- (2007) Volume 8, Issue 5
Ashwinikumar Kudari1, Jai Dev Wig1, Kim Vaiphei2, Rakesh Kochhar3, Siddarth Majumdar4, Rajesh Gupta1, Thakur Deen Yadav1, Rudra Prasad Doley1
Departments of 1General Surgery, 2Pathology, 3Gastroenterology and 4Experimental Medicine; Postgraduate Institute of Medical Education and Research. Chandigarh, India
Received February 18th, 2007 - Accepted June 21st, 2007
Context Experimental models of acute pancreatitis have been developed in order to understand its pathophysiology and extrapancreatic manifestations. Objective The objective of our study was to study sequential changes in the pancreas and distant organs in sodium taurocholate-induced acute pancreatitis in a rat model. Animals Sixteen male Wistar rats weighing 250-300 g. Design The rats were distributed into two groups: induced acute pancreatitis (study group: 8 rats) and a control group (8 rats). Within each group, the animals were divided into subgroups: those who were sacrificed early (24 h and 72 h; two each) and those who were sacrificed late (120 h and 240 h; two each). Intervention Acute pancreatitis was induced in the rats by multiple intraparenchymal injections of 10% sodium taurocholate solution. In the controls, the same amount of normal saline was injected into the pancreatic parenchyma. Main outcome measures Pathological examination of the pancreas, lungs, kidneys, intestine and liver was done. Results In this model of taurocholate-induced acute pancreatitis, the early changes observed in the pancreas were focal hemorrhages, parenchymal necrosis and neutrophil infiltration. At 72 hours, the changes observed were acinar necrosis, edema, fibrin deposition and inflammatory cell infiltration. Late changes were fibrinoid necrosis and fibroblast proliferation. In the acute phase, the histological changes in the lungs were congestion, focal pulmonary edema and intraalveolar hemorrhages while, in the late stage, there was persistence of vascular congestion. The changes observed in the kidneys were vacuolization of tubular epithelium in the subcapsular region and areas of hemorrhage in the interstitium. Intestinal changes included degenerative changes in the villous epithelium in the acute phase with normalization of the histology in the late phase. Conclusion Our findings correlate with the clinical observation of multisystem organ failure in acute pancreatitis. Early changes in these organs suggest that careful observation is mandatory in patients with acute pancreatitis in order to institute supportive treatment.
Inflammation; Kidney Diseases; Necrosis; Pancreas; Pancreatitis, Acute Necrotizing; Rats, Wistar; Respiration Disorders; Taurocholic Acid
Various experimental models of acute pancreatitis have been developed in order to understand its pathophysiology, histological features, degree of severity and failure of distant organ systems notably the lungs, kidneys, liver and intestine [1, 2, 3, 4, 5, 6]. Novel therapies targeted towards the recently discovered intracellular events need to be developed and tested using experimental models [2, 5, 6]. Chan and Leung [1] have summarized the most prevalent animal models of acute pancreatitis and their advantages and limitations. Pandol et al. [3] have also shown pancreatic inflammation, apoptosis, necrosis (mild to severe) and systemic inflammation in models of experimental acute pancreatitis. However, there are significant differences in the pathophysiological response among species and models. Various animal models have been described and the selection of a particular model depends upon the specific aims of the experiment. The course of pancreatitis in terms of the histological changes which take place during the course of the disease will help in identifying the relationship between the up/down regulation of cellular factors and the degree of the injury of the pancreas in experimental models. The time taken for changes to occur in the lungs, kidneys, intestine and liver will suggest the intensity of the inflammatory reaction and may help the clinician in supporting various systems in the early stages of acute pancreatitis. The aim of our study was to study the sequential changes in the various organs in sodium taurocholate-induced acute pancreatitis.
The study was carried out between the period of July 2005 and December 2006. The study included sixteen male Wistar rats weighing between 250-300 grams. They were distributed into two groups: an induced acute pancreatitis group (study group: 8 rats) and a control group (8 rats). Within each group, the animals were divided into subgroups: those who were sacrificed early (24 h and 72 h; two each) and those who were sacrificed late (120 h and 240 h; two each).
Operative Procedure
The acute pancreatitis was induced in the operating theatre of the Department of Experimental Medicine and Biotechnology. General anesthesia was induced by an intraperitoneal injection of 60 mg/kg of phenobarbital. The abdomen was then shaved and prepared with a 5% povidone-iodine solution. A midline incision was made. The pancreatitis was induced by multiple intraparenchymal injections of 0.3 mL/100 g body weight of 10% sodium taurocholate solution through a 30-gauge needle. In the controls, the same amount of normal saline was injected into the pancreatic parenchyma in a similar fashion. After the injection, the organs were carefully returned to the peritoneal cavity, and the incision was closed with a continuous 3-0 silk suture. The animals were left to recover for one hour and were then put back into metallic cages, one rat per cage, and allowed unlimited access to standard rat chow.
