Keywords
Heavy metals; Soku oil field; Niger delta; Aluminium; Enrichment factor
Introduction
Soku oil field and the associated Gas Plant are the major oil field facilities discovered and exploited after Oloibiri oil field in the Niger Delta of Nigeria. The Soku oil facility supplies the bulk of the gas to Nigeria Liquefied Natural Gas (NLNG) at Finima, Bonny for export [1]. The oil field and the Gas plant are surrounded by a number of tidal estuarine water bodies like the Sombrero and Saint Bartholomew estuaries. With the increase in Oil exploration and production, the incidences of oil spills have equally increased considerably within the Niger Delta area. Spills occurred accidentally and through the deliberate actions of some local people who sabotaged pipelines to steal oil or protested against the operations of the Federal government and oil companies. Available records showed that a total of 6,817 oil spills occurred between 1976 and 2001 with a loss of approximately three million barrels of oil [1,2]. Environmental pollution from crude oil/oil spillages had become critical and a major topic of popular concern worldwide as it is known to affect public health. Industrial effluents contaminated land, swamps, rivers, estuaries and coastal waters. Such anthropogenic pollutants were the main sources of heavy metal contaminants in aquatic environment [3].
Metals which occur naturally in the earth’s crust are released to the soil and the hydrologic cycle during physical and chemical weathering of igneous and metamorphic rocks. The background concentrations of these elements are mainly controlled by the geologic characteristics of the watershed [4].The essential trace (heavy) metals at low concentrations are micro-nutrients and as such are essential for life but become toxic at high concentrations. Some heavy metals as inorganic elements are essential for plant growth in traces or very minute quantities but toxic and poisonous in relatively higher concentrations. Lead (Pb) and cadmium (Cd) are non-essential, hence exposure to these may lead to several health implications. In natural aquatic ecosystems these metals occur in low concentrations usually in Nano grams to microgram per litre. The occurrence of these heavy metal contaminants especially in excess of natural levels in recent times has become a problem of increasing concern especially as high heavy metal levels in humans have been traced back to the high concentrations of heavy metals in aquatic resources [5].
The natural sources of heavy metals include coastal supply, deep sea supply which by-passes the near shore environment and includes metal transported in the atmosphere as dust particles (or as vapour in the case of mercury) and also materials which are produced by glacial erosion and transported by floating ice in polar regions [6]. Though Turekian’s investigations are particular for the marine environment, rivers share similar characteristics such as the product of mechanical and chemical weathering of rocks. It seems probable that continental weathering plays a major part in determining the heavy metal composition of large rivers [6]. Concentrations of metals in many so-called uncontaminated rivers may not be strictly natural because the rate of weathering has been accelerated by agriculture [7]. Heavy metals in aquatic environment through human activities include industrial and municipal waste products, rural and urban water runoff, fine sediments eroded from catchments, atmospheric deposition (e.g. burning of fossil fuels, incineration of waste and industrial emissions) and petroleum industry [8,9]. Heavy metals such as Nickel (Ni), Mercury (Hg), copper (Cu), Iron (Fe), manganese (Mn), Pb and Cd are found in fuel and antifouling paints from ships [10]. Metal contamination tends to be localized to areas situated in close proximity to an industrial activity, installations or large cities [11]. Heavy metals in the aquatic environment are partitioned among various aquatic environmental compartments viz: Soluble (water) phase, suspended solids, bottom sediments and biota and the main processes governing their distribution and partitioning include dilution, adsorption, advection, dispersion, precipitation, sedimentation and biological uptake [4]. In tide dominated water ways such as deltas, estuaries and tidal creeks, flanking environment are the main traps for fine sediments and these include mangroves, salt marshes and inter-tidal flats [12- 14].The aim of this study was to determine the levels of selected heavy metals (Cd, Al, Fe, V and Pb) in water and sediments in Soku oil field and compare obtained values with the World Health Organization’s recommended limits.
