Keywords
Heavy metals; Water; Sediments; Oreochromi; Sewage water
Introduction
Water contamination caused when an input from human activities cause an increase of a substance in fresh water, sediments and organisms above the natural background level for that area and for those organisms [1]. Occurrence of potential toxicants in aquatic ecosystem causes a reduction in the quality of the aquatic environment that results in impaired level of dissolved oxygen (DO), pH, temperature, biological oxygen demand, and chemical oxygen demand [2]. Though chemical monitoring of water and sediment is a common and reliable measure to describe the degree of contamination, it is not the case for the overall assessment for evaluating the effects of pollution on the environment as toxic or biological effects on organisms cannot be obtained by this method.
Organisms in aquatic environments are usually exposed to a complex mixture of chemicals causing multiple damages at the organisms, population and ecosystem level, in organ function [3,4]. Different compounds in polluted water are reported to damage the genetic material of exposed organisms and therefore cause genotoxic effects. The toxicity of these elements is due to their ability to cause, oxidative damage to living tissues [5]. Heavy metal toxicity includes the binding of heavy metals more strongly to functional sites that are normally occupied by essential functional groups of biologically important molecules thus disrupting the integrity of entire cells and their membranes, making them inactive, decomposing essential metabolites and changing the osmotic balance around the cells [6]. The degeneration and malformation (morphological alterations) of the cells reflect one aspect of the cytotoxic impacts after exposure to pollutants and serve as an index of cytotoxicity [7-9].
Metals were of particular concern due to their toxicity and ability to bio-accumulate in aquatic ecosystems [10] as well as persistency in the natural environment. Recently, different types of biomarkers have been studied and evaluated for their acceptability to detect the biological effects as a biomonitoring tool [11-13]. Biomonitoring, a promising tool for identifying pollutants (bio-indicators) that affect human and environmental health, is especially useful with organisms, thus exposed in biological systems (biomarkers) [14].
In Egypt, the problems of the drainage canals have extremely increased in the past years [15] where disposing of partially treated or untreated domestic and industrial wastewater into agricultural drains deteriorates their water quality [16]. El-Shikh pond in Elkharja city, New Valley lies between latitudes 25° 41’ N to 25.43N and longitudes 30° 56’ E to 30° 57’ E. It is receiving agricultural and domestic wastes without purification in addition to sewage of El-kharja city.
The present study aimed to evaluate the physicochemical characteristics of water, sediments and determination of heavy metals in different tissues Oreochromis niloticus from El-Shikh Pond.
Materials and Method
Study area
The present study was extended from April to July 2014 and carried out in El-shikh Pond, which located in El-kharja city, New Valley governorate, Egypt lies between latitudes 25° 41’ N to 25.43N and longitudes 30° 56’ E to 30° 57’ E which extends for 4.33 Km. This drain receives untreated domestic sewage from numerous villages in addition to the agricultural and industrial wastes (Figures 1A and 1B). The renewal of the water in the pond is very low. The pond fauna exhibits low diversity, with Oreochromis niloticus as the dominant species.
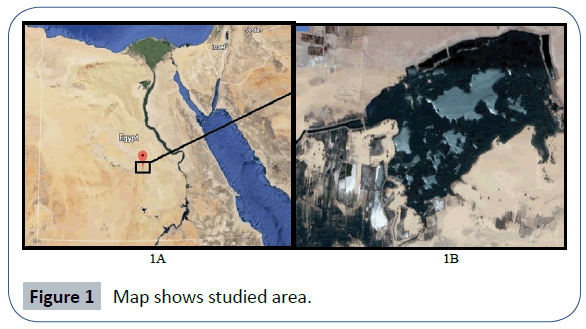
Figure 1 Map shows studied area.
• Samples collection: 150 specimens of Oreochromis niloticus were collected (from April to July 2014). The wet weight and total body length of the fish were measured, blood sampling was done.
• Sample Preparation: Fish were transported to the laboratory for blood smears analysis. Ten water samples and sediment were collected and transported to the laboratory for water quality analysis.
