Singh O1*, Meena RN2, Srivastava V3, Pandey V3, Shah AG2 and Meena RK1
1Department of Medicine, Institute of Medial Sciences, Banaras Hindu University, Varanasi, India
2Department of General Surgery, Institute of Medial Sciences, Banaras Hindu University, Varanasi, India
3Department of Pediatric Surgery, Institute of Medial Sciences, Banaras Hindu University, Varanasi, India
*Corresponding Author:
Singh O
Department of Medicine, Institute of Medial Sciences, Banaras Hindu University, Varanasi, India
Tel: +91-542-6701602
E-mail: opbhu07@gmail.com
Received date: December 30, 2015, Accepted date: January 28, 2016, Published date: February 01, 2016
Citation: Singh O, Meena RN, Srivastava V, et al. Impact of Brain Tumour Treatment on Quality of Life in Children: A Health Perspective. Neurooncol Open Access. 2016, 1:1.
Keywords
Brain tumor; Cancer treatment; Neuro-oncology; Children
Introduction
Tumours arising from in and around the brain in which some cells grow and multiply uncontrollably due to loss of mechanisms that control normal cells growth. Because of the location, significant long-term impairment to intellectual and neurological function is possible. They can also be life threatening if not treated promptly. The cause of primary brain tumors is unknown. However, primary brain tumours are the second most common cancer in children and the most common solid neoplasm of childhood, representing about 20% of all pediatric cancers [1,2]. For example, between 2001 and 2005, 4,181children aged 0-14 years were diagnosed with cancer in Canada, and between 2000 and 2004, 676 died from this disease [1]. The incidence of primary pediatric brain tumours is approximately 2.76 to 4.28 cases per 100,000 children [2]. The reported incidence of pediatric brain tumours has been increasing over the last three decades, probably due to improved diagnostics [3]. Refinements in imaging, surgical technique and adjunctive therapies have led to longer survival in children with brain tumours [2,4,5]. As more children survive a cancer diagnosis, the need for long-term monitoring and follow-up care continues to grow [1].
Treatment
Treatment of brain tumours is complicated because of the delicate surrounding tissue. Among all tumors, pediatric brain tumors are life-threatening, most children and adolescents with this diagnosis survive into adulthood. Treatment for pediatric cancers usually involves one or more of three main modalities: localized surgical resection, radiation therapy and/or chemotherapy. The trend in neurosurgery for pediatric neuro-oncological conditions has been toward less invasive procedures and non- surgical interventions, where possible [6], as treatment toxicities are cumulative [7]. The outcomes for children diagnosed with and treated for cancer are largely dependent on host factors, the type of cancer diagnosed, timing, and treatments received [8]. Aggressive therapy is often needed to cure this potentially lethal disease, yet late effects, such as injury to the developing brain, remain a profound concern [9]. With surgical resection, craniospinal irradiation and chemotherapy, cure rates as high as 85% are achieved with average-risk cases. However, these remarkable improvements in survival are achieved at high cost to survivors’ HRQL [10].
Child Development and Cancer Treatments
Children who receive treatment for brain tumours are uniquely challenged as their diagnosis and treatment occurs concurrently with ongoing development. Understanding the basic processes of normal child development helps to determine the potential consequences of receiving treatment during this formative time. Child development or developmental science is devoted to understanding constancy and change from conception through adolescence [11]. Discreet periods of development have been identified including (a) the prenatal period (conception to birth), (b) infancy and toddlerhood (birth to two years), (c) early childhood (two to six years), (d) middle childhood (six to eleven years) and (e) adolescence (eleven to eighteen years). Each developmental stage is characterized by new capacities and social expectations that define the three broad domains of cognitive, physical and emotional/social development. During infancy and toddlerhood, brain and body growth supports the development of motor, perceptual and intellectual capacities including early language development. The first independent steps usually take place in this phase. In early childhood, as the body becomes longer and leaner, refinement in motor skills occurs and self-control and self-sufficiency emerge. Language development is rapid during this period, with morality and socialization with peers becoming important. During the first year of life, the auditory and visual cortexes responsible for body movement develop at a rapid rate. Language areas are particularly active during late infancy through preschool years. Previous studies have suggested that the cerebral cortex is highly plastic during these early years; this protects it from damage as other parts can take over any lost cortical functions [11]. Receiving cancer treatment for a brain tumour during this period of child development can pose many challenges to normal neurodevelopment.
