Keywords
Glioma; Tumor; Neurocognitive; Brain
Introduction
The ability to define and ultimately manage normal tissue injury in the brain remains limited as neither pathologic nor radiographic features have a clear correlation with clinical presentation (Figure 1). Normal tissue injury in the brain is often defined clinically by the development of late neurocognitive dysfunction affecting memory, executive function, learning ability and attention, not attributed to tumor progression or other comorbidities. The clinical manifestations of normal tissue injury affect 50-90% of glioma patients at some point in the course of their treatment [1,2]. Pathologic features of radiation injury include coagulative necrosis, vascular alterations with fibrinoid necrosis, edema, and demyelination [3] while radiologic features include increased signal on proton-density and T2-weighted MR images [4]. With increasing life expectancy in both adult and pediatric patients [5,6] and the advanced understanding of the glioma tumor`s molecular characteristics [7], which may define prognosis as well as treatment, the ability to address both is becoming increasingly relevant. With a prolonged period of time necessary to elicit overall survival benefit in low grade glioma patients [8] and the absence of a change in overall survival with newer therapies in high grade glioma [9-12], normal tissue toxicity and its clinical implications will become an increasingly important outcome measure in patients with glioma that merits both a better definition (clinical, radiographic, molecular) and better management options as normal tissue toxicity transitions from side effect to a potential biomarker for outcome. The use of imaging as a tool to investigate normal tissue injury represents a noninvasive modality that in conjunction with thorough neurocognitive and quality of life testing can help define and guide management of normal tissue injury related treatment sequelae in patients with glioma. We will discuss the imaging modalities currently employed to investigate normal tissue toxicity incurred by patients with glioma as a result of tumor-associated injury as well as treatment-associated injury and future directions for research and treatment.
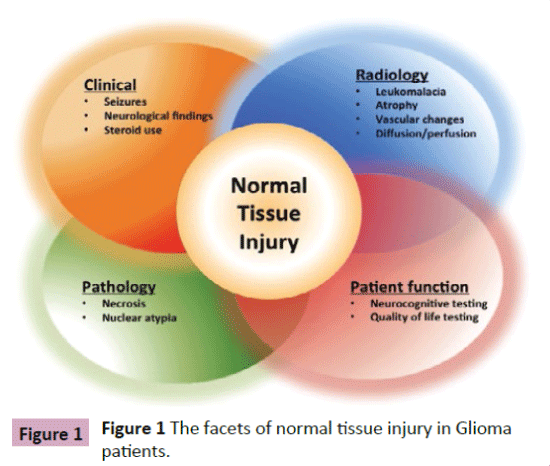
Figure 1: The facets of normal tissue injury in Gliomapatients.
The Multifactorial Causes of Normal Tissue Injury in Glioma
Prior to discussing available imaging modalities, a better definition of the underlying principles of normal tissue injury is in order. The etiology of brain dysfunction in patients with glioma is multifactorial [13-16] with significant correlations to patient related factors such as age, comorbidities, psychological factors and genetics as well as characteristics of the underlying malignancy, such as its natural history and infiltrative nature and finally the treatment modalities involved in management. Treatment for brain tumors is likewise multidisciplinary including surgical resection, radiation therapy and the administration of systemic targeted and non-targeted agents (Figure 2). With respect to surgical resection, cognitive function has long been studied in conjunction with epilepsy surgery [17] and also more recently in the setting of brain tumor resection with the identification of relationships of tumor location to neurocognitive outcome post resection [16]. Evidence for pre-treatment cognitive change [18] and the administration of multiple treatments have prompted the term “cancer and cancer treatment associated cognitive change” [19].
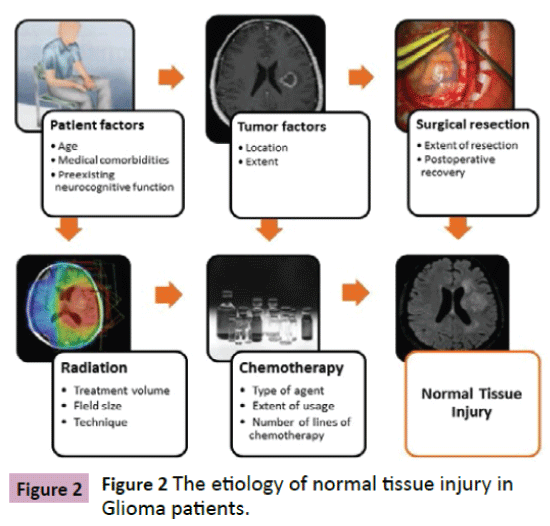
Figure 2: The etiology of normal tissue injury in Glioma patients.
Radiation induced brain injury is described in the form of acute (within days to weeks after irradiation), early -delayed (within 1-6 months post irradiation) and late (>6 months post irradiation) [20]. With respect to radiation therapy induced normal tissue toxicity imaging and animal models have been employed to study this phenomenon [21].
With respect to chemotherapy the concept of “chemobrain” is well described, though less so as applied to patients with glioma where the administration of radiation therapy may make distinguishing radiation toxicity from chemotherapy toxicity more difficult. Nonetheless increasing evidence is becoming available showing a neurocognitive detriment with the use of chemotherapy such as with the use of dose-dense temozolomide [22] and that of anti-angiogenic agents [23]. Most studies however are inadequately powered to distinguish the independent effects of multiple treatments and pretreatment neurocognitive testing is often lacking. Specific nervous system toxicities associated with emerging novel immunotherapies are now being discussed and will further alter the understanding of normal tissue injury in these patients.