• The physicochemical characteristics
Some physicochemical characters as temperature, pH, Conductivity and turbidity were measured by using water checker U-10 Horiba Ltd at the same time of sample collection. Other characters such as Dissolved oxygen, Biological Absorbed oxygen, Chemical consumed oxygen, Total dissolved Solids, Nitrate, Total nitrogen; Total alkalinity and Organic carbon were measured by traditional manual methods [17,18].
• Determination of heavy metals: Sampling water, sediments and fish samples were collected from the studied sites during summer 2014.
Water sampling: Duplicate water samples were taken with a water sampler from five localities in the studied area between 10:00 and 12:00 a.m. at a depth of 30 cm below the water surface and stored at 4 ?C in a clean 1000 ml sampling glass bottles [19].
Sediment sampling: Duplicates of five core samples of sediment up to 20 cm in length were taken from selected area with polyvinyl chloride (PVC) corers [20]. The corers were immediately sealed and stored at 4°C.
Fish sampling: fish were collected from the same area where water and sediment samples were collected. Then stored frozen for further investigation. Fish gills, liver, kidney, muscles, skin and gonads were minced and well mixed, then approximately 1.0 g was placed in a 150 ml beaker and 10 ml concentrated nitric acid was added. The mixture was then heated until the appearance of heavy metals in different dense white fumes that indicate the nitric acid had evaporated. The mixture was cooled, then 10 ml of 25% hydrochloric acid was added and the solution was transferred to 250 ml volumetric flasks that were subsequently brought to volume with deionized water.
Each sample of water was put in screw-capped tube till complete dryness. Ten ml of concentrated nitric acid was added to the sample to dryness, and then diluted to 20 ml with deionized water. The solution was filtrated, transferred to 250 ml volumetric flask, marked and stored refrigerated till analysis. For the digestion of sediment samples, 1-2 g wet weight is digested with repeated additions of nitric acid and hydrogen peroxide, the resultant digested is reduced in volume while heating and then diluted to a final volume of 250 ml. All metals concentrations in the samples were determined by inductively coupled plasma mass spectrometry (ICP-MS) (Thermo Fisher Scientific, Bremen, GmbH).
Ethical Statement
All experiments were carried out in accordance with Egyptian laws and University guidelines for the care of experimental animals. All procedures of the current experiment have been approved by the Committee of the Faculty of Science (New Valley branch) of Assiut University, Egypt.
Statistical Analysis
The basic statistics of the measured parameters were estimated. The patterns of variation due to different tissues were tested by using one-way ANOVA. The differences between means were done by using The Tukey-HSD test. Range test was used as a post hoc test to compare between means at p>0.05. The software SPSS, version10 (SPSS, 1998) was used.
Results
Water quality: Physical-chemical parameters
The increased metals level in water resources caused by agricultural and domestic wastes are one of the main indicators of chemical contamination. So the water physical-chemical parameters shown in Table 1. The physicochemical characteristic of water in selected area recorded in Table 1 during the period of investigation (April to July 2014). The negative impact of different sources of pollutants (sewage and agricultures) discharged into the selected area; water confirmed by conductivity, turbidity, dissolved oxygen, nitrogen and nitrate content in El-Shikh pond as shown in table 1. Except temperature, pH, Nitrates, DO, BOD and COD in the studied area, the physical character of water was above the permissible limits of EOS (1993). Metals concentration in water following trend: Fe>Al>Mn>Ni>Zn>Pb> Cr>Se>Cu>Co>Cd. Also, the concentration of Al, Fe, Mn, Ni and Pb were above the permissible limits (Table 1) recommended by EOS (1993), and WHO (2008) referring to bad condition of the aquatic ecosystems studied. Iron then Aluminum showed the highest concentration of metals in sediments, followed by Ni>Mn>Cr>Cu>Zn>Pb>Cd however, Cobalt the Selenium showed the lowest one (Table 2).
