Introduction
Periodontitis is a chronic inflammatory disease prevalent in
most populations, and is characterized by inflamed gingiva and
loss of connective tissue attachment between the tooth and its
surrounding alveolar bone. Chronic bacterial exposure and host
immune- inflammatory response is believed to play an
important role in the destruction of connective tissue and bone,
which are key features in the progression of periodontitis.
Periodontal treatment through the ages has focused on the
reduction of bacterial infection by mechanical removal of
infectious agents. Scaling and Root Planing (SRP) still remains
the “gold standard”; however, it may fail to reduce or eliminate
the anaerobic infection at the base of the pocket, at inaccessible
areas such as the furcation, and within the gingival tissues and
structures inaccessible to periodontal instrumentation such as
sites with probing depths (PDs) >5mm.
Local drug delivery systems act as an effective adjunct to
conventional SRP. It provides controlled release of therapeutic
agents at specific subgingival sites, thus translating into high
concentrations at the target site with reduced dosage, fewer
side effects, fewer applications, and higher patient acceptance
compared to systemic drugs.
For thousands of years, various herbal and natural products
have been used for chemotherapeutic purposes in the field of
medicine and dentistry, with the advantage of minimal side
effects and cost-effectiveness. Aloevera is one such medicinal, a
perennial succulent plant belonging to the Xanthorrhoeaceae
family with anti-inflammatory, antioxidant, antibacterial,
antiviral, antifungal, and immune boosting properties. It also
promotes wound healing by accelerating epithelial cell migration
and collagen maturation, facilitating tissue restoration.
It is member of tree Lily family known as Aloe barbadensis.
Aloe barbadensis consists of two parts which differ completely
in their composition and therapeutic properties. The
parenchymal tissue makes up inner portion of the aloe leaves and produce the aloe vera gel, a clear, thin, tasteless jelly like
material. It also promotes wound healing by accelerating
epithelial cell migration and collagen maturation, facilitating
tissue restoration. Acemannan, a polysaccharide from aloe vera,
has been found to have osteogenic properties. The use of this
herbal product may serve to widen the treatment options for
Chronic periodontitis.
There is limited literature available regarding the use of aloe
vera in the form of chip as a local drug delivery system in
dentistry. Hence, the present study is designed to evaluate the
clinical effects of locally delivered AV chip and compare them
with scaling and root planing in patients with Chronic
Periodontitis.
Aim
The objective of this study was to compare and evaluate the
clinical effects of SRP and aloevera chip; SRP and SRP along with
locally delivered aloevera chip in chronic periodontitis patients
at baseline and 3 months’ post therapy.
Materials and methodology
This randomized controlled split mouth clinical trial involved
15 healthy chronic periodontitis patients aged between 20 and
40 years, who visited the Department of Periodontology,
Rajarajeswari Dental College and Hospital, Bangalore; between
September 2019 and January 2020. An approval was obtained
from the Institutional Ethical Committee (RRDCHET/05PERIO/
2019).
The participants were selected based on the following
inclusion criteria:
1. Chronic periodontitis patients with a pocket depth ≥5mm.
2. Systemically healthy patients
Exclusion criteria were:
1. Smokers and alcoholic patients.
2. Patients allergic to herbal medications.
3. Aggressive periodontitis patients.
4. Subjects on any medication taken within the last 6 months
which may alter the
periodontal status.
5. Pregnant and lactating mothers.
6. Patients who have undergone periodontal treatment within
a period of one year.
All the participants will be explained about the need and
design of the study. Written informed consent for the study will
be obtained from each patient.
Randomization and treatment groups
This randomized split mouth clinical study was carried out on
randomly selected male and female subjects in the age group of 20-40 years with chronic periodontitis having a pocket depth of
≥ 5mm and was categorized as follows:
Group I: Scaling and root planing alone
Group II: Scaling and root planing + aloe vera chip.
Clinical examination to assess the periodontal
condition
Clinical examination was performed on all teeth at baseline,1
month and 3 months. Clinical parameters, such as Gingival index
(GI) (Loe H & Silness P, 1963), Probing Pocket Depth (PPD) and
Clinical Attachment Level(CAL) were recorded.