The relationship between a patient`s clinical presentation and normal tissue injury remains ill defined. Radiographic definitions are lacking as conventional MRI typically employed to follow these patients for tumor progression has limited ability to distinguish normal tissue injury from treatment related changes such radiation necrosis or vascular changes [24]. Structural changes may be present on scans in previously radiated patients [25] but may have no bearing on the patient`s function, while conversely the patient`s function may be compromised in the absence of structural changes. Surgical re-resection in previously treated patients may help distinguish tumor recurrence from treatment related changes however is subject to sampling error and lack of clear pathological features distinguishing between the two. Since normal tissue injury may be more functional than structural in nature there is a growing emphasis to combining imaging modalities with extensive neurocognitive studies to better examine its relationship to treatment [26,27].
Normal Tissue Injury and Anatomical Relationships
The relationship between brain anatomy and the development of normal tissue injury is complex and remains a growing area of research. With this in mind a number of research efforts have been undertaken to determine which areas of the brain may be responsible for the sequelae we note in patients who have undergone treatment. The next step however will require a correlation between clinical manifestations and potential damage to these areas to enable both potential sparing and the identification of a biomarker for normal tissue injury. Studies in the literature have examined changes in whole brain, gray matter, white matter, ventricles, and combinations thereof [21,25,28].
One of the areas often discussed, is the hippocampus. Study of the hippocampus is prompted by the notion that creation of new memories has been associated with neural stem cells located in the subgranular zone of the hippocampal dentate gyrus and hence radiation related injury to this area may be associated with cognitive decline. A dose relationship has been elucidated in conjunction with neurocognitive testing and has prompted further studies involving hippocampal avoidance in the setting of radiation therapy [29].
Broader changes in white matter and grey matter and their relationship to neuro-cognition [30-32], have previously been analyzed in breast cancer patients in the setting of chemotherapy alone as well as in pediatric patients post chemotherapy and radiation therapy. Since white matter makes up 50% of the brain volume and contains an abundance of glial cells, oligodendrocytes and astrocytes [33], disruption of central conduction pathways by tumor or treatment can result in neurocognitive dysfunction. Grey matter including basal ganglia, thalamus, and hippocampus are responsible for attention, memory, and represent the primary domains typically affected by treatment.
In addition to white and grey matter and specifically the hippocampus as relevant anatomical areas structurally affected by treatment, ventricular changes have also been described, with ventricular dilation and progressive brain atrophy identified post chemo-irradiation for CNS patients [25,30,34]. These may also be related to clinical deficits, although the relationship is less clear.
Imaging Modalities to Detect Normal Tissue Injury
While CT scans have been shown to document gross changes in neuroanatomy post-radiation therapy, new advances in imaging are being applied to assess, anatomically and temporally normal tissue changes post-treatment. Advantages of non-invasive imaging include 1) the ability to characterize normal tissue injury potentially acting as a biomarker as well as 2) the possibility of intervention prior to clinically significant decline and 3) the ability to alter treatment with respect to both radiation and chemotherapy to reduce toxicity. We will review imaging methods employed to date (Figure 3).
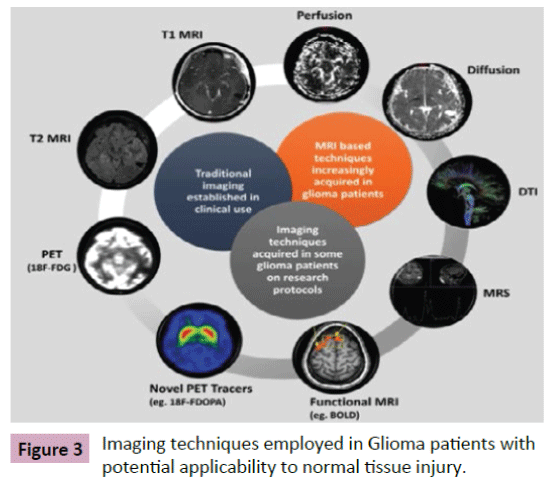
Figure 3: Imaging techniques employed in Glioma patients with potential applicability to normal tissue injury.
PET
Positron emission tomography (PET) imaging has been used as a non-invasive technique to quantify metabolic changes in the body due to physiologic processes, pathology, and treatment interventions. Using radioactive nuclides attached to various metabolic markers, PET scanning can localize areas of altered cellular metabolism, hypoxia, and proliferation [35-37]. The most common radiotracer, 18F-FDG, an analogue of glucose, localizes cellular energy metabolism. 18F-FDG-PET has been used to analyze normal tissue injury. Despite its lack of specificity, it can highlight areas of inflammation, apoptosis, or delineates areas of non-functional necrosis [37]. PET provides an interesting avenue of normal tissue investigation for three important reasons: 1) long term cognitive deficits post-radiation have been found to coincide with altered glucose metabolism in the brain. Studies of rhesus monkeys demonstrated a decline in cognitive function post-whole brain irradiation [21] which was associated with altered glucose uptake during neurocognitive tasks 9 months after radiation; 2). It has been shown to have a relationship to radiation dose level. In a trial comparing 38 adult survivors of acute lymphocytic leukemia, Krull et al. [38] compared the 18F-FDG-PET images of patients who had received 18Gy versus 24Gy of prophylactic cranial irradiation. They found an increase in metabolic activity in the basal ganglia of patients who had received the higher 24Gy dose, with both groups demonstrating higher activity in the cerebral cortex, basal ganglia, and thalamic regions. Increased metabolic activity was associated with oral naming speed in the frontal lobes, and negatively correlated with cognitive flexibility in the basal ganglia. More importantly sub-acute changes in neurocognitive function have been linked to uptake on PET scans. In a small group of 11 patients receiving 3D conformal radiation therapy, reductions in PET uptake were noted in regions of the brain receiving greater than 40Gy and were associated with worsening performance on neurophysiological tests [39]; 3) 18F-FDG-PET post-treatment can be employed to distinguish tumor progression from radiation necrosis [37].
Novel radiotracers are also increasingly available and may improve the specificity of PET imaging in these situations. 11C-methionine, 11C-choline, and 18F-FDOPA, are examples of radiotracers that are showing potential clinical use in CNS imaging [37,39,40].