Materials and Methods
Study area
Soku Oil field is situated between the co-ordinates of 04º61’21.9’’E and 07º28’58.08’’N and 04º63’ 52.21’’E and 07º20’ 48.34’’ in Soku, Akuku toru local government area of rivers state of Nigeria. The Soku oil field is a host of several oil and gas industrial facilities operated by petroleum development companies in the area. These facilities include gas and oil wells, flow stations, gas plant, gas flaring sites and gas and oil pipelines. The oil field has several small and medium settlements such as Elem-Sangama, Etukukiri, Russiakiri, Ijawkiri, Ekulama and others which are separated by smaller creeks. The area has suffered long term impacts of accidental discharges and spillages of hazardous substances (industrial wastes and effluents, raw materials, chemicals, crude oil and refined petroleum products). Dredging activities and out board engine boat transportation also occur in the area. The vegetation is typically a mangrove intertidal forest with patches of fresh water forests existing in between. The major occupation of the rural dwellers is fishing (Figure 1).
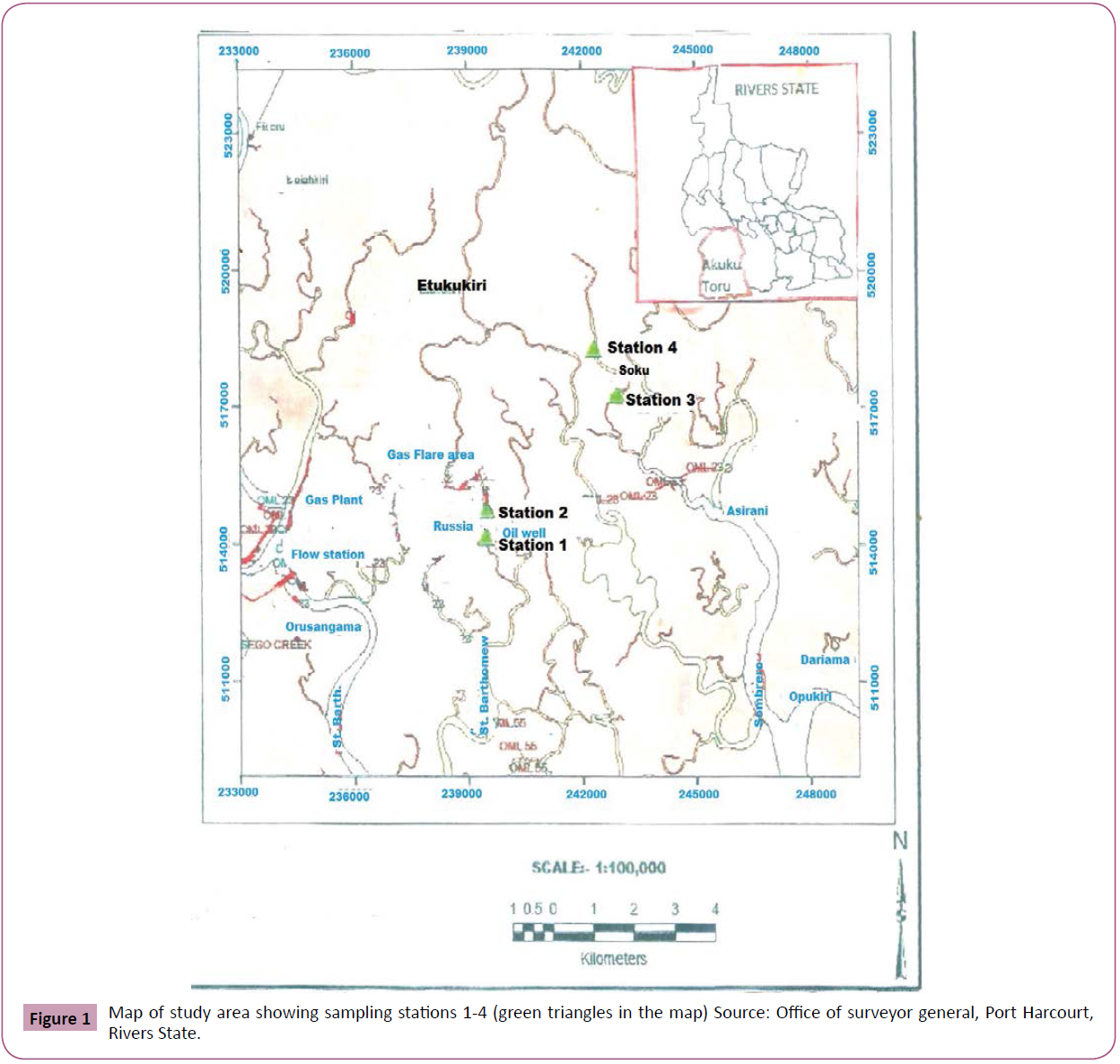
Figure 1: Map of study area showing sampling stations 1-4 (green triangles in the map) Source: Office of surveyor general, Port Harcourt, Rivers State.