Neuro-developmental Challenges
There are many aspects of cancer treatment that can cause neurodevelopmental deficits in children being treated for cancer. The serious consequences for normal brain development have been studied using in utero exposure to radiation [12]. This research has demonstrated that the developing brains in children are much more sensitive than the adult brain. Treatments that target the central nervous system, including chemotherapy or radiation to the brain or spinal cord, can lead to neurodevelopmental deficits [13]. Developmental theory helps to explain the ongoing interaction between a child and his/her environment when he/she is undergoing treatment for a brain tumour.
Synactive Theory
The synactive theory of development provides a framework for understanding the neurobehavioural capabilities of a fetus, newborn and young infant during early development [14]. The theory helps practitioners understand the interaction between the nervous system and child development by observing physiological cues including motor behaviour, attention and social interaction [15]. It describes a dynamic continuous interaction between the autonomic system, the motor system, the organizational system (e.g. sleep-wake cycles), the attentional- interactive system (e.g. adaptive skills) and the self-regulatory balancing system [14]. Functional competence is achieved as the child moves through the developmental agenda while interacting with his or her environment [14]. The five subsystems of the synactive theory are described as interdependent and interrelated with a loss of integrity in one system affecting the others [16]. Developmental challenges occur when neurobehavioural demands become overwhelming for the child, and functioning in one area affects the others [15]. Although this theory has been used almost exclusively to plan nursing care for premature infants in neonatal intensive care units [16], it has also been used to guide brain development and early childhood education curriculums [15]. This model can be used in everyday practice to facilitate the integration of neurobehavioural consideration of infants, toddler and preschoolers and their families [15]. This model also provides an important context for understanding the developmental challenges faced by a child following treatment for a brain tumour with respect to motor, behaviour and social interactions. Undergoing treatment for a brain tumour during normal child development can result in developmental challenges that can have adverse outcomes in survivors.
Surviving Childhood Brain Tumours
Survivorship is conceptualized as the phase of cancer care that begins after the completion of definitive therapy [17]. Brain tumour survivors include children and youth who have completed treatment for a brain tumour and who have received surgery, chemotherapy and/or radiation therapy. Long-term survivors of childhood central nervous system malignancies are at increased risk for late mortality, development of second neoplasms, multiple endocrinopathies and adverse neurologic health conditions [18]. Children diagnosed with brain tumours during infancy have been found to have developmental delays in a number of areas of adaptive function. By the time they reached school age, children displayed further compromise in cognitive functions, academic skills and adaptive behaviour. Higher levels of deficit at follow- up were associated with tumour location in the supratentorium, a younger age at diagnosis and a longer time since diagnosis [19]. These results were supported by the finding that posterior fossa tumours disturb the normal development of higher mental functions, especially the development of linguistic and emotional traits [20] and by a study of the developing cognitive profiles of children diagnosed with cerebellar tumours before the age of five [21], which suggested that greater damage to right cerebellar structures are associated with a plateauing in verbal and/or literacy skills. In contrast, greater damage to left cerebellar structures is associated with delayed or impaired non-verbal/ spatial skills [21].
Developmental Outcomes
A recent multicentre, Canadian study provides critical insight into the developmental outcomes in childhood cancer including specific deficits following treatment for brain cancer. In a retrospective cohort study, 800 survivors, age 17 years or younger were matched by age and gender with a group of 923 controls [22]. Significant finding with survivors than controls were found with repeated a grade, attended learning disability or special education programs, had educational or other school problems, had no close friends and were less likely to use friends as confidants. Brain tumour survivors reportedly were more likely than controls to have educational problems and no close friends, followed by survivors of leukemia, and survivors of neuroblastoma. Among survivors, those who had received cranial radiation were more likely to have educational difficulties and to have no close friends than survivors who did not receive cranial radiation [22]. Parent-reported HRQL in child and adolescent cancer survivors was also explored in the same cohort and comparison group [23]. According to parents, the HRQL for survivors was somewhat poorer, overall, than for controls. Survivors of brain tumours, lymphoma, and leukemia and patients treated with cranial radiation had the poorest HRQL [23]. Both of these studies were limited by the exclusive use of parent proxy measures. In a retrospective Canadian cohort study of 2,152 long-term survivors and 2,432 controls, aged 5 to 37, surviving cancer during childhood or adolescence resulted in deficits in dexterity, ambulation, hearing, speech and cognition [24]. Brain tumour survivors were the most likely to show impairments across multiple domains. Impairments in cognition were found most commonly in survivors exposed to craniospinal radiation at young ages [24]. These findings have been replicated in numerous publications using the Childhood Cancer Survivor Study (CCSS), which documents an American cohort of long-term survivors of childhood cancer who were diagnosed between 1970 and 1986, 13% of whom are brain tumour survivors [25,26]. Pediatric brain tumour survivors are at risk for many adverse outcomes related to physical, social, emotional and cognitive function. Patient variables such as tumour location, treatment type (e.g. radiation treatment) and age at treatment may be important variables affecting outcomes.
