Keywords
Domperidone 10 mg, D2 receptor, Degradation studies, Assay.
Introduction
Chemical stability of pharmaceutical
molecules is a matter of great concern as it
affects the safety and efficacy of the drug
product. The FDA and ICH guidance’s state
the requirement of stability testing data to
understand how the quality of a drug
substance and drug product changes with
time under the influence of various
environmental factors Knowledge of the
stability of molecule helps in selecting
proper formulation and package as well as providing proper storage conditions and
shelf life, which is essential for regulatory
documentation. Forced degradation is a
process that involves degradation of drug
products and drug substances at conditions
more severe than accelerated conditions and
thus generates degradation products that can
be studied to determine the stability of the
molecule. The ICH guideline states that
stress testing is intended to identify the
likely degradation products which further helps in determination of the intrinsic
stability of the molecule and establishing
degradation pathways, and to validate the
stability indicating procedures used [1].
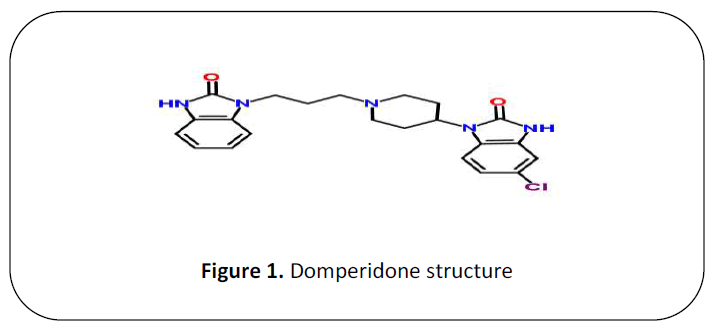
Domperidone (DOM), chemically
known as 5-Chloro-1-{1-[3-(2-oxo-2, 3-
dihydro-1H-benzimidazol-1-yl) propyl]-4-
piperidinyl}-1, 3-dihydro-2H-benzimidazol-
2-one (Figure 1) [2]. It is a selective D2 receptor antagonist, the D2Receptors in the
CTZ (Chemo Trigger Zone) and causes
prolactin release from the anterior pituitary.
It is official in British Pharmacopoea and
European Pharmacopoea [3-5]. It speeds
gastrointestinal peristalsis, and is used as
antiemetic for nausea or vomiting associated
with gastrointestinal disorders e.g.,
dyspepsia, heartburn, epigastric pain and
with cytotoxic and other drug treatments. It
does not enter the CNS (Blood Brain
Barrier) to a significant extent. Side effects
include galactorrhea, gynecomastia, or
menstrual irregularities and it may be 91%-
93% binds with protein [6,7].
Figure 1. Domperidone structure
Literature review reveals that some
of the UV, HPLC methods have been
reported for the simultaneous estimation of
Domperidone [5,8-14]. The aim of the study was
to perform forced degradation studies of the
different brands of domperidone under
hydrolytic (acidic and basic), and thermal
stress conditions, defined under ICH
guideline by using spectrophotometer. These
types of degradation studies of drugs and
these are very helpful for health care
professionals [15-18].
Methods
Reagents
Analytical grade reagents were used
1N sodium hydroxide, 1N hydrochloric acid,
de-mineralized water and distilled water.
Wavelength Selection
About 100 ppm of domperidone was
accurately prepared in distill water. The
wavelength maxima (λmax) was observed at
287 nm and this wavelength was adopted for
absorbance measurement.
Standard Stock Solution
The seven different brands were
purchased from a local medicine shop
located in Bayzid Bostami, Chittagong. All
tablets of brand have the same batch number
and were labeled to contain domperidone 10
mg per tablet. All the seven brands have 3
year shelf life. Weigh and finally crushed
tablets accurately for making primary
solutions of domperdone 10 mg, Motigut
(0.1878 gm) Square pharmaceuticals,
Domilux (0.0963 gm) Popular
pharmaceutical, Dysnov (0.1484 gm)
Unimed & Unihealth, Dominol (0.1119 gm)
Whitehorse pharma, Domar (0.2805 gm)
Pacific pharmaceuticals, Perion (0.1467 gm)
Globe Pharmaceutical, Doperon (0.1845
gm) United chemicals & pharmaceuticals
were weighed accurately and introduced in
100 ml volumetric flasks. Distill water was
added and shaken vigorously and make up
the volume up to 100 ml to make the
strength of the solution 100ppm in 100 ml.
For Acid
To study the effect of acid, 5 ml of
100 ppm solution of each brand was taken in
seven separated test tubes then 5ml of 1N
HCl was added in each test tube. They were
then left for a period of 60 minutes. Upon
completion of time period, solutions were
transferred to acuvette separately and then
absorbance of the solutions was recorded at
the wavelength of 287 nm.