GI was assessed using a mouth mirror and a probe. PPD was
measured using the Williams graduated periodontal probe from
the gingival margin to the base of the pocket. The
measurements were made to the nearest mm at 6 surfaces
(disto-buccal, disto-lingual, midbuccal, mesio-buccal, mesiolingual,
midlingual) of all teeth present. Clinical attachment level
will be determined from a fixed landmark on individually
fabricated stents using a William’s periodontal probe. The
precise assessment and comparison of the clinical attachment
level at different intervals of time can determine whether the
attachment is being lost, which indicates that the lesion is active.
The exact measurement of this important clinical parameter was
done from the CEJ to the bottom of the pocket and therefore
requires that the location of this landmark be determined
exactly and reproducibly.
Treatment procedures
In all selected patients, a full mouth ultrasonic scaling and
root planing was in a single visit. At the start of the study, oral
hygiene instructions were given and clinical parameters namely
Gingival Index (GI), probing pocket depth (PPD) and clinical
attachment level (CAL) were recorded in all the patients. Root
planing was done with area specific curettes in all the groups
along with ultrasonic scaling. Group II received aloe vera chip
following SRP. Instructions for optimal oral hygiene was
reinforced during each postoperative visit at baseline, 1 month
and 3 months. No additional therapy was performed during
these visits.
Formulation of aloe vera chip
Ingredients are as follows:
Aloe vera- 5%
Hydroxy Propyl Methyl Cellulose- 600mg
Hydroxy Propyl Cellulose- 100mg
Polyethylene Glycol- 50mg
Water- 10ml
10ml of water in a 100ml glass beaker was taken and kept on
a Magnetic Stirrer. Temperature was set to 0 C. A magnetic bead
was added into the glass beaker and RPM was set to 500.
Required quantity of ingredients were weighed. While stirring,
Hydroxy Propyl Methyl Cellulose was added in smaller quantities and RPM was increased to 1000. After addition of Hydroxy
Propyl Methyl Cellulose, Hydroxy Propyl Cellulose and
Polyethylene Glycol was added. Stirring was continued for 2 to 3
hours at an RPM of 1000 to 1500. Further, Aloe vera was added
and stirring was continued. Entire glass beaker contents were
transferred to a petri-dish pre coated with glycerine and kept for
drying at normal room temperature.
Results
The descriptive statistics, including mean, minimum and
maximum values, of the demographic variables of Group 1 and
Group 2 are listed in Table 1 and Table 2. The age of subjects
selected for the study ranged from 20 to 40 years with a mean
age of 29.47 years (Table 1).
|
N |
Minimum |
Maximum |
Mean |
Std. Deviation |
Age |
15 |
20 |
40 |
29.47 |
7.08 |
Table 1: mean age distribution of the subjects
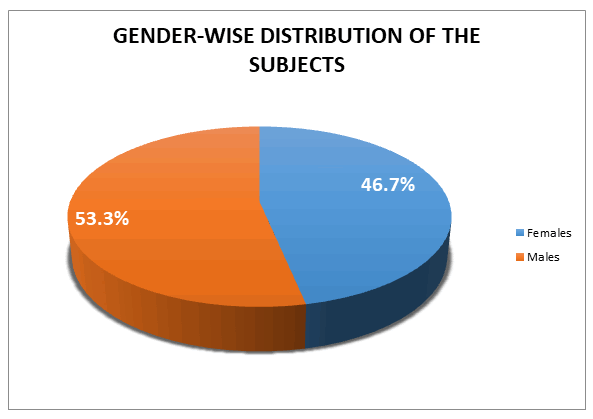
There were a total of 15 participants with 7 females and 8 males in the study (Table 2).