Chronic Diseases and Physical Activity
Undergoing treatment for cancer can put an individual at risk for developing a number of chronic health conditions which can limit their physical activity. For this reason, cancer is being increasingly conceptualized as a chronic condition. These potential complications which can affect HRQL are particularly important for a young survivor who potentially still has many years of life ahead of them. This issue has been highlighted in many CCSS studies. Chronic diseases identified in the CCSS cohort were found to involve multiple organs and occurred in varying degrees of severity [27,28]. Complications included endocrine and reproductive dysfunction, pulmonary toxicity, cerebrovascular injury, and neurologic and neurosensory sequelae.
Endocrine abnormalities include thyroid disease, growth hormone abnormalities affecting height, and disorders of body weight. Other possible conditions included alterations in pubertal development, osteonecrosis, cardiopulmonary disease and neurologic/neurosensory disorders. All of these effects were disease and treatment specific. High-risk populations have been identified for specific organ toxicity and secondary carcinogenesis including brain tumour and hematologic malignancy survivors [27,28]. Childhood cancer survivors were also found to be at substantial and increasing risk for the development of second neoplasms [29,30]. Late mortality after initial five-year diseasefree survival has been well established, with higher risks of late recurrence noted in the early decades and increasing rates of second malignancies and treatment- related deaths occurring in the later decades [31]. These chronic conditions may be associated with symptoms that may affect a child’s HRQL and ability to remain physically active [32]. Contributing factors that increased the risk for physical performance limitations were musculoskeletal, neurologic, cardiac, pulmonary, sensory, and endocrine organ system dysfunctions [32].
Social Outcomes
Social outcomes have also been studied in the CCSS cohort. Childhood cancer survivors generally had similar high school graduation rates, but required more special education services than sibling controls (33). Survivors were slightly less likely than expected to attend college, and were more likely to be unemployed and single as young adults [33]. HRQL and life satisfaction outcomes were compared in the CCSS cohort with sibling and normative data. A significant percentage of survivors reported more symptoms of global distress and poorer physical but not emotional domains of HRQL [34]. Cranial irradiation affected neurocognitive outcomes, especially in brain tumour survivors. Psychological distress also predicted poor health behaviours, including smoking, alcohol use, fatigue and altered sleep [34].
Pediatric brain tumour survivors have been frequently studied and consistently perform poorly when compared to healthy controls or siblings on a number of HRQL outcome measures including global distress and diminished social functioning [35]. Most of these studies have included adult survivors over the age of 18. These studies highlight the fact that not all cancers are the same with respect to long-term outcomes. Several high-risk groups consistently emerge. It is these high-risk groups that best exemplify the possible detrimental effects of cancer treatment on child development. Brain tumour and acute leukemia childhood cancer survivors have been identified as particularly vulnerable to cognitive, physical, emotional and social developmental issues. Being able to further distinguish the specific symptoms that occur within each of the HRQL domains, can provide caregivers with important information to guide care.
Symptoms in Children with Cancer
Children with cancer experience many symptoms related to their disease and its treatment [1,36-38] which can cause distress [39] and can diminish their HRQL [38]. Symptom assessment is complex and should involve characterizing symptoms based on their intensity, location, temporal nature, frequency and affective effect [40]. Numerous studies have shown that developmental stage as well as verbal and cognitive abilities need to be considered when measuring symptoms in children and adolescents [40- 43]. To develop age-appropriate interventions, it is necessary to understand how children at different developmental stages experience cancer-related symptoms [42]. Previous studies have verified that child self-report is the gold standard for subjective symptom measures [44-46], although parent proxy measures generally correlate positively with child reports [45]. Multidimensional, self-report tools that measure symptoms in children as young as seven years of age, such as the Memorial Symptom Assessment Scale (MSAS), have been shown to have acceptable reliability and validity [37,38,45]. Knowing the most common symptoms and understanding how they affect children’s HRQL, can help care providers tailor their care to meet patients’ individual needs.