Pathological Examination
The tissue specimens were examined at the Department of Histopathology, by an expert pathologist (K.V.). The pancreas was harvested and trimmed of fat. Its macroscopic appearance was recorded and the gland was fixed in 10% formaldehyde, processed in paraffin, and stained with hematoxylin and eosin. The injury was scored by recording the number of lobes affected by edema, hemorrhage, leukocyte infiltration, parenchymal necrosis, fat necrosis and fibrosis. Each criterion was graded on a scale of 0-3 [7]. Edema and hemorrhage were assessed according to their location, and whether they were observed within intra- or interlobular areas. Leukocyte infiltration, parenchymal necrosis and fibrosis were evaluated according to the number of lobules involved. Fat necrosis, inflammation and fibroblastic proliferation were scored in the peripancreatic tissue. Pathological examination of pancreas, lungs, kidneys, intestine and liver were done after fixing them in 10% formaldehyde.
Biochemical Parameters
Serum amylase was measured at 24 hours by a reagent kit for the quantitative estimation of alpha-amylase in serum (Reckon Diagnostic Pvt Ltd, Baroda, India). The reference range of serum amylase was 35-140 IU/L as per the manufacturer’s instructions. Samples were collected from all the animals of both groups.
The experimental protocol was duly approved by the Ethical Committee of the Institute of Medical Education and Research (Chandigarh, India). All animals received humane care according to the criteria outlined in the "Guide for the Care and Use of Laboratory Animals (1996)" prepared by the National Academy of Sciences.
All continuous variables were expressed as mean and standard deviation, and were analyzed by means of the Mann-Whitney Utest. Median, range, and frequencies were also used as descriptive statistics. Statistical analysis was done using the SPSS version 12 software. Two-tailed P values less than 0.05 were considered significant.
Biochemical Parameters
The study group rats had a significantly higher (P=0.001) amylase level (mean±SD: 1,448±82 IU/L; median: 1,447 IU/L; range: 1,339-1,565 IU/L) than the control group (mean±SD: 550±84 IU/L; median: 556 IU/L; range: 402-650 IU/L).
Pathological Examination
In the control group, the injection of saline into the pancreatic parenchyma did not result in any macroscopic damage and all the abdominal organs looked normal. The animals in the study group showed severe morphological changes in the pancreas including focal hemorrhage and necrosis. In addition there was pronounced intestinal dilatation. The lungs, kidneys and liver looked normal on gross examination.
Histopathological Changes
Pancreas
The values of total histopathologic scores in the control and the test groups are shown in Table 1. The total histological score of the control group was significantly lower than t hat of study group (P<0.001). Control group. The histopathological examination of the pancreas in the control group was normal except for grade 1 edema in two out of eight rats (Table 1).
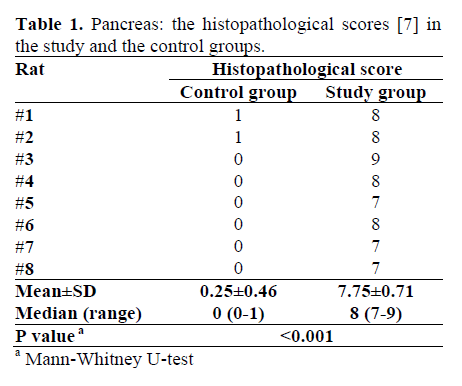
Study group. Early changes were a focal area of parenchymal necrosis and inflammation with neutrophil infiltration (Table 2). There were collections of lymphomononuclear cells in and around the pancreatic parenchyma with edema in the interlobular region. The animals sacrificed at 72 hours showed more extensive pathological changes, including acinar necrosis, peripancreatic fat necrosis, inter and intralobular edema, hemorrhage, fibrin deposition and inflammatory cell infiltration consisting of neutrophils, histiocytes and lymphocytes. The late changes were peripancreatic fat inflammation, fibroblastic proliferation, fibrinoid necrosis and focal patc hy persisting edema in the parenchyma. Fibrosis was seen extending focally into the pancreatic lobule with mild lymphocytic infiltration. At 240 hours, the changes tended to be more chronic with replacement of acini by chronic inflammatory cells and crowding of islets of Langerhans because of acinar atrophy. In addition there was proliferation of fibroblasts in the peripancreatic area along with lymphomononuclear cell infiltration (Figure 1).