| Parameters |
Unit |
Results |
*Guide Line |
| * PHYSICOCHEMICAL PARAMETERS: |
| Temperature |
0C |
25.5 ± 4.25 |
35.0 |
| Electric Conductivity |
µS/Cm |
381.30 ± 5.09 |
|
| Turbidity |
NTU |
3.51 ± 0.45* |
1.00 |
| pH |
|
8.33 ± 1.25 |
6.5-8.5 |
| Total Hardness as (CaCO3) |
Mg/l |
1347 ± 29* |
500 |
| Calcium Hardness |
Mg/l |
1210 ± 33* |
350 |
| Magnesium Hardness |
Mg/l |
540+21* |
150 |
| Magnesium Mg+2 |
Mg/l |
129+33 |
|
| Ammonia as (N) |
Mg/l |
1.99+0.21* |
0.40 |
| Nitrites as (N) |
Mg/l |
0.23+0.03* |
0.06 |
| Nitrates as (N) |
Mg/l |
1.06+0.41 |
10 |
| Fluoride |
Mg/l |
0.62+0.01 |
0.8 |
| Cyanide (CN- ) |
Mg/l |
Nil |
0.05 |
| Sodium |
Mg/l |
159+23 |
200 |
| Potassium |
Mg/l |
28.9+11.2 |
|
| Calcium Ca+2 |
Ppm |
61.12 ± 6.20 |
|
| * Organic Chemical Parameters |
| Dissolved Oxygen (D.O) |
mg/l |
4.36+0.31 |
≥ 4 |
|
| Bio Chemical Oxygen Diamond (BOD5) |
mg/l |
12.8+2.11 |
60 |
|
| Chemical Oxygen Diamond (COD) |
mg/l |
32+3.1 |
80 |
|
| * Heavy Metals |
EOS, 1993 |
WHO, 2008 |
| Cadmium(Cd) |
Ppm |
0.003 ± 0.0004# |
0.01 |
0.003 |
| Chromium(Cr) |
Ppm |
0.20 ± 0.01 |
1.0 |
0.50 |
| Lead(Pb) |
Ppm |
0.33 ± 0.14*# |
0.10 |
0.01 |
| Selenium(Se) |
Ppm |
0.09 ± 0.02 |
0.40 |
0.20 |
| Cobalt(Co) |
Ppm |
0.01 ± 0.002 |
0.20 |
0.20 |
| Nickle(Ni) |
Ppm |
0.82 ± 0.03*# |
0.10 |
0.10 |
| Copper(Cu) |
Ppm |
0.08 ± 0.01 |
1.0 |
1.00 |
| Zinc(Zn) |
Ppm |
0.42 ± 0.06 |
5.0 |
3.00 |
| Iron(Fe) |
Ppm |
9.06 ± 0.86*# |
0.30 |
0.30 |
| Aluminum (Al) |
Ppm |
7.94 ± 1.83*# |
3.0 |
1.00 |
| Manganese(Mn) |
Ppm |
0.90 ± 0.24*# |
0.10 |
0.10 |
Table 1: Some physicochemical characteristics of sewage water of El-Shikh pond at Elkharja, New Valley; presented data are means ± SE of measurements undertaken during April-July 2014.
| * Heavy Metals |
Cd |
Cr |
Pb |
Se |
Co |
Ni |
Cu |
Zn |
Fe |
Al |
Mn |
2.04 ±
0.02 |
4.85 ±
0.05 |
3.43 ±
0.05 |
1.50 ±
0.06 |
0.25 ±
0.01 |
150.2 ±
7.3 |
4.17 ±
0.02 |
3.57 ±
0.03 |
175.1 ±
20.8 |
173.9 ±
3.8 |
124.4
± 5.2 |
Table 2: Metals concentration (ppm) of sewage Sediments of El-Shekh pond at Elkharja, New Valley; presented data are means ± SE of measurements undertaken during April-July 2014.
Estimation of heavy metal residues in fish organs exhibited different patterns of heavy metal accumulations, some above and other under the permissible limits (Table 3) reflecting fish history, organ specificity, and variability of aquatic ecosystem status. Liver showed a highly significant increase in Fe, Ni, Co, Cu, Zn, Se and Cd as a target organ. However, Al and Mn accumulated significantly in kidney (Table 3). So, the highest concentration of metals recorded in liver while muscles recorded the lowest concentration of metal. The accumulation pattern of metals in different tissue recorded as Table 3. This study showed that Al (66.42 ± 3.26 ppm) and Fe (38.53 ± 1.52 ppm) were above the permissible limits that recommended by EOS (1993) and WHO (2008).