Sampling stations and design
Four sampling stations (stations 1 and 2 from the tributaries of San Bartholomew and stations 3 and 4 from Sombrero River) along the Soku oil and gas plant area were established on the basis of up and down streams of the point source which is the Soku oil field. Station 1 situated at the co-ordinates of 4.6529ºN and 6.66648ºE was behind a multi-national Company’s platform (oil well No. 015 and 037) with oil and gas pipe lines. The leaks from pump valves on the oil wells and precipitation of flared gases are main sources of anthropogenic pollutants on the downstream flow. Station 2 with co-ordinates 4.6633ºN and 6.65979ºE, 1 km away from Station 1 on the up-stream direction, is at a rural settlement called Russia camp where sources of pollutants include runoffs from surrounding oil wells, domestic and human wastes and the precipitation of flared gases. Station 3 with co-ordinate 4.68135ºN and 6.68059ºE, 1.5 km away from station 1 is by Soku Community sand fill with dredging activities, domestic wastes and untreated sewage from human as well as out board engine boat transportation activities as sources of pollution. Station 4 with co-ordinates 4.7014ºN and 6.67727ºE, a distance of 2 km away from the community with minimal human activities served as control. The surface water and sediments for this study were sampled bimonthly for one year at the respective stations.
Collection of surface water sampling
Plastic containers measuring 1000 ml were used to collect waist deep surface water for laboratory analysis in replicates. The sampling bottles/containers were washed and rinsed twice with the water at specific sampling locations before the collection of the samples. The samples were collected by lowering the containers by hand to about 20 cm below the surface level. The bottles/containers were completely immersed before opening, allowed to fill and corked (stopped) under water. The surface water samples were acidified with 5 ml of concentrated nitric acid (69% pure produced by Lobachemie) in the 1000 ml container [15].Physicochemical parameters (temperature, pH, dissolved oxygen DO, turbidity and salinity) were measured using standard procedures [15].
Laboratory analysis of heavy metals in surface water
Heavy metals (Cd, Pb, Fe, Al and V) in the surface water were determined by the use of Atomic Absorption Spectrophotometer [(AAS) (model: AANALYST 400 Perkin Elmer)]. The surface water sample collected was filtered using Watchman No 1 filter paper (Diameter: 8.5 cm and Pore size 11 μm) and the supernatant collected for AAS analysis. The concentration of individual metal was determined by aspirating a portion of the filtered sample directly with no pre-treatment using the methods described in previous study [15].
Collection of sediments
Sediments samples were collected randomly from three spots per location per sampling day. The method of collection used was destructive sampling of the littoral zone. The sediments were excavated using a scooper to a depth of 4-5 cm and collected into plastic buckets. The sample was thoroughly mixed forming 1:1 sediment to water slurry and some representative quantities sub sampled into a labeled polythene bag. pH and temperature were measured using a pH meter and a thermometer. The sediments were air dried at room temperature in the laboratory, crushed to fine texture in a porcelain mortar, repackaged in labeled polythene bags and stored in the laboratory for heavy metal analysis
Laboratory analysis heavy metals in sediments
From the air dried sediment samples, 4 g was measured into a 200 ml boro-silicate conical flask. 50 ml of distilled water was added and stirred to mix. Then 12 ml of concentrated hydrochloric acid and 10 ml of concentrated nitric acid were added with some anti pumping chips to avoid spill. The mixture was placed in a hot water bath in a fume chamber to evaporate gradually to half the original volume (25 ml). It was then allowed to cool and filtered with Watchman No 1 filter paper. The supernatant was diluted to 100 ml by the addition of distilled water and taken for AAS analysis as in surface water [15].
Statistical analysis
The following statistical analyses were adopted to interpret the results of this study.
Test to compare stations and months (ANOVA), separation of means and Enrichment factor (EF) of sediment. Enrichment Factor was calculated with the application of the original formula introduced by Buat-Menard and later adopted by Likuku [16,17]:
EF=(Cm/Cref)/(Bm/Bref)
Where,
EF=Enrichment Factor, Cm=Contamination of examined metal in the sediment of the study area. Cref=Background concentration of the examined metal in sediment. Bm=Concentration of reference metal in the sediment of the study area. Bref=Background concentration of the reference metal in sediment [18].
