maggiore Hospital. Bologna, Italy
Pancreatic carcinoma is one of the most lethal cancers in humans. The poor prognosis of this malignancy depends on several factors, such as: lack of early symptoms, advanced stage at detection, early metastatic spread and no effective systemic treatment. To date, only few risk factors for this malignancy are known; therefore, considerable efforts are required to identify additional causative agents involved in the process of pancreatic carcinogenesis. In the last years, a large series of epidemiological investigations have suggested that both bacteria and viruses may play a important role in the initiation and progression of several animal and human cancers. In particular, some studies have showed that hepatitis B (HBV) andhepatitis C (HCV) viruses, two hepatotropic pathogens with well-known oncogenic properties for liver, may be detected alsoin extra-hepatic tissues, such as pancreas. The aim of this paper is to briefly report the results of available studies, assessingthe possible association between HBV/HCV and pancreatic cancer development as well as to discuss the limiting factors ofthese researches.
Carcinoma, Hepatocellular; Hepacivirus; Hepatitis B virus; Pancreatic Carcinoma; Pancreatic Neoplasms; Risk Factors
Pancreatic adenocarcinoma is a very aggressive disease with a poor prognosis and an overall fiveyear survival less than 5% [1], accounting for about 250,000 deaths worldwide yearly [2]. Advanced stage at presentation, aggressive behavior, early metastatic dissemination and lack of effective systemic therapies explain the dismal outcome of this neoplasia [3, 4]. To date, causes of this malignancy are not yet completely understood [4]. Cigarette smoking habit [5] and family history strongly increase the risk of this tumor [6] as well as a more elevated incidence of pancreatic adenocarcinoma has been observed in patients with high dietary fat consumption [7], alcohol abuse [8], diabetes mellitus [9], metabolic syndrome [10, 11] and chronic pancreatitis history [12]. Therefore, a better knowledge of risk factors for pancreatic adenocarcinoma development is absolutely required. Epidemiological and experimental researches suggest that about 15-20% of human cancers are associated in some way with chronic infection [13].
In the last decade, a large series of studies reported that both bacteria and viruses may play a role as risk factors for pancreatic carcinogenesis. In particular, concerning infections induced by bacteria, a recent meta-analysis has considered six observational studies, suggesting that Helicobacter pylori infection increases risk of pancreatic cancer development [14]. According to previous studies, it has been already reported that this pathogen is involved in the pathogenesis of peptic ulcer disease [15] as well as of gastric carcinoma and lymphoma [16]. Additional researches proposed a possible relationship between oral pathologies, such as periodontitis, and a higher risk of pancreatic cancer [17, 18]. It has been also suggested that an association exists between variations of patients’ oral flora (in particular some species of salivary microorganisms, such as Neisseria elongate and Streptococcus mitis) and an enhanced incidence of pancreatic diseases, such as neoplasia in this organ [19]. Nevertheless, it is not yet completely understood whether oral microbiota has a causative role in the process of pancreatic carcinogenesis.
Some studies have underlined an association between some viral infection and an increased probability of pancreatic adenocarcinoma. In particular, it has been reported that patients with HIV-related infection, during highly active antiretroviral therapy (HAART) [20, 21], present a significantly higher risk of this tumor in comparison with general population, although other studies did not confirm this relationship [22]. Furthermore, it has been shown an association between exposure to poultry oncogenic viruses and an increased probability of pancreatic adenocarcinoma. Recent reports have suggested that hepatitis B virus (HBV) and hepatitis C virus (HCV) may be involved in the process of oncogenesis in the pancreas. One of the pathogenetic mechanisms by means of which these pathogens promote cancer initiation and progression is represented by the development of a persistent local inflammation, at sites where chronic infection is established [23].