| Gender |
Frequency |
Percent |
| Females |
7 |
46.7 |
| Males |
8 |
53.3 |
| Total |
15 |
100 |
Table 2: gender-wise distribution of the subjects
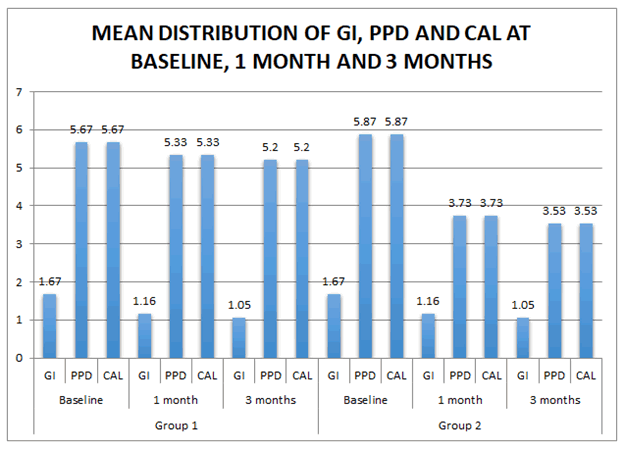
Table 3 describes the mean distribution of GI, PPD and CAL at
baseline, 1 month and 3 months. In Group 1 and Group 2, mean
baseline GI was 1.67 which was reduced to 1.16 at 1 month and
gradually reduced to 1.05 at 3 months. In Group 1, mean PPD
and CAL at baseline was 7 which was reduced to 6 at 1 month
and 3 months whereas in Group 2, mean baseline PPD and CAL
was 7 which was reduced to 6 at 1 month and gradually reduced
to 5 at 3 months.
| Groups |
Time intervals |
Clinical Parameters |
N |
Minimum |
Maximum |
Mean |
S.D |
| Group 1 |
Baseline |
GI |
15 |
1.1 |
2 |
1.67 |
0.25 |
| PPD |
15 |
5 |
7 |
5.67 |
0.62 |
| CAL |
15 |
5 |
7 |
5.67 |
0.62 |
| 1 month |
GI |
15 |
0.7 |
1.6 |
1.16 |
0.24 |
| PPD |
15 |
4 |
6 |
5.33 |
0.62 |
| CAL |
15 |
4 |
6 |
5.33 |
0.62 |
| 3 months |
GI |
15 |
0.7 |
1.4 |
1.05 |
0.21 |
| PPD |
15 |
4 |
6 |
5.2 |
0.56 |
| CAL |
15 |
4 |
6 |
5.2 |
0.56 |
| Group 2 |
Baseline |
GI |
15 |
1.1 |
2 |
1.67 |
0.25 |
| PPD |
15 |
5 |
7 |
5.87 |
0.64 |
| CAL |
15 |
5 |
7 |
5.87 |
0.64 |
| 1 month |
GI |
15 |
0.7 |
1.6 |
1.16 |
0.24 |
| PPD |
15 |
2 |
6 |
3.73 |
1.1 |
| CAL |
15 |
2 |
6 |
3.73 |
1.1 |
| 3 months |
GI |
15 |
0.7 |
1.4 |
1.05 |
0.21 |
| PPD |
15 |
2 |
5 |
3.53 |
0.83 |
| CAL |
15 |
2 |
5 |
3.53 |
0.83 |
Table 3: mean distribution of gi, ppd and cal at baseline, 1 month and 3 months
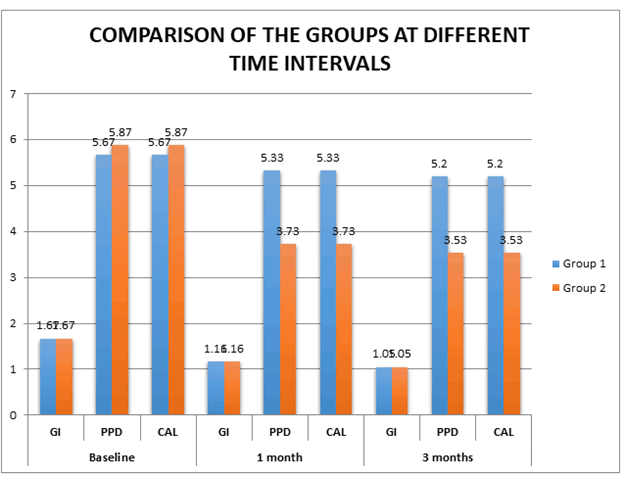
Table 4 describes comparison of GI, PPD and CAL between the
groups at different time intervals. At baseline, with respect to GI,
there was no significant difference seen between the groups.