> > > > >
| Tissues |
G |
INT |
K |
L |
M |
OV |
SK |
T |
EOS, 1993 |
WHO, 2008 |
| Al |
63.87 ± 12.36*#B |
63.59 ± 3.80*#B |
92.54 ± 7.73*#D |
42.29 ± 5.77#A |
66.42 ± 3.26*#B |
73.07 ± 4.27*#C |
69.12 ± 4.10*#BC |
70.84 ± 5.46*#C |
50.0 |
30.0 |
| K>OV>T>SK>M>G>INT>L |
| Cr |
1.14 ± 0.09A |
1.75 ± 0.07D |
1.55 ± 0.08C |
1.31 ± 0.15B |
1.98 ± 0.10E |
1.74 ± 0.07D |
1.86 ± 0.09DE |
1.72 ± 0.09D |
20.0 |
10.0 |
| M>SK>INT>OV>T>K>L>G |
| Mn |
2.53 ± 0.29CD |
2.71 ± 0.38D |
11.37 ± 1.99*#F |
3.67 ± 0.75E |
1.47 ± 0.07A |
2.26 ± 0.12C |
1.62 ± 0.09B |
2.28 ± 0.16C |
10.0 |
10.0 |
| K>L>INT>G>T>OV>SK>M |
| Fe |
56.48 ± 4.48*#C |
117.50 ± 19.50*#D |
154.33 ± 27.10*# |
158.17 ± 38.59*#E |
38.53 ± 1.52*#A |
144.83 ± 8.57*#E |
46.91 ± 2.35*#B |
44.79 ± 2.76*#B |
30.0 |
30.0 |
| L>K>OV>INT>G>SK>T>M |
| Co |
0.05 ± 0.007C |
0.15 ± 0.02E |
0.19 ± 0.03E |
0.25 ± 0.04F |
0.03 ± 0.001A |
0.25 ± 0.02F |
0.04 ± 0.002B |
0.10 ± 0.007D |
10.0 |
1.0 |
| L=OV>K>INT>T>G>SK>M |
| Ni |
0.72 ± 0.05A |
1.14 ± 0.10C |
1.33 ± 0.17CD |
1.41 ± 0.19D |
0.91 ± 0.03B |
0.90 ± 0.04B |
0.89 ± 0.04B |
0.85 ± 0.04B |
10.0 |
2.0 |
| L>K>INT>M>OV>SK>T>G |
| Cu |
0.57 ± 0.13D |
0.95 ± 0.16F |
0.85 ± 0.18EF |
1.49 ± 0.36G |
0.30 ± 0.01A |
0.38 ± 0.01C |
0.33 ± 0.01B |
0.75 ± 0.19E |
20.0 |
20.0 |
| L>INT>K>T>G>OV>SK>M |
| Zn |
5.43 ± 0.85B |
9.58 ± 1.05E |
11.17 ± 2.04F |
16.41 ± 1.83G |
4.38 ± 0.17A |
6.71 ± 0.28C |
14.55 ± 0.98G |
8.06 ± 0.91D |
40.0 |
40.0 |
| L>SK>K>INT>T>OV>G>M |
| Se |
4.92 ± 1.42D |
14.80 ± 4.86E |
23.38 ± 8.29F |
47.59 ± 14.05G |
1.11 ± 0.07A |
1.52 ± 0.07B |
1.53 ± 0.09B |
2.50 ± 0.62C |
50.0 |
50.0 |
| L>k>INT>G>T>SK>OV>M |
| Cd |
0.03 ± 0.008C |
0.09 ± 0.03#D |
0.17 ± 0.05#E |
0.20 ± 0.05#E |
0.01 ± 0.001A |
0.01 ± 0.001A |
0.02 ± 0.001B |
0.03 ± 0.008C |
0.50 |
0.05 |
| L>K>INT>G>T>SK>OV>M |
| Pb |
0.73 ± 0.08#A |
1.67 ± 0.12#D |
1.42 ± 0.21#C |
1.24 ± 0.14#B |
1.89 ± 0.18#EF |
1.50 ± 0.17#CD |
1.44 ± 0.18#C |
1.73 ± 0.18#E |
2.0 |
0.05 |
| M>T>INT>OV>SK>K>L>G |
(*) Above the permissible limits of EOS (1993) (#) Above the permissible limits of WHO (2008)
Different letters indicate significance at 0.05.