While PET can in theory allow for the quantification of normal tissue changes its use remains limited by its lack of spatial resolution. Recent advancements in molecular imaging have allowed for the creation of combined PET-MRI scanners, which have allowed for improved anatomic and spatial resolution of PET signaling in the brain. With the use of these scanners, the metabolic information gained from PET can be co-registered with the anatomic and spatial information gained in MRI in a single scan. Thus merging the potential benefits of the two imaging modalities, and increasing the applicability of PET scanning in the anatomic localization of normal tissue injury in the brain.
MRI
Magnetic resonance imaging (MRI) is on the forefront of imaging techniques to delineate the anatomical and functional changes that occur as a result of treatment in glioma patients. Benefits of MRI imaging include improved soft tissue resolution, functional and perfusion sequences, and novel techniques that are able to image endogenous contrast agents. The post-operative syndrome of cerebellar mutism is currently under investigation with MRI in an effort to elucidate the etiology. In a series of patients followed for one year post-operatively with sequential MRI, at one year, increased brainstem and cerebellar atrophy was noted when compared to patient without symptoms [41]. Depending on the endpoint measured, the broad spectrum of available MRI techniques and sequences may be useful to build an overall picture of the changes that occur in normal tissue post-radiation therapy [42,43]. Correlations between imaging changes, pathologic tissue injury, and neurocognitive deficits have been reported [44]. However, limitations of this technique still remain, including variations in imaging protocols, contrast agents, magnet strength, operator dependence, data postprocessing and analysis which preclude effective comparison between studies [42,45,46]. Additional uses of MRI technique for the investigation of normal tissue injury that bear in depth discussion include perfusion and diffusion MRI and diffusion tensor imaging (DTI).
MR perfusion has been used to characterize vascularity within the brain, and alterations resulting from tissue damage and pathology. MR perfusion quantifies changes in cerebral blood volume, cerebral blood flow, and vascular permeability. The method of assessing MRI perfusion varies, and is dependent on the characteristic of interest.
Dynamic contrast-enhanced (DCE) perfusion MRI obtains serial T1-weighted images as intravenous gadolinium is injected, which derives a parametric map of the microvasculature, often quantified as a vascular leakage constant, K (trans). In a prospective study of patients receiving partial brain radiation therapy were assessed during treatment using the acute and subacute phase for temporal and dose-volume changes on DCE-MRI imaging, while also undergoing neurocognitive assessment [47]. Cerebral vascular volume and permeability significantly increased during radiation and acutely afterwards, then decreased in the subacute phase, with the most significant changes associated with high dose (>40Gy) regions. The intra-radiation changes in cerebral blood volumes of left temporal and frontal lobes and vascular permeability of left frontal lobes were significantly associated with changes in verbal memory and learning scores at 6 months post-RT. In addition, a dose-volume effect was noted. Future studies may help link overall prognosis and late neurocognitive changes to the changes in perfusion seen on imaging.
Additional methods of assessing altered vasculature include Dynamic susceptibility-contrast (DSC) MRI and arterial spin labeling (ASL). DSC perfusion uses T2-weighted images during an initial bolus of gadolinium contrast to assess relative cerebral blood volume, which quantifies the vessel density in a region of interest [48] Reductions in vascular density have been correlated with late cognitive impairment post-radiation therapy in animal models, and therefore may be a biomarker of future deficits [49]. A significant benefit of ASL is that it relies on endogenous magnetically labeled arterial blood, not exogenous contrast agents, and therefore allows for repeat imaging of during the same MRI session [50]. A dose-response reduction in cerebral blood flow has also been found after radiation therapy using ASL [42,51]. Additionally, ASL [52] demonstrated decreased cerebral blood flow in recurrent GBM treated with bevacizumab chemotherapy, not only in primary tumor, but also in the contralateral normal hemisphere. While human models have also shown changes in cerebral blood volume and blood flow after radiation therapy using this technique, further correlation with neurocognitive effects is required [53].
Diffusion weighted imaging (DWI) measures the random Brownian motion of water molecules in 3 dimensions within tissue, and based on this information apparent diffusion coefficient (ADC) maps can be created [54]. DWI and ADC maps delineate areas of increased cellularity and altered viscosity on brain MRI, and ADC values have been correlated with prognosis in low and high grade gliomas [55-58]. Due to the altered diffusion between unaffected brain parenchyma, necrosis, and highly cellular areas, DWI and ADC maps have been helpful in differentiating tumor recurrence in the CNS from radiation necrosis [59].
Diffusion tensor imaging (DTI) creates parametric maps measuring the direction and magnitude of water diffusion. The fractional anisotropy (FA) of DTI allows for directionality as well as diffusivity to be outlined. DTI has been suggested as a biomarker for radiation induced normal tissue injury, it has been shown to be more sensitive than T1 and T2 images for white matter changes [60,61], can be used to outline neuroplasticity and altered brain function [62], and directionality allows for characterization of myelinated tracts within the brain [63]. Subacute changes in DTI have been detected in the temporal lobed of patients treated with radiation for nasopharyngeal carcinoma [64], with associated alteration in MRS signals. Co-registration of prior radiation therapy plans with DTI and ADC maps, demonstrate late white matter changes in patients with primary brain tumors, that appear at dose levels as low as 5 to 15 Gy [65]. Irradiation of the spinal cord in children shows altered DTI signal changes non-uniformly throughout the brainstem, independent of dose, suggesting non-uniform radiation sensitivity of white matter tracts [66].
The pathologic correlations of DTI images in various animal models include demyelination, edema, persistent axonal injury, and cognitive impairment, [67-69]. In a prospective study of partial brain radiation for low-grade and benign tumors wherein the neurocognitive effects and DTI-MRI images post radiation were analyzed (61), altered diffusivity was noted within 3 weeks of beginning radiation, and remaining until week 78, with the percent change in the cingula signal at the end of radiation, predicting percent change 24 weeks later. Signal changes in the cingula 30 weeks after starting radiation were associated with both altered neurocognitive function, and changes during the course of RT and were predictors of later neurocognitive decline. DTI has also been used to follow pediatric survivors of acute lymphocytic leukemia treated with systemic chemotherapy, as they show lower grey matter volumes in the neocortical and subcortical structures as adults [70]. Lower hippocampal volumes in these patients correlated with lower adult neurocognitive performance.