An integrative review of symptoms in children with cancer identified 219 distinct symptoms or problems, indicating that children and adolescents experience numerous and complex symptoms [47]. Thirty-six percent of the symptoms identified were psychological or emotional in nature (e.g. anxiety), 31% were physiological (e.g. obesity), 22% were both physical and psychological (e.g. fatigue) and 11% were school-related (e.g. behavioural). The review also identified the importance of health care providers being able to understand the child’s perspective when communicating with a child about his or her symptom experience. In particular, care providers need to consider the verbal and cognitive skills of younger children [47].
Symptoms and HRQL
HRQL assessment in chlidren is an important measure of the impact of the disease, effect of treatment and other variables affecting people's lives (Figure 1). The relationship between symptom experience and HRQL has been explored in children on treatment. Symptom characteristics and HRQL outcomes were explored in 61 patients following the administration of myelosuppressive chemotherapy, including four patients being treated for brain tumours [48]. Patients experienced a mean of 10.6 symptoms. The five most common symptoms were nausea (80%), fatigue (70%), pain (69%), alopecia (66%) and drowsiness (57%). A higher number of symptoms and higher symptom distress scores were associated with poorer HRQL scores [48]. Another systematic review including studies of multiple symptoms in pediatric oncology patients identified nine cross-sectional studies with convenience sampling [38]. Twenty-eight percent to 100% of the patients in these studies were actively receiving treatment. The most commonly occurring symptoms were weight loss or weight gain, fever, sore throat, lack of energy, alopecia, drowsiness, bruising, round face, pain and anorexia. Fatigue, and lethargy or lack of energy was the most frequently reported symptoms in all of the studies. The relationship between demographic and clinical characteristics and the occurrence of multiple symptoms were not elucidated. No studies were identified that examined the relationships between symptom characteristics and HRQL. The authors of the review concluded that more studies are required on the prevalence and effect of multiple symptoms and the link between patient symptoms and outcomes, such as HRQL [38].
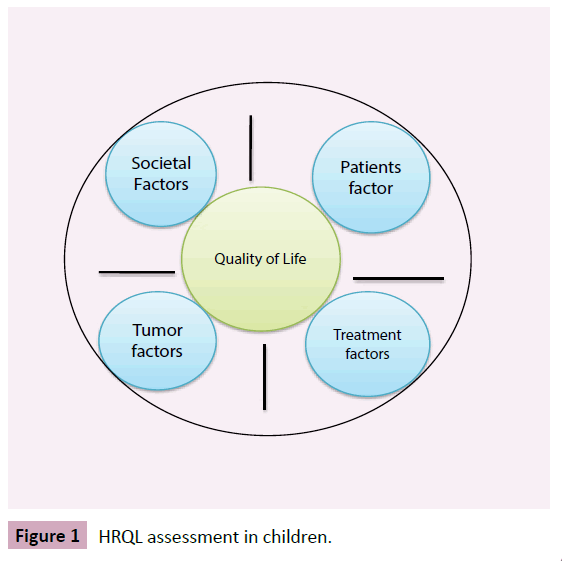
Figure 1 HRQL assessment in children.
Concluding Remarks
Being diagnosed with a brain tumour is a life-changing event, and in children it become complex and represent an important health issues on quality of life. Management of this challenging disease requires a delicate balance between optimizing treatment to cure the disease and prevent recurrence, while minimizing the significant treatment toxicities to the developing brain. Several studies with quality-of-life outcome assessment identified that survivors of pediatric brain tumors have a poor HRQL relative to other cancer survivors or healthy peers across social, emotional, physical and cognitive domains. Validated tools exist to measure symptoms and HRQL in young children, but the symptom experience and the relationship between symptoms and HRQL in children and youth who have completed treatment for a brain tumor are not well understood. Clinicians are accepting that addition of HRQL as both primary and secondary end points play an important role and may in turn increase overall survival. There are currently limited interventions available to enhance quality of life for those affected by a brain tumour. More research is needed in this aspect to increase the survivorship and as patients experience potential morbidities associated with therapies.
References
- Ellison LF, De P, Mery LS, Grundy PE (2009) Canadian Cancer Society's Steering Committee for Canadian Cancer Statistics. Canadian cancer statistics at a glance: cancer in children. CMAJ Canadian Medical Association Journal 180:422-424.
- Maher CO, Raffel C (2004) Neurosurgical treatment of brain tumors in children. Pediatr Clin North Am 51: 327-357.