HBV and HCV are liver-tropic pathogens with wellknown oncogenic properties. It has been estimated that these microorganisms infect approximately 400 and 180 million people, respectively. Therefore, they represent a serious public health problem in the world. HBV and HCV may cause a necroinflammatory hepatic disease with different patterns of severity and course. Persistent liver injury represents a high risk condition for developing cirrhosis and hepatocellular carcinoma [24, 25]. Furthermore, a relationship between HBV/HCV and intrahepatic cholangiocarcinoma [26] and some forms of non-Hodgkin lymphoma have been reported [27, 28]. However, antigens and replicative sequences of both pathogens have been also detected in different extra-hepatic tissues, including pancreas [29, 30]. It is well-known that an association exists between HBV- and HCV-related hepatitis and acute pancreatitis [31, 32]. These pathogens might replicate in pancreas, inducing persistent inflammation and injury in this organ. Experimental data suggest that the establishment of a chronic damage may induce the development of metaplasia in tissues where an inflammatory process occurs [33]. This event represents a high risk condition for malignant transformation in involved organs, as well as in pancreas, because it promotes a permissive microenvironment for carcinogenic stimuli [34]. In addition it has to be considered that, according to available data, liver and pancreas originate from common multipotent cells of endoderm origin [35, 36] and that experimental studies have suggested that hepatocytes may trans-differentiate into pancreatic cells and vice versa, when they are cultured upon definite conditions [37, 38]. These data suggest the hypothesis that pancreas is not merely a reservoir of HBV and HCV, but it might support their replication. Therefore, both viruses may have a role not only in liver cancer development, but also in the initiation and progression of pancreatic carcinogenesis. Nevertheless, although in the last decade of the twentieth century some researchers have detected antigens and/or intermediate replicative forms of HBV [39, 40, 41] and HCV [32] in pancreas, no further studies have been performed soon afterwards with the aim of investigating the possible oncogenic activity of both pathogens in this organ. Recently, the interest in the study of the possible association between HBV or HCV infection and pancreatic cancer development, has progressively increased and some researchers have been designed to investigate this topic. In addition, the possible mechanisms involved in the process of HBV- or HCV-induced malignant transformation have been synthesized in a recent paper [42].