To elucidate the clinical applicability of DTI as a biomarker for normal tissue toxicity, additional studies are required to correlate imaging changes with specific neurocognitive effects. DTI has been used to quantify in vivo radiation induced changes in the structural integrity of white matter in the CNS over time, with respect to permanence and reversibility; however, continued advances in DTI acquisition, distortion correction, and improved fiber tractography will improve its clinical applicability.
MRS
Magnetic resonance spectroscopy (MRS) has been used in CNS imaging to quantify metabolite distribution in the brain, as each molecule yields a characteristic resonance frequency [71-73]. Metabolites examined include, N-acetylaspartate (NAA), myoinositol, creatine, glutamate, lactate, taurine, and choline. MRS has been used to characterize malignant tumors [73-76] and differentiate necrosis from recurrent tumor [77]. New evidence suggests that neurochemical changes in the surrounding normal brain may correlate with post-radiation neurocognitive effects. MRS has shown changes, not detected on standard MRI sequences, in the metabolic profile of normal brain parenchyma during, and after, a course of radiation therapy [78-82]. In a recent study investigating hippocampal neuron density, a cohort of patients undergoing whole brain radiation for cerebral metastases were found to have a decrease in neuronal marker, NAA, in the subacute phase after radiation. While not statistically significant, a simultaneous decline in scores for Auditory Verbal Learning Test, and Brief Visuospatial Memory Test - Revised was observed between hippocampal NAA concentrations and test scores [83]. Further research is needed in MRS to delineate the anatomic, temporal, and neurochemical changes that coincide with progressive clinical effects.
Data translation
The fundamental scientific challenges of translating normal tissue injury findings in glioma patients involve understanding what exactly particular imaging changes represent on biological and cellular levels. Comparing these imaging findings to normal brain injury in other pathologic states such as traumatic brain injury, neurodegenerative disorders such as Alzheimer’s, and discoveries in normal brain injury from other tumors such as brain metastases, as well as developing de novo hypotheses specific to glioma patients are currently the main conduits for bringing novel discoveries to use in glioma patients. Previously, most attempts at clinical trials into normal tissue injury in glioma patients have focused on clinical endpoints defined by shortterm changes as a consequence the limited lifespan of patients. Instead of laboratory bench findings directing bedside studies, previous and current trials have mainly focused on the bedside as dictator of research goals [27,84]. As a result, limited successes have been realized. However, in the past decade, with increases in expected survival times, a newer focus on late effects and early intervention, as well as prevention have moved to the forefront.
The fundamental questions to translate our current pool of data to the clinic involve understanding the effect of glioma and its treatments on normal brain injury, and subsequently, how to reverse or prevent adverse effects most efficiently. These questions include: How do changes in current imaging parameters (typically MRI-based), translate to neurocognitive outcomes, prognostic outcomes, quality of life outcomes, and biomarker fluctuations? How can we use this information to successfully define and target the individual contributions of adverse effects from tumor, chemotherapy, and/or radiation? What pharmacologic and non-pharmacologic strategies are effective?
However, several recent studies have reported data that could have implications for management of normal brain structure and function in glioma patients. Polymorphisms in inflammation, DNA repair, and metabolism pathways have been found to be associated with neurocognitive function in glioma patients and may affect clinical outcomes [85]. These data suggest that neurocognitive outcomes are not merely defined by tumor or interventions for treatment, but are determined by the genetic background on which those changes occur. Surgical studies using intraoperative mapping with direct electrical stimulation (DES) has been used to identify cortical and subcortical white matter pathways critical for language, motor, and sensory function. Overall, 33.3% of patients exhibited loss of function at one or more motor or language sites between surgeries for recurrent glioma. Loss of function at these sites was not associated with neurological impairment at the time of a repeat surgery, suggesting that neurological function is preserved through neural circuit reorganization or activation of latent functional pathways [86]. These investigations have demonstrated that the adult central nervous system reorganizes motor and language areas in patients with glioma. Determining how and being able to predict where the brain reorganizes these functions will be the goal of future investigations.
Differentiation of recurrent glioma from radiation necrosis is one of the most vexing normal tissue considerations in glioma patients. A recent meta-analysis of imaging studies attempting to differentiate between the two has demonstrated that magnetic resonance spectroscopy alone has moderate diagnostic performance in differentiating glioma recurrence from radiation necrosis using metabolite ratios like Cho/Cr and Cho/NAA ratios [77]. However, future studies will determine how magnetic resonance spectroscopy should be combined with other advanced imaging technologies to improve diagnostic accuracy.
Attempts at sparing brain sub compartments from injury are borrowed from whole brain radiation studies. RTOG 0933 used advanced radiation planning techniques to spare the neural stemcell population in the adult hippocampus in patients receiving whole brain radiation with brain metastases with limited benefit at short time intervals [29]. Likewise, the INTRAGO Trial is utilizing intraoperative radiotherapy for newly diagnosed glioblastoma in a phase I/II dose escalation trial to minimize extraneous normal tissue damage from radiotherapy [87].
Pharmacological interventions under consideration for prevention of neurological dysfunction in glioma patients include memantine, an NMDA receptor antagonist used in the treatment of Alzheimer’s patients and patients with obsessive-compulsive disorder [84]. In RTOG 0614, patients receiving whole brain radiation treatment and concurrently treated with memantine had better cognitive function over time; specifically, memantine delayed time to cognitive decline and reduced the rate of decline in memory, executive function, and processing speed in patients receiving. Although there was decreased decline in the primary endpoint of delayed recall at 24 weeks, the study may be criticized for a lack of statistical significance, possibly due to significant patient loss. The benefit of non-pharmacological strategies using exercise-induced stimulation of hippocampal neurogenesis, cognitive therapies and mesenchymal stem cell replacement to prevent or delay onset of normal tissue injury in patients are currently under investigation [88,89].