Results
Heavy metal concentrations in surface water
The mean, range and standard error of the heavy metals in mg/L and the maximum permissible limit of heavy metal concentrations in water are presented in Table 1. Mean concentrations Cd, Pb, Fe and V in the study area were all above the permissible limits [19-23].
| Parameters |
Mean |
Range |
Standard Error (SE) |
Standard Limits |
Source |
| Cadmium (mg/L) |
0.01 |
0.00-0.10 |
0.01 |
0.001 |
USEPA [19] |
| Lead (mg/L) |
0.64 |
0.03-2.055 |
0.11 |
0.04 |
FMEnv, USEPA [20,21] |
| Iron (mg/L) |
1.95 |
0.026-9.606 |
0.49 |
0.3 |
IRIS, FAO/WHO [22,23] |
| Aluminium (mg/L) |
0.88 |
0.00-6.540 |
0.34 |
1 |
USEPA, FAO/WHO [21,24] |
| Vanadium (mg/L) |
0.08 |
0.001-0.410 |
0.03 |
0.009 |
IRIS, FAO/WHO [22,23] |
Table 1 Means and standard error of heavy metals (Cd, Pb, Fe, Al and V) in surface water.
Figure 2 indicate that during the wet season (May-July), Cd ranged from 0.00-0.10 mg/L with highest value at station 1 (downstream station) recorded in May, while Cd concentrations were below detectable limits at stations 1,2,3 and 4 (Table 2).
| Parameters |
July, 2017 |
Sept., 2017 |
Nov, 2017 |
Jan, 2018 |
March, 2018 |
May, 2018 |
| Cd (mg/L) |
0.00b |
0.01b |
0.01b |
0.01b |
0.01b |
0.05a |
| Pb (mg/L) |
0.04c |
0.12c |
0.49bc |
1.07a |
0.90ab |
1.22a |
| Fe (mg/L) |
2.89b |
6.01a |
1.27bc |
0.22c |
0.21c |
1.07bc |
| Al (mg/L) |
2.87a |
2.39a |
0.00b |
0.00b |
0.01b |
0.00b |
| V (mg/L) |
0.26a |
0.21a |
0.01b |
0.00b |
0.01b |
0.01b |
Superscript along row that are similar are not significantly different at p=0.05. ‘a’ is not different from ‘ab’ but is different from’ b’, ‘c’
Table 2 Mean values of heavy metals off or all stations for surface water in study area from respective sampling period.
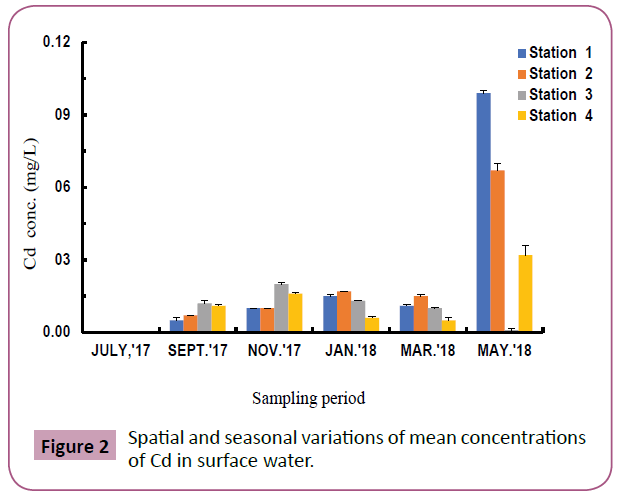
Figure 2: Spatial and seasonal variations of mean concentrations of Cd in surface water.
Figure 3 confirms the presence of Pb in surface water which ranged from 0.03-2.06 mg/L with highest value at station 4 (Upstream station) during the wet season (May) and the lowest values at stations 2, 3 and 4in the wet season month of July.