Figure 1. Photomicrographs of the pancreas. a. Interstitial edema, with sparse inflammatory cell infiltration between the pancreatic acini (H&E, x150). b. Large areas of hemorrhage, interstitial edema and infiltration by inflammatory cells between the acini and the peripancreatic area (H&E, x55). c. Areas of acinar necrosis, interstitial edema, sparse inflammatory cells and hemorrhage (H&E, x150). d. Fibroblastic proliferation, inflammation, hemorrhage and fibrin deposition (H&E, x150).
Lungs
Control group. The histopathological examination of the lungs was normal in all rats of the control group. Study group. Early changes in the study group were congestion, focal pulmonary edema and intra-alveolar hemorrhage. The histological changes at 72 hours were interstitial edema and lymphomononuclear cells in the interstitium with vascular congestion. The late change was the persistence of the vascular congestion with resolution of the other changes (Figure 2).
Kidneys Control group. Histopathological examination of the kidneys was normal in all rats of the control group. Study group. Early histopathological examination of the kidneys in the study group showed the presence of vacuolization of the tubular lining epithelium mainly in the subcapsular region and patchy areas of hemorrhage in the interstitium. Some of the tubules towards the medullary region showed necrosis of the tubular lining epithelium suggestive of early acute tubular necrosis (Figure 3). Kidneys which were studied in the late period did not show any histopathological abnormality.
Small and large intestines
Control group. The histopathological examination of the intestine was normal in all rats of the control group. Study group. Early histopathological examination of the small intestines in the study group showed the presence of degenerative changes in the epithelium lining the villi (Figure 4). Histopathological examination of the intestine in the late period did not sho w the changes. On histopathological examination, the colon was essentially within the normal limits both in the early and the late groups.
Liver
The histopathological examination of the liver was normal in all the study and control animals.
Experimental models of acute pancreatitis resembling the human situation are an integral tool for increasing the understanding of complex mechanisms as well as for developing therapeutic strategies for this disease. Nevalainen and Aho [8] showed that edema is the common denominator of pancreatitis induced by any method and reflects inflammatory mechanisms. Parenchymal necrosis can be peripheral or centrilobular. Non-specific polymorphonuclear leukocytic infiltration appears first, later shifting towards mononuclear lymphoplasmacytic and macrophagic types, but polymorphs persist. Fat dissolution begins during the first few minutes after induction. The necrotizing process is followed by fibroblastic proliferation which is pronounced on the 3rd day of the disease. In our model, changes observed at 24 hours were focal areas of necrosis with neutrophil infiltration and edema in the interlobular region. At 72 hours, there was acinar necrosis and peripancreatic fat necrosis along with interlobular edema and more chronic inflammatory cell reaction. There was also the onset of fibrin deposition. Later, fibroblastic proliferation started and, by 10 days, crowding of islets of Langerhans also occurred along with more intense fibroblast activity. These responses are in accordance with the observations of other authors. Edema was found in all study animals and was the earliest change. It was followed by leukocytic infiltration and parenchymal and peripancreatic fat necrosis. Fibroblasic proliferation started after 72 hours.
In patients with acute pancreatitis, lung injury occurs in 60-70% and adult respiratory distress syndrome in 10-15% [9, 10]. The first sign of multiorgan dysfunction is impaired lung function. The lung injury is characterized by increased endothelial permeability, activation of the macrophages and the release of mediators, and activation and sequestration of neutrophils in the interstitium. Several rodent models of pancreatitis associated with lung injury suggest that activated neutrophils and the release of proinflammatory mediators after the activation of the inflammatory cells within pancreas play an important role in translating the pancreatic inflammation to the lungs [9, 10]. In our model of acute pancreatitis, histopathological examination of the lungs at 24 hours showed a marked accumulation of mononuclear cells and neutrophils which thickened the alveolar membrane. At 72 hours, histologic injury decreased with minimal changes at 240 hours. Few previous studies have investigated the early changes in pulmonary mechanics after the initiation of acute pancreatitis by taurocholic acid. Lichtenstein et al. [11] described alveolar edema at day 1 associated with neutrophil infiltration. Milani Júnior et al. found a significant increase in pulmonary elastance [12]. Lung sections showed an uneven distribution of ventilation, edema in the alveoli, and neutrophil infiltration. Two hours after bile acid injection, the alveoli were filled with fluid, macrophages, red blood cells, and cellular debris [12]. Enlargement of the alveolar septa was observed and severe endothelial changes were obvious with disintegration of type I epithelial cells, adherence of platelets to the capillary endothelium, and a loss of endothelial cell cytoplasm. An increase in pulmonary wet-todry ratio was found at one hour after induction of pancreatitis. Six hours after taurocholic acid administration, the infiltration of neutrophils was significantly increased, and the alveolar walls were enlarged.