Overwhelmingly, what these data demonstrate is there is a dire need for improved understanding of the biology related to the effects of tumor, chemotherapies, and radiation on the short term and late changes in brain microenvironment, neuroimmunity, neural circuitry and pathways, blood-brain barrier regulation, and subcellular mechanisms of neurological dysfunction and neurodegeneration. Imaging-related biomarker identification for normal tissue injury and response in glioma patients is one avenue that may show potential promise in identifying patients in whom interventional strategies are needed. A MRI-based vascular hippocampal marker parameter related to blood-brain barrier permeability, K(trans), and the fraction of blood plasma volume, Vp, have recently been investigated [90], correlating significantly with changes in memory function at 6 and 18-months after treatment, suggesting that K(trans) could serve as one predictor of late neurocognitive dysfunction for glioma patients.
Future Directions and Recommendations
We have identified as ongoing areas of improvement the lack of reasonable comparison between retrospective studies aimed at the analysis of imaging data in conjunction with normal tissue toxicity in glioma patients. This extends from the wide heterogeneity with respect to designation of region of interest, post-processing of images and the general approaches to data analysis and is compounded by selection bias with respect to both patients and anatomical areas of the brain. There is an ongoing lack of correlation of imaging to clinical presentation, quality of life and neurocognitive testing as well as tissue obtained from resection and re-resection. Conclusions are further limited by data drawn from small studies of 10 to 50 patients which represent a great proportion of literature on the subject. This reflects one of the fundamental barriers to increased understanding of normal tissue injury in glioma patients which stems from a lack of support of research funding in normal tissue research proposals from leading cancer funding agencies. Currently, most major funding mechanisms have a requirement for a research focus on tumor as opposed to normal tissue toxicities. This is in contrast to other malignancies such as breast cancer where a growing body of literature exists on neurocognition in relationship to systemic treatment. A task force such as the Brain Tumor Clinical Trial Endpoints Workshop on at NCI have been established to gather consensus and build a more robust basis for improving clinical outcome assessments as study endpoints and advance their inclusion into adult primary brain tumor clinical trials.
As a scientific community, collaborative research is needed into exceptional responders to better distinguish the biological mechanisms operating in those patients who experience severe changes from tumor and its associated treatments in comparison to those patients with minimal to no damage from glioma. As suggested by current data, the underlying difference may be due in part to possible differences in genetic polymorphisms.
Finally, more research is needed into differentiation of tumorassociated injury versus therapy-associated injuries in glioma patients. Development of novel imaging techniques to demonstrate additional functional and metabolic differences as well as structural variations may assist in answering this question. Ultimately further understanding of normal tissue toxicities in glioma patients will benefit the advancement of research and ultimately improve the outcome of other malignancies where the underlying disease process may involve the central nervous system.
Acknowledgment
This work was supported in part by the Centers for Cancer Research, NCI.
References
- Taphoorn MJ (2003) Neurocognitive sequelae in the treatment of low-grade gliomas. SeminOncol 30: 45-48.
- Correa DD, DeAngelis LM, Shi W, Thaler HT, Lin M, et al. (2007) Cognitive functions in low-grade gliomas: disease and treatment effects. J Neurooncol 81: 175-184.
- Tiller-Borcich JK, Fike JR, Phillips TL, Davis RL (1987) Pathology of delayed radiation brain damage: an experimental canine model. Radiat Res 110: 161-172.
- Valk PE, Dillon WP (1991) Radiation injury of the brain. AJNR Am J Neuroradiol 12: 45-62.
- Mulhern RK, Merchant TE, Gajjar A, Reddick WE, Kun LE (2004) Late neurocognitive sequelae in survivors of brain tumours in childhood. Lancet Oncol 5: 399-408.
- Jemal A, Center MM, DeSantis C, Ward EM (2010) Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol Biomarkers Prev 19: 1893-1907.
- Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, et al. (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17: 98-110.
- Shaw EG, Wang M, Coons SW, Brachman DG, Buckner JC, et al. (2012) Randomized trial of radiation therapy plus procarbazine, lomustine, and vincristine chemotherapy for supratentorial adult low-grade glioma: initial results of RTOG 9802. J ClinOncol 30: 3065-3070.
- Brandes AA, Stupp R, Hau P, Lacombe D, Gorlia T, et al. (2010) EORTC study 26041-22041: phase I/II study on concomitant and adjuvant temozolomide (TMZ) and radiotherapy (RT) with PTK787/ZK222584 (PTK/ZK) in newly diagnosed glioblastoma. Eur J Cancer 46: 348-354.
- Chinot OL, Wick W, Mason W, Henriksson R, Saran F, et al. (2014) Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N Engl J Med 370: 709-722.
- Gilbert MR, Dignam JJ, Armstrong TS, Wefel JS, Blumenthal DT, et al. (2014) A randomized trial of bevacizumab for newly diagnosed glioblastoma. N Engl J Med 370: 699-708.
- Taal W, Bromberg JE, van den Bent MJ (2015) Chemotherapy in glioma. CNS Oncol 4: 179-192.
- Belka C, Budach W, Kortmann RD, Bamberg M (2001) Radiation induced CNS toxicity--molecular and cellular mechanisms. Br J Cancer 85: 1233-1239.
- Hurria A, Rosen C, Hudis C, Zuckerman E, Panageas KS, et al. (2006) Cognitive function of older patients receiving adjuvant chemotherapy for breast cancer: a pilot prospective longitudinal study. J Am GeriatrSoc 54: 925-931.
- Ahles TA (2012) Brain vulnerability to chemotherapy toxicities. Psychooncology 21: 1141-1148.