For Base
To study the effect of base, 5 ml of
100 ppm solution of each brand was taken in
seven separated test tubes then 5 ml of 1N NaOH was added in each test tube. The
samples were then left for a period of 60
minutes. Upon completion of time period,
solutions were transferred to a cuvette
separately and then absorbance of the
solutions was recorded at the wavelength of
287 nm.
For Heat
To study the effect of heat, 5 ml of
100 ppm solution of each brand was taken in
seven separated test tubes each containing 5
ml of water, than place these solutions in
water bath for 60 min and absorbance of the
solutions was recorded at the wavelength of
287 nm.
Results and Discussion
This research was performed with the
purpose to compare the degree of degradation
in seven different brands of domperidone 10
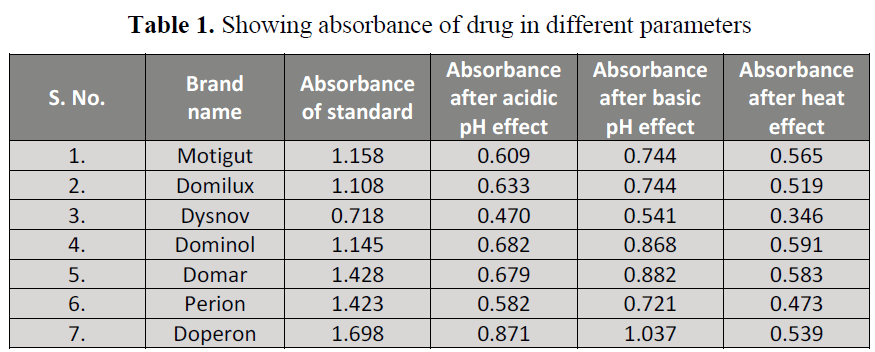
mg. Table 1 shows the variation in
absorbance after the effect of different
degradation parameters. The limit of assay by
USP specified that the content should not be
less than 95% and not more than 105% of
labeled amount. All brands were degraded in
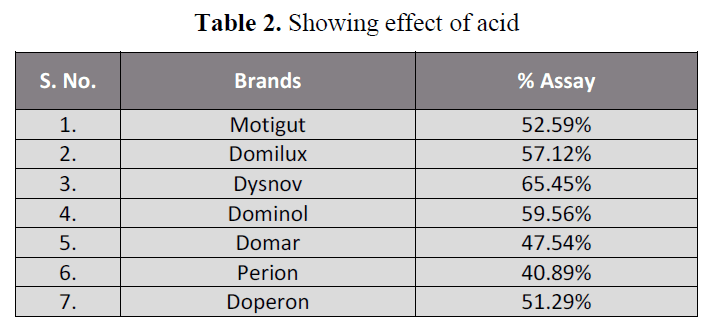
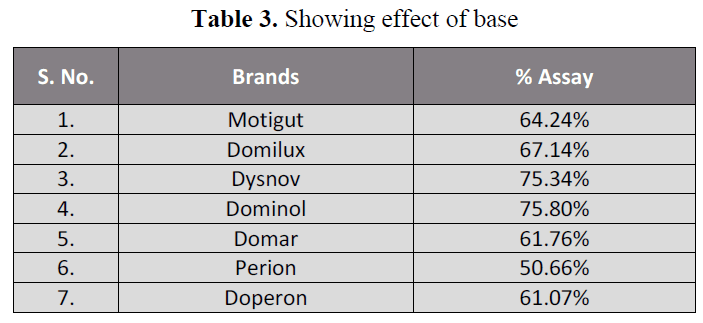
acidic and basic pH (table 2, 3) showing that
pH alteration has the most degradation impact
on these products. All brands was also
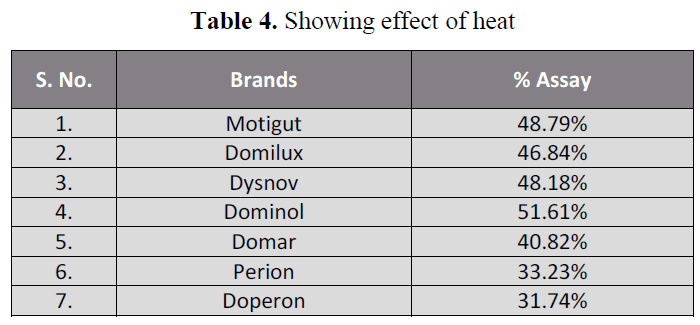
degraded after the heat exposure (table 4).