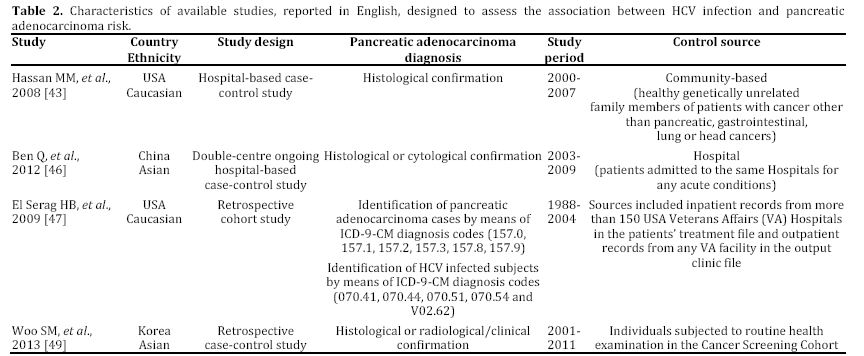
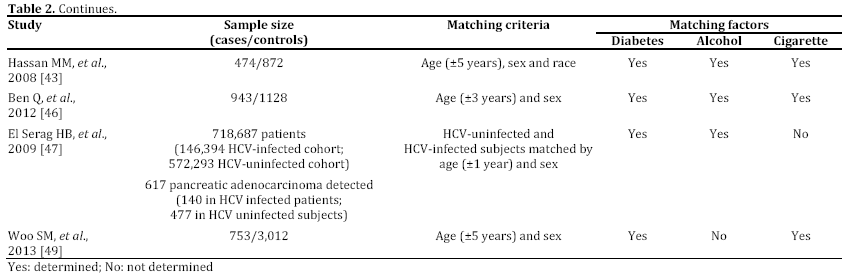
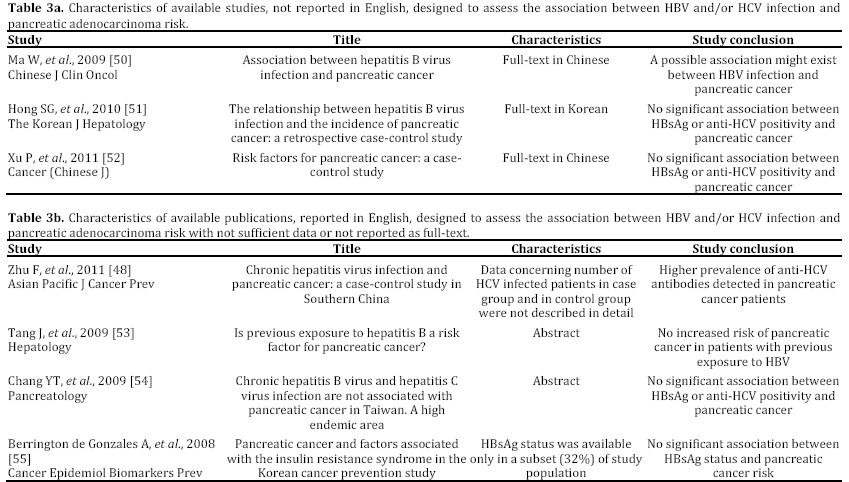
In the last years, some epidemiological studies have been published, suggesting that HBV and HCV may be risk factors not only for liver, but also for pancreatic cancer. Most of these studies, both casecontrol or cohort trials, have been reported in English and performed in a period ranging from 2008 to 2012 and mainly carried out in China [43, 44, 45, 46, 47, 48, 49] (Tables 1 and 2). Some publications are in Chinese or in Korean [50, 51, 52], or in English but not in full-text, or with insufficient data [53, 54, 55] (Table 3). In the last two years, between the end of 2012 and the beginning of 2013, almost 4 meta-analyses, examining the possible association between HBV and HCV infection and pancreatic carcinogenesis, have been published (Table 4). The results of these pooled-analyses support the hypothesis that both viruses may be risk factors also for pancreatic cancer [56, 57, 58, 59] and that patients with signs of persistent HBV or HCV infection have an increased probability to develop this malignancy in comparison to controls without previous contact with both viruses. On the whole, presence of HBsAg is associated with a higher incidence of pancreatic adenocarcinoma in comparison with individuals never exposed to HBV [56, 57, 58, 59]. Furthermore, although no statistically significant relationship has been found between anti-HCV positivity and risk of carcinogenesis in pancreas, a borderline value has been observed in this analysis [59]. The small number of available studies (n=3) assessing this association accounts for this indefinite result. Additional investigations are required to confirm this possible correlation. Therefore, the results from these recent researches provide for the first time very interesting insights on the possible role of hepatotropic viruses in pancreatic adenocarcinoma initiation and progression as well as they may contribute to develop a new field of study. Nevertheless, several questions and uncertain points, requiring a better definition and explanation, remain. In particular, the following problems have to be underlined.
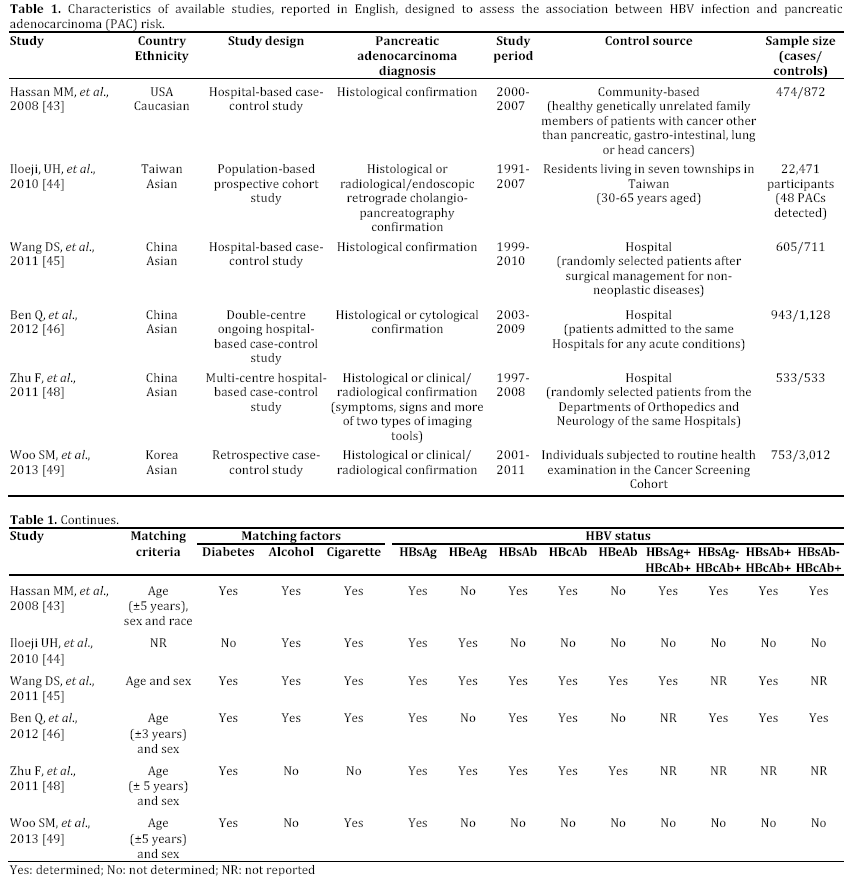
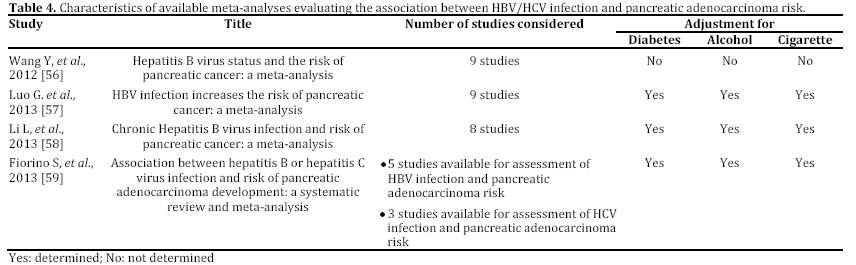
a) Available studies, concerning this topic, are still few, heterogeneous in design, and, in some cases, underpowered.