- Ye X, Wang L, Tse ZT, Tang G, Song G (2015) Effects of high-energy electro-pulsing treatment on microstructure, mechanical properties and corrosion behavior of Ti-6Al-4V alloy. Mater Sci Eng C Mater Biol Appl 49: 851-860.
- Ye X, Wang L, Tse ZTH, Tang G, Song G (2015) Mechanical properties and phase transition of biomedical titanium alloy strips with initial quasi-single phase state under high-energy electropulses. Journal of the Mechanical Behavior of Biomedical Materials. 42: 100-115.
- Smith MA, Freidlin B, Ries LA, Simon R (1998) Trends in reported incidence of primary malignant brain tumors in children in the United States. J Natl Cancer Inst 90: 1269-1277.
- Mainprize TG, Taylor MD, Rutka JT (2000) Perspectives in pediatric neurosurgery. Childs Nerv Syst 16: 809-820.
- Eiser C (2004) Children with cancer: The quality of life. Mahway, New Jersey: Lawrence Erlbaum Associates.
- Bhatia S, Landier W (2005) Evaluating survivors of pediatric cancer. Cancer J 11: 340-354.
- Walter AW, Hilden JM (2004) Brain tumors in children. Curr Oncol Rep 6: 438-444.
- Gottardo NG, Gajjar A (2008) Chemotherapy for malignant brain tumors of childhood. J Child Neurol 23: 1149-1159.
- Berk LE, William RL (2009) Child development. 3rdCanadian edition. ed. Toronto: Pearson Allyn and Bacon.
- Verheyde J, Benotmane MA (2007) Unraveling the fundamental molecular mechanisms of morphological and cognitive defects in the irradiated brain. Brain Res Rev 53: 312-320.
- Peterson CC, Drotar D (2006) Family impact of neurodevelopmental late effects in survivors of pediatric cancer: review of research, clinical evidence, and future directions. Clin Child Psychol Psychiatry 11: 349-366.
- Als H (1982) Toward a synactive theory of development: Promise for the assessment and support of infant individuality. Infant Mental Health Journal 3:229-243.
- Gilkerson L (2001) Integrating an understanding of brain development into early childhood education. Infant Mental Health Journal 22:174-187.
- van den Berg H, Langeveld NE (2008) Parental knowledge of fertility in male childhood cancer survivors. Psychooncology 17: 287-291.
- Grunfeld E, Earle CC (2010) The interface between primary and oncology specialty care: treatment through survivorship. J Natl Cancer Inst Monogr 2010: 25-30.
- Armstrong GT (2010) Long-term survivors of childhood central nervous system malignancies: the experience of the Childhood Cancer Survivor Study. Eur J Paediatr Neurol 14: 298-303.
- Stargatt R, Rosenfeld JV, Anderson V, Hassall T, Maixner W, et al. (2006) Intelligence and adaptive function in children diagnosed with brain tumour during infancy. J Neurooncol 80: 295-303.
- Larysz D, Blamek S, Larysz P, Pietras K, Mandera M (2010) Posterior fossa brain tissue injury: developmental, neuropsychological, and neurological consequences of brain tumors in children. Acta Neurochir Suppl 106: 271-274.
- Scott RB, Stoodley CJ, Anslow P, Paul C, Stein JF, et al. (2001) Lateralized cognitive deficits in children following cerebellar lesions. Developmental Medicine & Child Neurology 43:685-691.
- Barrera M, Shaw AK, Speechley KN, Maunsell E, Pogany L (2005) Educational and social late effects of childhood cancer and related clinical, personal, and familial characteristics. Cancer 104: 1751-1760.
- Speechley KN, Barrera M, Shaw AK, Morrison HI, Maunsell E (2006) Health-related quality of life among child and adolescent survivors of childhood cancer. J Clin Oncol 24: 2536-2543.
- Pogany L, Barr RD, Shaw A, Speechley KN, Barrera M, et al. (2006) Health status in survivors of cancer in childhood and adolescence. Qual Life Res 15: 143-157.
- Leisenring WM, Mertens AC, Armstrong GT, Stovall MA, Neglia JP, et al. (2009) Pediatric cancer survivorship research: experience of the Childhood Cancer Survivor Study. J Clin Oncol 27: 2319-2327.
- Robison LL, Armstrong GT, Boice JD, Chow EJ, Davies SM, et al. (2009) The Childhood Cancer Survivor Study: a National Cancer Institute-supported resource for outcome and intervention research. Journal of Clinical Oncology 27:2308-2318.