There was no significant difference seen with respect to GI, PPD
and CAL at baseline. At 1 month, there is a significant difference
between PPD and CAL but there is no significant difference in GI
between the groups. Greater reduction of PPD and CAL is seen
in Group 2 at 3 months when compared to Group 1.
| Time intervals |
Clinical parameters |
Groups |
N |
Mean |
S.D |
Mean diff |
t value |
p value |
| Baseline |
GI |
Group 1 |
15 |
1.67 |
0.25 |
0 |
0 |
1 |
| Group 2 |
15 |
1.67 |
0.25 |
| PPD |
Group 1 |
15 |
5.67 |
0.617 |
-0.2 |
-0.87 |
0.39 |
| Group 2 |
15 |
5.87 |
0.64 |
| CAL |
Group 1 |
15 |
5.67 |
0.617 |
-0.2 |
-0.87 |
0.39 |
| Group 2 |
15 |
5.87 |
0.64 |
| 1 Month |
GI |
Group 1 |
15 |
1.16 |
0.235 |
0 |
0 |
1 |
| Group 2 |
15 |
1.16 |
0.235 |
| PPD |
Group 1 |
15 |
5.33 |
0.617 |
1.6 |
4.91 |
0.00* |
| Group 2 |
15 |
3.73 |
1.1 |
| CAL |
Group 1 |
15 |
5.33 |
0.617 |
1.6 |
4.91 |
0.00* |
| Group 2 |
15 |
3.73 |
1.1 |
| 3 Months |
GI |
Group 1 |
15 |
1.05 |
0.213 |
0 |
0 |
1 |
| Group 2 |
15 |
1.05 |
0.213 |
| PPD |
Group 1 |
15 |
5.2 |
0.561 |
1.66 |
6.42 |
0.00* |
| Group 2 |
15 |
3.53 |
0.834 |
| CAL |
Group 1 |
15 |
5.2 |
0.561 |
1.66 |
6.42 |
0.00* |
| Group 2 |
15 |
3.53 |
0.834 |
Table 4: comparison of the groups at different time intervals using independent sample t test
Table 5 describes comparison of the clinical parameters within
the group among different time intervals. There was a significant
difference in GI, PPD and CAL within the groups for both the
groups.
| Clinical Parameters |
Groups |
Repeated measures value |
p value |
| GI |
Group 1 |
102.5 |
0.00* |
| Group 2 |
102.5 |
0.00* |
| PPD |
Group 1 |
8.27 |
0.003* |
| Group 2 |
108.26 |
0.00* |
| CAL |
Group 1 |
8.27 |
0.003* |
| Group 2 |
108.26 |
0.00* |
Table 5: comparison of the clinical parameters within the group among different time intervals using repeated measures anova
There was a significant difference seen from baseline to 1
month and 3 months with respect to GI in group 1 and group 2.
But, from 1 month to 3 months there was no significant
difference with respect to GI in group 1. From baseline to 1
month and baseline to 3 months, there was a significant
difference in PPD and CAL in Group 2 and no significant
difference seen with respect to PPD from 1 month to 3 months
in Group 2. Conversely there was no significant difference in PPD
and CAL at baseline to 1 month and 1 month to 3 months. But
there was a significant difference with respect to PPD and CAL at
baseline to 3 months in both Group 1 and Group 2. (Table 6)
| Clinical parameters |
Time intervals |
Group 1 |
Group 2 |
| Mean Difference |
p value |
Mean Difference |
p value |
| GI |
Baseline Vs 1 month |
0.513 |
.000* |
0.513 |
.000* |
| Baseline Vs 3 months |
0.62 |
.000* |
0.62 |
.000* |
| 1 month Vs 3 months |
0.107 |
.037* |
0.107 |
.037* |
| PPD |
Baseline Vs 1 month |
0.333 |
0.058 |
2.133 |
.000* |
| Baseline Vs 3 months |
0.467 |
.011* |
2.333 |
.000* |
| 1 month Vs 3 months |
0.133 |
0.493 |
0.2 |
0.247 |
| CAL |
Baseline Vs 1 month |
0.333 |
0.058 |
2.133 |
.000* |
| Baseline Vs 3 months |
0.467 |
.011* |
2.333 |
.000* |
| 1 month Vs 3 months |
0.133 |
0.493 |
0.2 |
0.247 |
Table 6: post hoc bonferroni
Table 7 shows mean percentage reduction within the groups.