- Noll KR, Sullaway C, Ziu M, Weinberg JS, Wefel JS (2015) Relationships between tumor grade and neurocognitive functioning in patients with glioma of the left temporal lobe prior to surgical resection. NeuroOncol 17: 580-587.
- Rausch R, Kraemer S, Pietras CJ, Le M, Vickrey BG, et al. (2003) Early and late cognitive changes following temporal lobe surgery for epilepsy. Neurology 60: 951-959.
- Tucha O, Smely C, Preier M, Lange KW (2000) Cognitive deficits before treatment among patients with brain tumors. Neurosurgery 47: 324-333.
- Ahles TA, Root JC, Ryan EL (2012) Cancer- and cancer treatment-associated cognitive change: an update on the state of the science. J ClinOncol 30: 3675-3686.
- Tofilon PJ, Fike JR (2000) Theradioresponse of the central nervous system: a dynamic process. Radiat Res 153: 357-370.
- Robbins ME, Bourland JD, Cline JM, Wheeler KT, Deadwyler SA (2011) A model for assessing cognitive impairment after fractionated whole-brain irradiation in nonhuman primates. Radiat Res 175: 519-525.
- Armstrong TS, Wefel JS, Wang M, Gilbert MR, Won M, et al. (2013) Net clinical benefit analysis of radiation therapy oncology group 0525: a phase III trial comparing conventional adjuvant temozolomide with dose-intensive temozolomide in patients with newly diagnosed glioblastoma. J ClinOncol 31: 4076-4084.
- Johannesen TB, Lien HH, Hole KH, Lote K (2003) Radiological and clinical assessment of long-term brain tumour survivors after radiotherapy. RadiotherOncol 69: 169-176.
- Prust MJ, Jafari-Khouzani K, Kalpathy-Cramer J, Polaskova P, Batchelor TT, et al. (2015) Standard chemoradiation for glioblastoma results in progressive brain volume loss. Neurology 85: 683-691.
- Meyers CA, Brown PD (2006) Role and relevance of neurocognitive assessment in clinical trials of patients with CNS tumors. J ClinOncol 24: 1305-1309.
- Shaw EG, Rosdhal R, D'Agostino RB, Lovato J, Naughton MJ, et al. (2006) Phase II study of donepezil in irradiated brain tumor patients: effect on cognitive function, mood, and quality of life. J ClinOncol 24: 1415-1420.
- Gondi V, Tomé WA, Mehta MP (2010) Why avoid the hippocampus? A comprehensive review. RadiotherOncol 97: 370-376.
- Gondi V, Pugh SL, Tome WA, Caine C, B Corn A, et al. (2014) Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J ClinOncol 32: 3810-3816.
- Stylopoulos LA, George AE, de Leon MJ, Miller JD, Foo SH, et al. (1988) Longitudinal CT study of parenchymal brain changes in glioma survivors. AJNR Am J Neuroradiol 9: 517-522.
- Simó M, Rifà-Ros X, Rodriguez-Fornells A, Bruna J (2013) Chemobrain: a systematic review of structural and functional neuroimaging studies. NeurosciBiobehav Rev 37: 1311-1321.
- Kaiser J, Bledowski C, Dietrich J (2014) Neural correlates of chemotherapy-related cognitive impairment. Cortex 54: 33-50.
- Goldberg MP, Ransom BR (2003) New light on white matter. Stroke 34: 330-332.
- Omuro AM, Ben-Porat LS, Panageas KS, Kim AK, Correa DD, et al. (2005) Delayed neurotoxicity in primary central nervous system lymphoma. Arch Neurol 62: 1595-1600.
- Chen W, Cloughesy T, Kamdar N, Satyamurthy N, Bergsneider M, et al. (2005) Imaging proliferation in brain tumors with 18F-FLT PET: comparison with 18F-FDG. J Nucl Med 46: 945-952.
- Lopci E, Grassi I, Chiti A, Nanni C, Cicoria G, et al. (2014) PET radiopharmaceuticals for imaging of tumor hypoxia: a review of the evidence. Am J Nucl Med Mol Imaging 4: 365-384.
- Takenaka S, Asano Y, Shinoda J, Nomura Y, Yonezawa S, et al. (2014) Comparison of (11)C-methionine, (11) C-choline, and (18) F-fluorodeoxyglucose-PET for distinguishing glioma recurrence from radiation necrosis. Neurol Med Chir (Tokyo) 54: 280-289.
- Krull KR, Minoshima S, Edelmann M, Morris B, Sabin ND, et al. (2014) Regional brain glucose metabolism and neurocognitive function in adult survivors of childhood cancer treated with cranial radiation. J Nucl Med 55: 1805-1810.
- Fink JR, Muzi M, Peck M, Krohn KA (2015) Multimodality Brain Tumor Imaging: MR Imaging, PET, and PET/MR Imaging. J Nucl Med 56: 1554-1561.
- Lizarraga KJ, Allen-Auerbach M, Czernin J, DeSalles AA, Yong WH, et al. (2014) (18)F-FDOPA PET for differentiating recurrent or progressive brain metastatic tumors from late or delayed radiation injury after radiation treatment. J Nucl Med 55: 30-36.
- Wells EM, Khademian ZP, Walsh KS, Vezina G, Sposto R, et al. (2010) Postoperative cerebellar mutism syndrome following treatment of medulloblastoma: neuroradiographic features and origin. J NeurosurgPediatr 5: 329-334.
- Knutsson L, van Westen D, Petersen ET, Bloch KM, Holtas S, et al. (2010) Absolute quantification of cerebral blood flow: correlation between dynamic susceptibility contrast MRI and model-free arterial spin labeling. MagnReson Imaging 28: 1-7.
- Kimura M, da Cruz LC Jr (2016) Multiparametric MR Imaging in the Assessment of Brain Tumors. MagnReson Imaging Clin N Am 24: 87-122.