Clinical data indicate a high frequency (from 37 to 60%) of renal involvement in acute pancreatitis [13]. But there is no unanimity regarding the explanation of the morphological nature of the lesions. Autopsy data in the literature do not rule out the possibility of obscuring the morphological picture due to the postmortem changes. The sequences of the morphological changes in the kidneys in acute pancreatitis have not been studied [13]. Acute renal failure of variable magnitude is now considered an important and frequent problem in acute pancreatitis. It is reported with a frequency as high as 22%, and the combination of acute renal failure and acute pancreatitis have mortality rates as high as 80% [14]. In their study of experimental acute pancreatitis, Levy et al. [14] have attributed the occurrence of renal failure to a decrease in intravascular volume. Zhang et al. [2] have attributed renal failure to the release of inflammatory mediators and the redistribution of the visceral blood flow, leading to a sharp decline in renal blood flow. There is also the development of granulocytosis and an accumulation of neutrophils in the microvasculature of the renal capillaries resulting in renal injury. Our observation of a lack of significant histological changes also supports the contention that vascular changes per se or in combination with the release of inflammatory mediators seem to be responsible for renal failure. In the early phase, there were areas of patchy necrosis of tubular lining epithelium suggestive of acute tubular necrosis close to the medullary area. Infectious complications in patients with severe acute pancreatitis are thought to result from bacterial translocation from the gastrointestinal tract. Such translocation is induced by the gastrointestinal derangement which may disrupt mucosal integrity, accompanied by an increase in the permeability of the gastrointestinal tract. However, the mechanism of increasing intestinal permeability in severe acute pancreatitis is still unknown. In their experimental study, Yasuda et al. [15] have reported an increase in intestinal permeability and subsequent bacterial/endotoxin translocation. They also demonstrated that the tight junction proteins (ZO-1 and occludin) were not changed but that apoptosis of the intestinal epithelial cells was accelerated. Pancreatitis-associated ascitic fluid reproduced the increased intestinal cell apoptosis and permeability, in vitro. These results suggest that the breakdown of the intestinal mucosa by accelerated apoptosis may result in intestinal permeability in severe acute pancreatitis and that pancreatitisassociated ascitic fluid contains factors which accelerate the apoptosis of intestinal epithelial cells [15]. In their study on intestinal permeability in patients with severe acute pancreatitis, Ammori et al. [16] have demonstrated that early increased intestinal permeability may play an important role in the pathophysiology of severe acute pancreatitis. Therapies which aim to restore the intestinal barrier function may improve outcome [16]. In our model histopathological examination of the intestine revealed the presence of degenerative changes in the epithelium lining the villi. The significance of this finding is not clear.
Clinical symptoms of liver damage are present in 18-30% patients with acute pancreatitis. Liver insufficiency accompanies pancreonecrosis in 25% of cases [17]. The incidence of liver failure is less than that of lung or kidney failure but is associated with high mortality. Liver failure could be due to the excessive activation of the hepatic inflammatory response. Pancreatogenic damage to the liver is characterized by hypoalbuminemia, hypofibrinogenemia, an increase in aminotransferase and lactate dehydrogenase activities and unconjugated bilirubin content in the blood. In their study of liver damage during experimental acute pancreatitis, Mozzhelin et al. [17] demonstrated that acute pancreatitis in rats caused by cooling of the pancreas with chloroethyl is characterized by the development of focal necrosis and the activation of lipid peroxidation in the liver, activation of blood aminotransferase and acid and alkaline phosphatase, and an increase in total and indirect bilirubin concentrations [17]. Our model did not show any histopathological changes in the liver. The limitations of our study are: a) the small number of animals in the study group; b) mediators of inflammation and apoptosis were not studied; and c) the contribution of microcirculatory disturbances in the pathogenesis of extrapancreatic organ damage has not been studied.
In conclusion, we observed that the process of pancreatic necrosis and inflammation progresses in the early phase (up to 72 h) which is then followed by fibroblastic proliferation and subsequent fibrosis which sets in by 10 days. Changes in the lung were vascular congestion, pulmonary edema and intra-alveolar hemorrhages in the acute phase, with only vascular congestion persisting in the late phase. Renal changes in the late phase were suggestive of patchy tubular necrosis which resolved itself rapidly. The intestine showed degenerative changes in the epithelium lining the villi in the acute phase reverting to normal by the late phase. The liver was not affected histologically in our experimental model. These findings correlate with the clinical observation of multisystem organ failure in acute pancreatitis. The early changes in these organs suggest that careful observation of potential organ failure is mandatory in patients with acute pancreatitis and early organ support may help in decreasing mortality.
The authors have no potential conflicts of interest