Table 1. Showing absorbance of drug in different parameters
Table 2. Showing effect of acid
Table 3. Showing effect of base
Table 4. Showing effect of heat
Conclusions
It was used to study the stressdegradation
studies as per ICH guidelines.
Domperidone was found to be degraded in
almost all types of stress conditions and was
found to be less stable. The method was
used is accurate and precise as well as
reproducible and economical and can be
successfully used degradation studies of
different dosage form. It was concluded that
all brands degraded from ranges for all the
stresses applied for degradation studies.
Acknowledgement
The authors greatly acknowledge to
Dr. Safila Naveed, Associate Professor,
Faculty of Pharmacy, Jinnah University for
Women, Karachi, Pakistan for her helpful
information and suggestions during the period
of laboratory work.
References
- Blessy M, Patel RD, Prajapati PN, Agrawal YK. Development of forced degradation and stability indicating studies of drugs. Review Paper. Journal of Pharma Analysis 2014; 4(3):159.
- Chemspider, Domperidone, https://www.chemspider.com/Chemical-Structure.3039.html, date accessed on 05/11/ 2014.
- European Pharmacopoeia. 3rd edition, Council of Europe Strasburg 1997:778- 780.
- British Pharmacopoeia, HMSO Publication, London; 1(2002); Through CD-ROM.
- Patel AH, Patel JK, Patel KN. Development and validation of derivative spectrophotometric method for simultaneous estimation of domperidone and rabeprazole sodium in bulk dosage forms. Int. J. Ph. Sci 2010; 2(1):464-469.
- Bennett and Brown, Clinical Pharmacology. 10th Edition, Churchill Livingstone, New York, U.S.A., 2008: 569.
- Pubchem, Domperidone, https:// pubchem.ncbi.nlm.nih.gov/compound/do mperidone#section=Top, date accessed on 05/11/2014.
- Sivakumar T, Manavalan R, and Valliappan K. Development and validation of a reversed-phase HPLC method for simultaneous determination of domperidone and pantoprazole in pharmaceutical dosage forms. Acta chromatographica 2007; 18:130-142.
- Chitlange SS, Mulla AI, Pawbake GR and Wankhede SB. Simultaneous Spectrophotometric Estimation Of Dexrabeprazole And Domperidone In Capsule Dosage Form. International Journal of Pharmaceutical Quality Assurance 2010; 2: 31-34.
- Tarkase KN, Tarkase MK, Dokhe MD and Wagh VS. Development And Validation of Spectrophotometric Method For Simultaneous Estimation of Cinnarizine And Domperidone Maleate In Pure And Tablet Dosage Form. Indian Journal of Pharmaceutical Sciences 2012; 3:2700-2704.
- Kulkarni AS, Balkrishna MV. Method Development and Validation for the Simultaneous Determination of Omeprazole and Domperidone in Solid Dosage form by Rp-Hplc. Indian Journal of Pharmacy and Pharmaceutical Sciences 2012; 4:109- 114.
- Babar SJ, Mane VB, Boise DSB. Development and Validation of UVSpectrophotometric Methods for Simultaneous Estimation of Paracetamol and Domperidone in Bulk and Tablet Dosage Form. Indian Journal of Pharmacy and Pharmaceutical Sciences 2012; 4:206-209.
- Pardeep K, Sarma GS, Vika P. Development and Validation of Derivative Spectrophotometric Method For Simultaneous Estimation Of Domperidone and Esomeprazole. International Journal of Universal Pharmacy and Life Sciences 2011; 1: 9- 15.
- Baldha RG, Vandana PB, and Bapna M. Simultaneous Spectrophotometric Estimation of Rabeprazole Sodium And Domperidone In Combined Dosage Forms. International Journal of Pharm Tech Research 2010; 2:1563-1568.
- Naveed S, Waheed N and Nazeer S. Degradation studies of ampicillin in API and formulations. J App Pharm 2014; 6(3):314-321.
- Naveed S, Naseem Y, Samie S, Khan S, Siddiqui S, Bushra and Ramzan S. Degradation Study of Five Different Brands of Ciprofloxacin Using UVVisible Spectrophotometer and Their Comparitive study. International Research Journal of Pharmacy 2014; 5(3):189-190.
- Naveed S, Shafiq A, Khan M, Jamal M, Zafar H, et al. Degradation Study of Available Brands of Metformin Karachi Using UV Spectrophotometer. J. Diabetes Metabolism 2014; 5:328.
- Naveed S*, Nazeer S and Waheed N. Degradation study of Meloxicam by UV-spectroscopy. British Journal of Research 2014; 1(3):105-112.