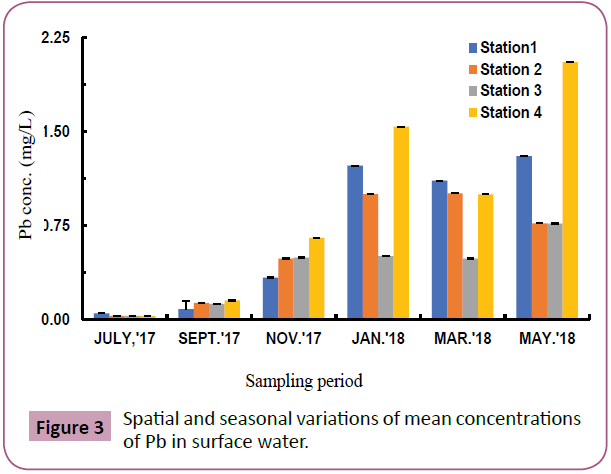
Figure 3: Spatial and seasonal variations of mean concentrations of Pb in surface water.
Fe: Fe concentrations in surface water ranged from 0.03-9.61mg/ L with highest values recorded at station 1 during the wet season (September) and the lowest values at station 3 during the dry season (January). The levels in September were significantly different from other months and July was significantly different from January and March but not with November and May (p<0.05) (Figure 4).
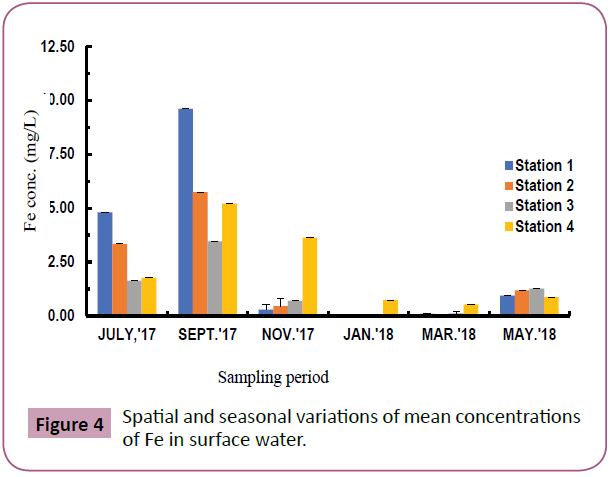
Figure 4: Spatial and seasonal variations of mean concentrations of Fe in surface water.
Aluminium (Al): Figure 5 shows the spatial and seasonal variations of mean concentrations of Al in surface water which ranged from BDL (Below Detection Limit)-6.54 mg/L with highest value at station 1 during the wet season (July). Generally, Al recorded higher concentrations in wet season than dry season. Spatial variations of Al concentrations showed no significant difference among sampling stations at p<0.05.
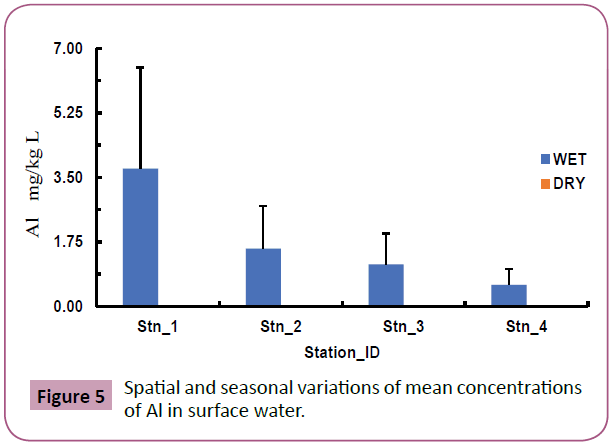
Figure 5: Spatial and seasonal variations of mean concentrations of Al in surface water.
Vanadium (V): Figure 6 shows the spatial and seasonal variation of V which ranged from 0.001-0.410 mg/L. Highest value of 0.410 mg/L was recorded at station 3 during the wet season (July). V in surface water was generally higher in the wet season and lower in the dry season. The variations were not significant among sampling stations at p<0.05. In comparison, levels of V like those of Al indicated no significant variations between July and September but were significantly different from May, November, March and January at P<0.05.
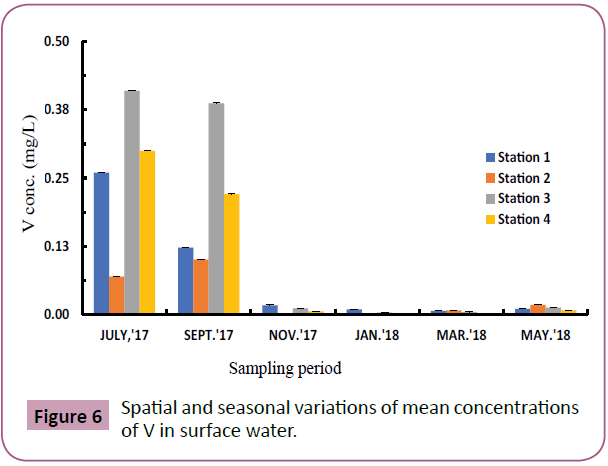
Figure 6: Spatial and seasonal variations of mean concentrations of V in surface water.