- Hanbury DB, Robbins ME, Bourland JD, Wheeler KT, Peiffer AM, et al. (2015) Pathology of fractionated whole-brain irradiation in rhesus monkeys (Macacamulatta).Radiat Res 183: 367-374.
- Bian W, Hess CP, Chang SM, Nelson SJ, Lupo JM (2014) Susceptibility-weighted MR imaging of radiation therapy-induced cerebral microbleeds in patients with glioma: a comparison between 3T and 7T. Neuroradiology 56: 91-96.
- Heye AK, Culling RD, Valdes C, Hernandez Mdel MJ, Thrippleton , et al. (2014) Assessment of blood-brain barrier disruption using dynamic contrast-enhanced MRI. A systematic review. NeuroimageClin 6: 262-274.
- Cao Y, Tsien CI, Sundgren PC, Nagesh V, Normolle D, et al. (2009) Dynamic contrast-enhanced magnetic resonance imaging as a biomarker for prediction of radiation-induced neurocognitive dysfunction. Clin Cancer Res 15: 1747-1754.
- Shiroishi MS, Castellazzi G, Boxerman JL, D'Amore F, Essig M, et al. (2015) Principles of T2*-weighted dynamic susceptibility contrast MRI technique in brain tumor imaging. J MagnReson Imaging 41: 296-313.
- Brown WR, Blair RM, Moody DM, Thore CR, Ahmed S, etal. (2007) Capillary loss precedes the cognitive impairment induced by fractionated whole-brain irradiation: a potential rat model of vascular dementia. J NeurolSci 257: 67-71.
- Petcharunpaisan S, Ramalho J, Castillo M (2010) Arterial spin labeling in neuroimaging. World J Radiol 2: 384-398.
- Petr J, Platzek I, Seidlitz A, Mutsaerts HJ, Hofheinz F, et al. (2016) Early and late effects of radiochemotherapy on cerebral blood flow in glioblastoma patients measured with non-invasive perfusion MRI. RadiotherOncol 118: 24-28.
- Andre JB, Nagpal S, Hippe DS, Ravanpay AC, Schmiedeskamp H, et al. (2015) Cerebral Blood Flow Changes in Glioblastoma Patients Undergoing Bevacizumab Treatment Are Seen in Both Tumor and Normal Brain. Neuroradiol J 28: 112-119.
- Jakubovic R, Sahgal A, Ruschin M, Pejovic-Milic A, Milwid R et al. (2015) Non Tumor Perfusion Changes Following Stereotactic Radiosurgery to Brain Metastases. Technol Cancer Res Treat 14: 497-503.
- Hagmann P, Jonasson L, Maeder P, Thiran JP, Wedeen VJ, et al. (2006) Understanding diffusion MR imaging techniques: from scalar diffusion-weighted imaging to diffusion tensor imaging and beyond. Radiographics 26 Suppl 1: S205-223.
- Desprechins B, Stadnik T, Koerts G, Shabana W, Breucq C, et al. (1999) Use of diffusion-weighted MR imaging in differential diagnosis between intracerebral necrotic tumors and cerebral abscesses. AJNR Am J Neuroradiol 20: 1252-1257.
- Tung GA, Evangelista P, Rogg JM, Duncan JA 3rd (2001) Diffusion-weighted MR imaging of rim-enhancing brain masses: is markedly decreased water diffusion specific for brain abscess? AJR Am J Roentgenol 177: 709-712.
- Bulakbasi N, Guvenc I, Onguru O, Erdogan E, Tayfun C et al. (2004) The added value of the apparent diffusion coefficient calculation to magnetic resonance imaging in the differentiation and grading of malignant brain tumors. J Comput Assist Tomogr 28: 735-746.
- Server A, Kulle B, Maehlen J, Josefsen R, Schellhorn T, et al. (2009) Quantitative apparent diffusion coefficients in the characterization of brain tumors and associated peritumoraledema. ActaRadiol 50: 682-689.
- Asao C, Korogi Y, Kitajima M, Hirai T, Baba Y, et al. (2005) Diffusion-weighted imaging of radiation-induced brain injury for differentiation from tumor recurrence. AJNR Am J Neuroradiol 26: 1455-1460.
- Assaf Y, Galron R, Shapira I, Nitzan A, Blumenfeld-Katzir T, et al. (2008) MRI evidence of white matter damage in a mouse model of Nijmegen breakage syndrome. ExpNeurol 209: 181-191.
- Chapman CH, V Nagesh, Sundgren PC, Buchtel H, Chenevert TL, et al. (2012) Diffusion tensor imaging of normal-appearing white matter as biomarker for radiation-induced late delayed cognitive decline. Int J RadiatOncolBiolPhys 82: 2033-2040.
- Blumenfeld-Katzir T, Pasternak O, Dagan M, Assaf Y (2011) Diffusion MRI of structural brain plasticity induced by a learning and memory task. PLoS One 6: e20678.
- Smits M, Vernooij MW, Wielopolski PA, Vincent AJ, Houston GC (2007) Incorporating functional MR imaging into diffusion tensor tractography in the preoperative assessment of the corticospinal tract in patients with brain tumors. AJNR Am J Neuroradiol 28: 1354-1361.
- Wang HZ, Qiu SJ, Lv XF, Wang YY, Liang Y, et al. (2012) Diffusion tensor imaging and 1H-MRS study on radiation-induced brain injury after nasopharyngeal carcinoma radiotherapy. ClinRadiol 67: 340-345.
- Ravn S, Holmberg M, Sorensen P, Frokjaer JB, Carl J (2013) Differences in supratentorial white matter diffusion after radiotherapy--new biomarker of normal brain tissue damage? ActaOncol 52: 1314-1319.
- Hua C, Merchant TE, Gajjar A, Broniscer A, Zhang Y, et al. (2012) Brain tumor therapy-induced changes in normal-appearing brainstem measured with longitudinal diffusion tensor imaging. Int J RadiatOncolBiolPhys 82: 2047-2054.