- Diller L, Chow EJ, Gurney JG, Hudson MM, Kadin-Lottick NS, et al. (2009) Chronic disease in the Childhood Cancer Survivor Study cohort: a review of published findings. J Clin Oncol 27: 2339-2355.
- Hudson MM, Mulrooney DA, Bowers DC, Sklar CA, Green DM, et al. (2009) High-risk populations identified in Childhood Cancer Survivor Study investigations: implications for risk-based surveillance. J Clin Oncol 27: 2405-2414.
- Friedman DL, Whitton J, Leisenring W, Mertens AC, Hammond S, et al. (2010) Subsequent neoplasms in 5-year survivors of childhood cancer: the Childhood Cancer Survivor Study. J Natl Cancer Inst 102: 1083-1095.
- Meadows AT, Friedman DL, Neglia JP, Mertens AC, Donaldson SS, et al. (2009) Second neoplasms in survivors of childhood cancer: findings from the Childhood Cancer Survivor Study cohort. J Clin Oncol 27: 2356-2362.
- Armstrong GT, Liu Q, Yasui Y, Neglia JP, Leisenring W, et al. (2009) Late mortality among 5-year survivors of childhood cancer: a summary from the Childhood Cancer Survivor Study. J Clin Oncol 27: 2328-2338.
- Ness KK, Hudson MM, Ginsberg JP, Nagarajan R, Kaste SC, et al. (2009) Physical performance limitations in the Childhood Cancer Survivor Study cohort. J Clin Oncol 27: 2382-2389.
- Gurney JG, Krull KR, Kadan-Lottick N, Nicholson HS, Nathan PC, et al. (2009) Social outcomes in the Childhood Cancer Survivor Study cohort. J Clin Oncol 27: 2390-2395.
- Zeltzer LK, Recklitis C, Buchbinder D, Zebrack B, Casillas J, et al. (2009) Psychological status in childhood cancer survivors: a report from the Childhood Cancer Survivor Study. J Clin Oncol 27: 2396-2404.
- Zebrack BJ, Gurney JG, Oeffinger K, Whitton J, Packer RJ, et al. (2004) Psychological outcomes in long-term survivors of childhood brain cancer: a report from the childhood cancer survivor study. J Clin Oncol 22: 999-1006.
- Barsevick AM (2007) The elusive concept of the symptom cluster. Oncol Nurs Forum 34: 971-980.
- Collins JJ, Devine TD, Dick GS, Johnson EA, Kilham HA, et al. (2002) The measurement of symptoms in young children with cancer: the validation of the Memorial Symptom Assessment Scale in children aged 7-12. J Pain Symptom Manage 23: 10-16.
- Baggott C, Dodd M, Kennedy C, Marina N, Miaskowski C (2009) Multiple symptoms in pediatric oncology patients: a systematic review. J Pediatr Oncol Nurs 26: 325-339.
- Hockenberry M1 (2004) Symptom management research in children with cancer. J Pediatr Oncol Nurs 21: 132-136.
- Dodd M, Janson S, Facione N, Faucett J, Froelicher ES, et al. (2001) Advancing the science of symptom management. J Adv Nurs 33: 668-676.
- Hockenberry M, Hooke MC (2007) Symptom clusters in children with cancer. Semin Oncol Nurs 23: 152-157.
- Linder LA (2008) Developmental diversity in symptom research involving children and adolescents with cancer. J Pediatr Nurs 23: 296-309.
- Vatne TM, Slaugther L, Ruland CM (2010) How children with cancer communicate and think about symptoms. J Pediatr Oncol Nurs 27: 24-32.
- Dodd MJ, Miaskowski C (2000) The PRO-SELF Program: a self-care intervention program for patients receiving cancer treatment. Semin Oncol Nurs 16: 300-308.
- Linder LA (2005) Measuring physical symptoms in children and adolescents with cancer. Cancer Nurs 28: 16-26.
- Collins JJ, Byrnes ME, Dunkel IJ, Lapin J, Nadel T, et al. (2000) The measurement of symptoms in children with cancer. J Pain Symptom Manage 19: 363-377.
- Ruland CM, Hamilton GA, Schjødt-Osmo B (2009) The complexity of symptoms and problems experienced in children with cancer: a review of the literature. J Pain Symptom Manage 37: 403-418.
- Baggott CR, Dodd M, Kennedy C, Marina N, Matthay KK, et al. (2011) An evaluation of the factors that affect the health-related quality of life of children following myelosuppressive chemotherapy. Supportive Care in Cancer 19:353-361.