Physicochemical parameters of surface water
Temperature ranged between 27.11ºC and 31.80ºC with mean value of 29.10 ± 0.30ºC.pH varied from 6.31 to 7.16 (slightly acidic to slightly alkaline).The salinity of water ranged from 0.79-9.95% with the mean of 3.55 ± 0.51%. The DO concentrations ranged from 2.18-6.63 mg/L with mean value of 4.37 ± 0.30 mg/L. The mean values are presented in Table 3.
| Parameters |
Station 1 |
Station 2 |
Station 3 |
Station 4 |
| Temperature (ºC) |
30.56a |
28.24b |
28.99b |
28.62b |
| pH |
6.71ab |
6.65b |
6.83a |
6.77ab |
| Salinity (mg/L) |
2.25b |
2.30b |
5.00a |
4.65a |
| DO (mg/L) |
4.27a |
4.56a |
4.14a |
4.50a |
| BOD (mg/L) |
26.56a |
25.62a |
26.50a |
26.45a |
Superscript along row that are similar are not significantly different at p=0.05. ‘a’ is not different from ‘ab’ but is different from’ b’, ‘c’
Table 3 Mean of physico-chemical parameters of surface water of the sampling stations.
Heavy metals concentration in sediment
The heavy metal concentrations (mg/kg) in sediment ranged from 0.00-6.05 for Cd, from 0.06-8.76 for Pb, from 78.23-18023.40 for Fe, from 0.003-10.79 for Al and from 0.017-18.43 for V. There was no significant difference at p<0.05 for all the heavy metals across the sampling stations. Seasonal differences were significant. The concentrations of all the heavy metals were above permissible limits [19,20,22,24,25]. As shown in table 5 (Tables 4 and 5).
| Parameters |
July, 2017 |
Sept, 2017 |
Nov, 2017 |
Jan, 2018 |
March, 2018 |
May, 2018 |
| Cd (mg/kg) |
0.01d |
0.76cd |
4.18a |
2.43bc |
1.51cd |
4.06ab |
| Pb (mg/kg) |
0.17c |
0.41c |
6.42a |
3.90b |
3.28b |
2.85b |
| Fe (mg/kg) |
261.98c |
476.31c |
12695.04a |
6269.17b |
6293.81b |
13655.48a |
| Al (mg/kg) |
8.64a |
6.13b |
0.38c |
0.01c |
0.08c |
0.01c |
| V (mg/kg) |
6.96a |
4.29ab |
0.04b |
0.03b |
0.02b |
0.05b |
Superscript along row that are similar are not significantly different at p=0.05. ‘a’ is not different from ‘ab’ but is different from’ b’, ‘c’
Table 4 Mean values of heavy metals of sediment from respective sampling period.
| Parameters |
Mean |
Standard Error (SE) |
Standard Limit |
Reference |
| Temperature (ºC) |
28.44 |
0.17 |
?40 |
FMEnv [20] |
| pH |
6.14 |
0.12 |
6-9.00 |
FMEnv [20] |
| Cd (mg/kg) |
2.16 |
0.39 |
0.03-0.3 |
FMEnv, DPR [20,25] |
| Pb (mg/kg) |
2.84 |
0.52 |
0.5 |
FMEnv, DPR [20,25] |
| Fe (mg/kg) |
6608.63 |
1230.19 |
20 |
FMEnv, DPR [20,25] |
| Al (mg/kg) |
2.54 |
0.75 |
1 |
USEPA, FAO/WHO [21,24] |
| V (mg/kg) |
1.9 |
0.88 |
0.009 |
IRIS, FAO/WHO [22,23] |
Table 5 Descriptive analysis of physico-chemical parameters and means of heavy metals in sediment.
Figures 7 to 11 shows the spatial and seasonal variations of the mean concentrations for Cd, Pb, Fe, Al and V in sediments of the study area. Concentration of Cd fluctuated throughout the study period but highest values were recorded in station 3 in the month of November and lowest values in July (wet season). Similar trends were observed for Pb and Fe and an opposite trend for Al and V.