Table 3: Metals concentration (ppm) in different Oreochromis niloticus organs (G: Gills, INT: Intestine, K: Kidney, L: Liver, M: Muscle, OV: Ovary, Sk: Skin and T: Testis) that collected from El-Shikh pond at Elkharja, New Valley; presented data are (means ± SE) of measurements undertaken during April-July 2014.
Discussion
Increases use of metal based fertilizers in agricultural revolution could result in the continued rises in the concentration of metal pollutants in fresh water due to the water run-off [21,22]. In this respect, [23] postulated that the bioaccumulation of heavy metal in fish body did not follow the same order of abundance in the ambient water. Moreover, Kock and Hofer [23] reported that even low concentration of heavy metal in water may result in high concentrations in fish flesh, which often collects the lowest percent of such metals.
Because of its great impact on aquatic life, water temperature is an important component of a water quality assessment [22]. Temperatures affect the ability of aquatic organisms to grow, reproduce, escape predators, and compete for habitat [24, 25]. The High levels of heavy metals during spring and summer could be attributed to the changes associated with higher water temperatures, which can cause higher activity and ventilation rates in fish [26].
Our results showed that the physicochemical characteristics of water in the selected area were recorded at intervals (Table 1) during the period of investigation. The concentration of Al, Fe, Mn, Ni and Pb were above the permissible limits (Table 1) recommended by EOS (1993), and WHO (2008) referring to bad condition of the aquatic ecosystems studied. Iron then Aluminum showed the highest concentration of metals in sediments, on the other hand, Cobalt the Selenium showed the lowest one (Table 2).
Aquatic organisms are exposed to contaminants through direct uptake from the water phase, indirect uptake through food or both. The direct uptake is influenced by the total concentration and the bioavailability of the contaminant, as well as by the physiological factors of the individual organisms [27]. The bioavailability and subsequent bioaccumulation of metals is related to the chemical specification of the elements, which is influenced by the physicochemical conditions of the environment (e.g., pH, conductivity, dissolved oxygen) which may be altered by the input of wastewaters [28].
Similar to our results, Omar [29] reported that high concentration of heavy metals studied in sediment samples result from precipitation of these metals from the water column under slightly elevated pH conditions recorded in the area, and from the adsorption of heavy metals onto organic matter and their settlement downwards.
Our results showed that kidney showed the highest concentration of Al and Mn. Similarly, Authman et al. (2012) and Ibrahim (2014) found that kidney of O. niloticus contained the highest concentration of Al+3 and Mn+2 respectively.
It can be noticed that the highest bioaccumulation were observed in the organs, mainly implicated in metal metabolism. High contaminated fish may cause health risks to human. The significant increases of Cu, Cd, Zn, Fe, Ni and Se in our results where confirmed by Kaoud and El-Dahshan [30] who found that the concentration of Cd and Cu in tissues was high in the liver. Also, the high accumulation of Zn in fish liver agrees with [31,32]. In addition, Ibrahim (2014) reported that the liver was the target organ for Fe and Ni in Oreochromis niloticus.
On the other hand, the lowest levels of these metals and minerals were found in the muscle compared with other tissues and organs. Similar results have been reported in Oreochromis niloticus [30] and other fish species [33-35]. The reason for this may be that muscle tissue is less active compared with other tissue samples and organs of fish. Muscle is the major tissue of interest under routine monitoring of metal contamination because it is consumed by people. Skin and muscle samples contained low concentrations of metals. It was shown that metal accumulation in fish tissues or organs depends on their physiological role. For instance, dermal route is usually a minimal contributor of exposure, due to the often effective barrier provided by the external epithelium [36]. Numerous recent studies have confirmed that muscle and skin in fish have many lower concentrations of metals than liver, kidney, gills and bones [37,38].
Conclusion
Pollution with metals in the study area, as reflected by the high metal concentrations recorded in water and sediment samples, because of the continuous discharge to the aquatic habitats. The concentrations of heavy metals reach to a dangerous level that affecting the health of local human communities. Therefore, this study strongly recommends the coordination of different efforts to Prohibition to fish from this area and rescuing the polluted habitats from serious ecological problems using proper management.