- Song SK, Sun SW, Ramsbottom MJ, Chang C, Russell J, et al. (2002) Dysmyelination revealed through MRI as increased radial (but unchanged axial) diffusion of water. Neuroimage 17: 1429-1436.
- Wang S, Wu EX, Qiu D, Leung LH, Lau HF, et al. (2009) Longitudinal diffusion tensor magnetic resonance imaging study of radiation-induced white matter damage in a rat model. Cancer Res 69: 1190-1198.
- Peiffer AM, Creer RM, Linville C, Olson J, Kulkarni P, et al. (2014) Radiation-induced cognitive impairment and altered diffusion tensor imaging in a juvenile rat model of cranial radiotherapy. Int J RadiatBiol 90: 799-806.
- Genschaft M, Huebner T, Plessow F, Ikonomidou VN, Abolmaali N, et al. (2013) Impact of chemotherapy for childhood leukemia on brain morphology and function. PLoS One 8: e78599.
- Urenjak J, Williams SR, Gadian DG, Noble M (1993) Proton nuclear magnetic resonance spectroscopy unambiguously identifies different neural cell types. J Neurosci 13: 981-989.
- Preul MC, Caramanos Z, Collins DL, Villemure JG, Leblanc R, et al. (1996) Accurate, noninvasive diagnosis of human brain tumors by using proton magnetic resonance spectroscopy. Nat Med 2: 323-325.
- Howe FA, Barton SJ, Cudlip SA, Stubbs M, Saunders DE, et al. (2003) Metabolic profiles of human brain tumors using quantitative in vivo 1H magnetic resonance spectroscopy. MagnReson Med 49: 223-232.
- Law M, Yang S, Wang H, Babb JS, Johnson G, et al. (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. AJNR Am J Neuroradiol 24: 1989-1998.
- Calvar JA, Meli FJ, Romero C, Calcagno ML, Yánez P, et al. (2005) Characterization of brain tumors by MRS, DWI and Ki-67 labeling index. J Neurooncol 72: 273-280.
- Zonari P, Baraldi P, Crisi G (2007) Multimodal MRI in the characterization of glial neoplasms: the combined role of single-voxel MR spectroscopy, diffusion imaging and echo-planar perfusion imaging. Neuroradiology 49: 795-803.
- Zhang H, Ma L, Wang Q, Zheng X, Wu C, et al. (2014) Role of magnetic resonance spectroscopy for the differentiation of recurrent glioma from radiation necrosis: a systematic review and meta-analysis. Eur J Radiol 83: 2181-2189.
- Rutkowski T, Tarnawski R, Sokol M, Maciejewski B (2003) 1H-MR spectroscopy of normal brain tissue before and after postoperative radiotherapy because of primary brain tumors. Int J RadiatOncolBiolPhys 56: 1381-1389.
- Lee MC, Pirzkall A, McKnight TR, Nelson SJ (2004) 1H-MRSI of radiation effects in normal-appearing white matter: dose-dependence and impact on automated spectral classification. J MagnReson Imaging 19: 379-388.
- Kaminaga T, Shirai K (2005) Radiation-induced brain metabolic changes in the acute and early delayed phase detected with quantitative proton magnetic resonance spectroscopy. J Comput Assist Tomogr 29: 293-297.
- Matulewicz L, Sokol M, Michnik A, Wydmanski J (2006) Long-term normal-appearing brain tissue monitoring after irradiation using proton magnetic resonance spectroscopy in vivo: statistical analysis of a large group of patients. Int J RadiatOncolBiolPhys 66: 825-832.
- Sundgren PC, Nagesh V, Elias A, Tsien C, Junck L, et al. (2009) Metabolic alterations: a biomarker for radiation-induced normal brain injury-an MR spectroscopy study. J MagnReson Imaging 29: 291-297.
- Pospisil P, Kazda T, Bulik M, Dobiaskova M, Burkon P, et al. (2015) Hippocampal proton MR spectroscopy as a novel approach in the assessment of radiation injury and the correlation to neurocognitive function impairment: initial experiences. RadiatOncol 10: 211.
- Brown PD, Pugh S, Laack NN, Wefel JS, Khuntia D, et al. (2013) Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. NeuroOncol 15: 1429-1437.
- Liu Y, Zhou R, Sulman EP, Scheurer ME, Boehling N, et al. (2015) Genetic Modulation of Neurocognitive Function in Glioma Patients. Clin Cancer Res 21: 3340-3346.
- Southwell DG, Hervey-Jumper SL, Perry DW, Berger MS (2015) Intraoperative mapping during repeat awake craniotomy reveals the functional plasticity of adult cortex. J Neurosurg,pp: 1-10.
- Giordano FA, Brehmer S, Abo-Madyan Y, Welzel G, Sperk E, et al. (2014) INTRAGO: intraoperative radiotherapy in glioblastomamultiforme-a phase I/II dose escalation study. BMC Cancer 14: 992.
- Biedermann SV, Fuss J, Steinle J, Auer MK, et al. (2016) The hippocampus and exercise: histological correlates of MR-detected volume changes. Brain StructFunct 221: 1353-1363.
- Donega V, Nijboer CH, van Tilborg G, Dijkhuizen RM, Kavelaars A, et al. (2014) Intranasally administered mesenchymal stem cells promote a regenerative niche for repair of neonatal ischemic brain injury. ExpNeurol 261: 53-64.
- Farjam R, Pramanik P, Aryal MP, Srinivasan A, Chapman CH, et al. (2015) A Radiation-Induced Hippocampal Vascular Injury Surrogate Marker Predicts Late Neurocognitive Dysfunction. Int J RadiatOncolBiolPhys 93: 908-915.
- Wen PY, Cloughesy TF, Ellingson BM, Reardon DA, Fine HA, et al. (2014) Report of the Jumpstarting Brain Tumor Drug Development Coalition and FDA clinical trials neuroimaging endpoint workshop (January 30, 2014, Bethesda MD) NeuroOncol 16: 36-47.