b) Nearly all researches have been performed in Asian populations and, in particular, in China, where viral liver infections are more prevalent. To date, no studies have been carried out in Europe and in Africa, whereas only two trials have been accomplished in Northern America (USA) [43, 47]. Therefore, the validity of the available researches and meta-analyses is limited to Asian peoples and it may be erroneous to extend their results to non Asian peoples.
c) Concerning HBV infection, it should be considered that in high HBV endemic regions worldwide, such as China and Taiwan, HBsAg prevalence is about 8%, whereas in low HBV endemic countries [60], including USA or Northern Europe, global HBsAg prevalence is less than 2% in general population [61]. In these areas, most of subjects with a previous contact with HBV have a serum profile characterized by absence of HBsAg and by presence of HBcAb with or without HBsAb. Therefore, these individuals present two different types of serum HBV antigen/antibody markers, including HBsAg-/HBcAb+/HBsAb- or HBsAg- /HBcAb+/HBsAb+ patterns. In the past these profiles have been considered as signs of complete recovery from a past exposure to HBV infection without or with natural immunity. Recently, progressive advancement in molecular biology and introduction of highly sensitive techniques for HBV genotyping allowed us to understand that this virus is able to persist and replicate at low level also in individuals with or without presence of serum HBcAb and/or HBsAb. In these HBsAg negative subjects, HBV-DNA is detectable in hepatic tissue, although it may be absent in serum. This condition is defined as “occult” HBV infection [62]. This term underlines the circumstance that this microorganism has not been completely eradicated, but it persists in the infected host. The biological and clinical significance of this serum and tissue pattern “in vivo” is uncertain and it has not yet been definitively understood. Therefore, presence of both antigen/antibody profiles (HBsAg-/HBcAb+/HBsAband HBsAg-/HBcAb+/HBsAb+) might be associated with a significant increased probability to develop pancreatic cancer, even if both viral profiles would mark the complete resolution of HBV infection. Unfortunately, to date, only 3 studies investigated this potential relationship. Therefore, no definitive conclusions may be drawn.
d) It is unclear whether some HBV or HCV genotypes may be associated with an increased risk of developing pancreatic cancer in comparison to different genotypes, according also with difference in their geographical distribution.
e) It has not be definitively explained whether some risk factors for pancreatic cancer, such as smoking status, history of diabetes and alcohol use, in association with HBV and/or HCV infection, may increase the risk to develop this type of malignancy.
In conclusion, new interesting insights are emerging from epidemiological studies, assessing the possible relationship between HBV/HCV infection and risk of pancreatic cancer development. However, several problems and uncertain points remain and they have to be accurately investigated and explained. Therefore, the planning of well-designed studies, focusing on populations of different regions in the world and enrolling a larger number of patients, is indispensable. This approach may help to establish the real impact of both viruses in pancreatic carcinogenesis and eventually define pathogenetic mechanisms involved in this process.
The Authors thanks Dr. Simonetta Righi (Integrate Library, S. Orsola- Malpighi Hospital, “Alma Mater Studiorum” - University of Bologna, Bologna, Italy) for her support in the search of scientific bibliography
None declared