References
- Clark RB (2001) Metals. In: Marine pollution.
- Roberts RJ (2001) Fish Pathology. Philadelphia: Saunders WB.
- Vorosmarty CJ, McIntyre PB, Gessner MO, Dudgeon D, Prusevich A (2010) Global threats to human water security and river biodiversity. Nature 468: 334.
- Ginebreda A, Kuzmanovic M, Guasch H, López de Alda M, Lopez-Doval JC (2014) Assessment of multi-chemical pollution in aquatic ecosystems using toxic units: Compound prioritization, mixture characterization and relationships with biological descriptors. Sci Total Environ 468/469:715-723.
- Bakar O, Demirçay Z, Yuksel M, Haklar G, Sanisoglu Y (2007) The effect of azithromycin on reactive oxygen species in rosacea. ClinExpDermatol32:197-200.
- Jjemba PK (2004) Environmental Microbiology. Principles and Applications. Enfield New Hampshire: Science Publishers Inc.
- Harabawy ASA, Ibrahim ATA (2014)Sublethaltoxicity of carbofuranpesticideonthe African catfish Clariasgariepinus (Burchell,1822):Hematological, biochemical and cytogenetic response. Ecotoxicol Environ Saf.
- Harabawy ASA, Mosleh YYI (2014) The role of vitamins A, C, E and selenium as antioxidants against genotoxicity and cytotoxicity of cadmium, copper, lead and zinc on erythrocytes of Nile tilapia, Oreochromis niloticus. Ecotoxicology and Environmental Safety. 104:28-35.
- Mahboob S, Al-Balwai HFA, Al-Misned F, Ahmad Z (2014)Investigationonthegenotoxicity of mercuric chloride to freshwater Clariasgariepinus. PakVetJ 34:100-103.
- Mohammadi M, Sary AA, Khodadadi M (2011) Determination of heavy metals in two barbs, Barbusgrypus and Barbusxanthopterus in KaroonandDezRivers, Khoozestan, Iran. Bull Environ ContamToxicol87:158-162.
- Amiard JC, Amiard-Triquet C, Barka S, Pellerin J, Rainbow PS (2006)Metallothioneins in aquatic invertebrates: their role in metal detoxification and their use as biomarkers. Aquat Toxicol 76:160-202.
- Magni P, De Falco G, Falugi C, Franzoni M, Monteverde M, et al. (2006)Genotoxicity biomarkers and acetylcholinesterase activity in natural populations of Mytilusgalloprovincialis along a pollution gradient in the Gulf of Oristano (Sardinia, western Mediterranean). Environ Poll 142:65-72.
- Nigro M, Falleni A, Del Barga I, Scarcelli V, Lucchesi P, et al. (2006) Cellular biomarkers for monitoring estuarine environments: transplanted versus native mussels. Aquat Toxicol 77:339-347.
- Silva J, Heuser V, Andrade V (2003)Biomonitoramentoambiental. In: Silva J EBaHJ, editor. GenéticaToxicológica. Alcance, Porto Alegre 167-178.
- Authman MMN, Abbas HH, Abbas WT (2013) Assessment of metal status in drainage canal water and their bioaccumulation in Oreochromis niloticus fish in relation to human health Environmental Monitoring and Assessment 185:891-907.
- El-Sheikh MA, Saleh HI, El-Quosy DE, Mahmoud AA (2010) Improving water quality in polluted drains with free water surface constructed Wetlands. Ecological Engineering36: 1478-1484.
- Eaton AD, Clesceri LS, Greenberg AR (1995) Standard methods for the examination of water and wastewater. Washington, DC.
- US EPA (1983) Methods for Chemical Analysis of Water and Wastes. In: Laboratory EMaS, editor. Office of Research and Development, US Environmental Protection Agency. Cincinnati, OH.
- AbouElnaga WM, Allam SM (1996) Heavy metal concentrations in the tissues of Tilapia zilliiGerv, exposed to waste water discharge of Egyptian copper factory. Comparative physiology 19: 21-35.
- Cabrera F, Conde B, Flores V (1992) Heavy metals in the surface sediments of the tidal river Tinto (SW Spain). Fresenius Environ Bull 1: 400-405.