There is mean percentage reduction in both the groups. With
respect to GI, there is -30.84% reduction from baseline to 1
month and -8. 44% from 1 month to 3 months in both Group 1
and Group 2. With respect to both PPD and CAL, there is -5.62%
reduction from baseline to 1 month and -2.22% from 1 month to
3 months in Group 1 whereas in Group 2 there is mean
percentage reduction of -36.86% from baseline to 1 month and
-3.78% from 1 month to 3 months.
| Groups |
Clinical parameters |
Time intervals |
N |
Minimum reduction (%) |
Maximum reduction (%) |
Mean (%) |
S.D (%) |
| Group 1 |
GI |
Baseline to 1 month |
15 |
-16.67 |
-43.75 |
-30.84 |
7.9 |
| Baseline to 3 months |
15 |
0 |
-20 |
-5.62 |
8.3 |
| 1 month to 3 months |
15 |
0 |
-33.33 |
-8.44 |
11.14 |
| PPD |
Baseline to 1 month |
15 |
0 |
-20 |
-5.62 |
8.3 |
| Baseline to 3 months |
15 |
0 |
-20 |
-7.84 |
8.75 |
| 1 month to 3 months |
15 |
0 |
-16.67 |
-2.22 |
5.86 |
| CAL |
Baseline to 1 month |
15 |
0 |
-20 |
-5.62 |
8.3 |
| Baseline to 3 months |
15 |
0 |
-20 |
-7.84 |
8.75 |
| 1 month to 3 months |
15 |
0 |
-16.67 |
-2.22 |
5.86 |
| Group 2 |
GI |
Baseline to 1 month |
15 |
-16.67 |
-43.75 |
-30.84 |
7.9 |
| Baseline to 3 months |
15 |
-14.29 |
-60 |
-36.86 |
14.93 |
| 1 month to 3 months |
15 |
0 |
-33.33 |
-8.44 |
11.14 |
| PPD |
Baseline to 1 month |
15 |
-14.29 |
-60 |
-36.86 |
14.93 |
| Baseline to 3 months |
15 |
-16.67 |
-60 |
-39.87 |
12.29 |
| 1 month to 3 months |
15 |
0 |
-20 |
-3.78 |
7.85 |
| CAL |
Baseline to 1 month |
15 |
-14.29 |
-60 |
-36.86 |
14.93 |
| Baseline to 3 months |
15 |
-16.67 |
-60 |
-39.87 |
12.29 |
| 1 month to 3 months |
15 |
0 |
-20 |
-3.78 |
7.85 |
Table 7: mean percentage reduction or increase within the groups
Discussion
Periodontal disease is an inflammatory disease of the
periodontium usually caused by pathogenic microflora in the
biofilm or dental plaque that forms on the tooth surfaces.
Treatment of periodontal diseases by different types of local
delivery systems has been investigated. Mouthwashes and
irrigating agents such as Chlorhexidine, sodium hypochlorite,
cetylpyridinium chloride and amine fluoride are widely used that
can inhibit the development of potentially pathogenic bacteria.
Although these antimicrobial agents are widely used,
immediate hypersensitivity reactions, toxicity, tooth staining and
other side effects have been reported. Moreover, it has been
reported that chlorhexidine on long term usage and sodium
hypochlorite are cytotoxic to human periodontal ligament cells, inhibit protein synthesis, and affect mitochondrial activity, thus
having detrimental effects on vital tissues.
The natural phytochemicals isolated from medicinal plants
used in traditional medicine have been considered useful
alternatives to synthetic drugs. Many medicinal plants and their
products are widely used for prevention and treatment of oral
diseases, and among them Aloe vera is of particular interest and
has been used therapeutically for a long time.
Aloe vera is a natural product contained in herbal dentifrices
with commercial appeal on the control of plaque and gingivitis.
Aloe latex contains anthraquinones, and enzymes bradykinase,
which are chemical compounds that are used in healing and
arresting pain because they are antiinflammatory in nature. Aloe
vera inhibits the cyclooxygenase pathway and reduces
prostaglandin E2 production from arachidonic acid. Also, Aloe
vera contains 6 antiseptic agents: Lupeol, salicylic acid, urea
nitrogen, cinnamonic acid, phenol and sulfur. They all have
inhibitory action on fungi, bacteria and viruses.