- Adefemi SO, Asaolu SS, Olaofe O (2008) Determination of Heavy metals in Tilapia mossambicusFish,Associated water and Sediment fom Ureje Dam in South Western, Nigeria. Research Jour of Environ Sci 2:151-155.
- Prasath PMD, Arivoli S (2008) Biochemical study of freshwater fish Catla catla with reference to mercury chloride. Iranian Jour Environ Health Sci Eng 3:109-116.
- Boyd CE (1990) Water quality in ponds for aquaculture. Alabama Agriculture Experiment Station,. Auburn Univ, Alabama, U. S. A.
- Chapman D, Chapman DE (1996) Water Quality Assessments A Guide to the Use of Biota, Sediments and Water in Environmental Monitoring. Chapman & Hall, London.
- Kazlauskiene N, Svecevicius G, Marciulioniene D, Montvydiene D, Kesminas V, et al. (2012) The Effect of Persistent Pollutants on Aquatic Ecosystem. Baltic International Symposium (BALTIC) 8-10: 1-6.
- Osman AGM, Abd El Reheem AMA, AbuelFadl K, GadEl-Rab A (2010) Enzymatic and Histopathologic Biomarkers as Indicators of Aquatic Pollution in Fishes. Natural Science 2:1302-1311.
- Obasohan EE, Eguaveon OI (2008) Seasonal Variations of Bioaccumulation of heavy metals in a freshwater fish (Erpetoichthyscalabaricus) from Ogba River, Benin City, Nigeria. Afr J Gen Agric4: 153-163.
- Sjöblom A, Meili M, Sundbom M (2000) The influence of humic substances on the speciation and bioavailability of dissolved mercury and methyl mercury, measured as uptake by Chaoborus larvae and loss by volatilization. The Science of the Total Environment 261:115?124.
- Kaoud HA, El-Dahshan AR (2010) Bioaccumulation and histopathological alterations of the heavy metals in Oreochromis niloticus fish. Nat Sci 2010 8:147?156.
- Kock G, Hofer R (1998) Origin of Cadmium and Lead in clear Soft Water Lakes of High-altitude and High-latitude, and their Bioavailability and toxicity to fish. J Exs87:225-257.
- Gagnon C, Saulnier I (2003) Distribution and fate of metals in the dispersion plume of a major municipal effluent. Environmental Pollution 124:47?55.
- El-Naggar SM, Tayel S (2009) Bioaccumulation of some heavy metals and histopathological alterations in liver of Oreochromis niloticus in relation to water quality at different localities along the River Nile, Egypt. World J Fish Mar Sci.
- Ibrahim ATA (2014) Seasonal variation of heavy metals concentrations in muscles of Oreochromis niloticus, River Nile water and sediments at Assiut Governorate, Egypt,Seventh International Conference on Environment and Development in the Arab World, Assiut, Egypt.
- Ibrahim ATA, Omar HM (2013) Seasonal variation of heavy metals accumulation in muscles of the African Catfish Clariasgariepinus and in River Nile water and sediments at Assiut Governorate, Egypt. J Biol Earth Sci3:B236-B48.
- Canpolat O (2013) The Determination of Some Heavy Metals and Minerals in the Tissues and Organs of the Capoetaumbla Fish Species in Relation to Body Size, Sex, and Age. Ekoloji22: 64-72.
- Kir I, Tumantozlu H (2012) Investigation of Some Heavy Metal Accumulation in Water, Sediment and Carp (Cyprinuscarpio) Samples of Karacaoren-II Dam Lake.Ekoloji21:65-70.
- Fernandes C, Fontainhas-Fernandes A, Peixoto F, Salgado MA (2007) Bioaccumulation of heavy metals in Liza saliens from the Esomriz-Paramos coastal lagoon, Portugal. Ecotoxcolog of Environ and Safety 66:426-431.
- Dsikowitzky L, Mengesha M, Dadebo E, Eduardo C, Calvalho VD, et al. (2012) Assessment of heavy metals in water samples and in fish tissues of edible fish species from Awassa and Koka Rift Valley Lakes, Ethiopia. . Environ Monit Assess 185:3117-3131.