In the present study, the clinical efficacy of AV chip for the
treatment of chronic periodontitis with a pocket depth of
5-6mm was evaluated as an adjunct to SRP. There was a
significant improvement in the clinical parameters in both the
groups, which can be attributed to the elimination of local
etiological factors through mechanical debridement.
The present study considers local drug delivery of aloevera
chip in the periodontal pockets of patients with CP because it
offers advantages of high concentrations at the target site with a
reduced dosage compared to a systemic regimen. LDD could
offer important benefits in terms of adverse reactions and
patient compliance.
The present study found a significant reduction in mean PPD
and gain in clinical attachment level (CAL), in alovera group,
which can be attributed to antibacterial and anti- inflammatory
properties.
Oliveria et al (2008) also reported significant reduction in
plaque and gingivitis with use of dentrifice containing Aloe vera.
Dilip et al demonstrated that Aloe vera was as effective against
Candida albicans, Streptococcus mutans, Lactobacillus
acidophillus, Entrococcus fecalis, Provetella intermedia and
Peptostreptococcus anaerobius.
Aloevera has strong immunomodulatory activity wherein it
downregulates lipopolysaccharide- induced inflammatory
cytokine production and the expression of inflammasome in
human macrophages.
Aloevera also accelerates collagen synthesis and promotes
wound healing, which can account for the significant PD
reduction and CAL gain. The findings of this study correlated
with those of Pradeep et al. and Abdelmonem et al. Acemannan,
a major component of AV, showed significantly- accelerated new
alveolar bone and cementum and periodontal ligament
formation in class II furcation defects.
The mean reduction in GI, PPD and the mean CAL gain were
significantly greater in the Aloevera group at 1 to 3months
interval, suggesting long- term benefits of this local delivery system. These findings are in commensurate with those of
Abdelmonem et al., who found AV LDD as an adjunct to
mechanical periodontal therapy to be associated with significant
improvement in microbiologic as well as clinical outcomes in
patients with CP.
Considering these facts, local drug delivery of aloevera chip as
an adjunct to SRP can be proposed as a better approach for
treatment of periodontal pockets in chronic periodontitis
patients.
Conclusion
Under the experimental conditions, this clinical trial demonstrated that Aloe vera chip in the form of local drug delivery ensued a significant pocket depth reduction and clinical attachment level gain compared to scaling and root planing in chronic periodontitis patients. The results of the present study suggested that the local drug delivery of Aloe vera chip improved the periodontal status. However long term, multicentre randomized, controlled clinical trials are required to know its clinical histologic effect on periodontal tissues in patients with chronic periodontitis.
Competing and conflicting interests:
No conflicts of interest
References
- Priyanka N, Kalra N, Saquib S, Kudyar N, Nikhil Malgaonkar N, Jain H. Clinical and microbiological efficacy of 3% satranidazole gel as a local drug delivery system in the treatment of chronic periodontitis: A randomized, controlled clinical trial. Contemp Clin Dent.2015; 6(3): 364–370.
- NagasreeM, Madhuri P B, Musalaiah S V V S, Kumar A, Indeevar P. Efficacy of ornidazole gel as an adjunct to scaling and root planing in chronic Periodontitis patients: a clinical and microbiological study. Journal of Biomedical and Pharmaceutical Research. 2016; 5(6).
- Adriaens PA, De Boever JA, Loesche WJ. Bacterial invasion in root cementum and radicular dentin of periodontally diseased teeth in humans. A reservoir of periodontopathic bacteria. J Periodontol 1988; 59:222-30.
- https://aap.onlinelibrary.wiley.com/doi/abs/10.1902/jop.1998.69.5.507
- Bansal K, Rawat MK, Jain A, Rajput A, Chaturvedi TP, Singh S. Development of satranidazole mucoadhesive gel for the treatment of periodontitis. AAPS PharmSciTech. 2009; 10(3):716-23.
- Silness, J. and Löe, H.: Periodontal disease in pregnancy. II. Correlation between oral hygiene and perio-dontal condition. Actaodont. Scand., 22:112-135, 1964.
- Löe, H. and Silness, J.: Periodontal disease in pregnancy. I. Prevalence and severity. Actaodont. Scand., 21:533-551, 1963.
- Mühlemann HR, Son S. Gingival sulcus bleeding--a leading symptom in initial gingivitis. Helv Odontol Acta. 1971; 15: 107-113.