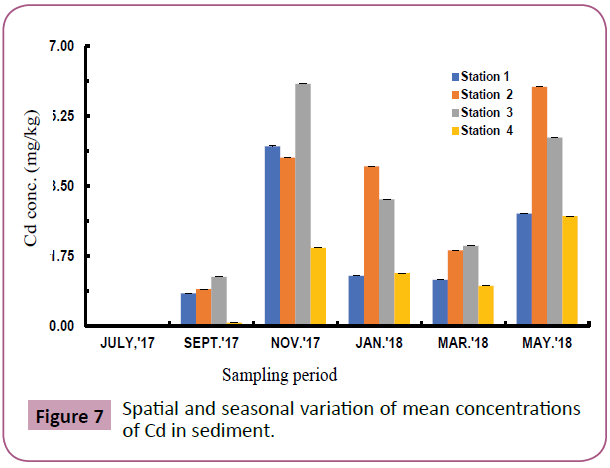
Figure 7: Spatial and seasonal variation of mean concentrations of Cd in sediment.
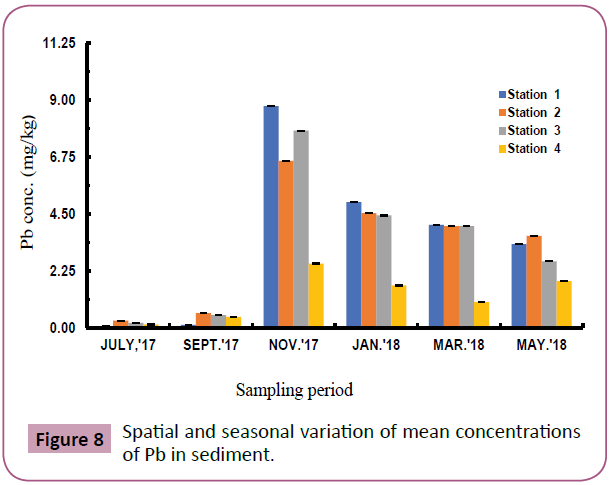
Figure 8: Spatial and seasonal variation of mean concentrations of Pb in sediment.
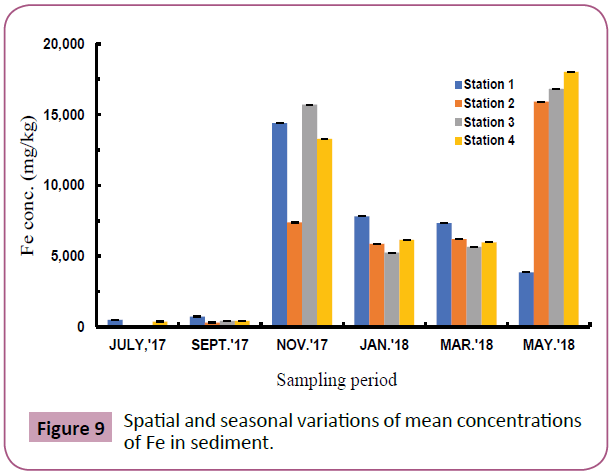
Figure 9: Spatial and seasonal variations of mean concentrations of Fe in sediment.
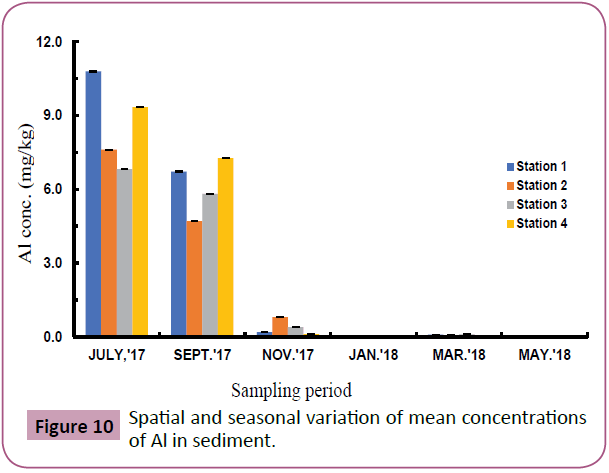
Figure 10: Spatial and seasonal variation of mean concentrations of Al in sediment.
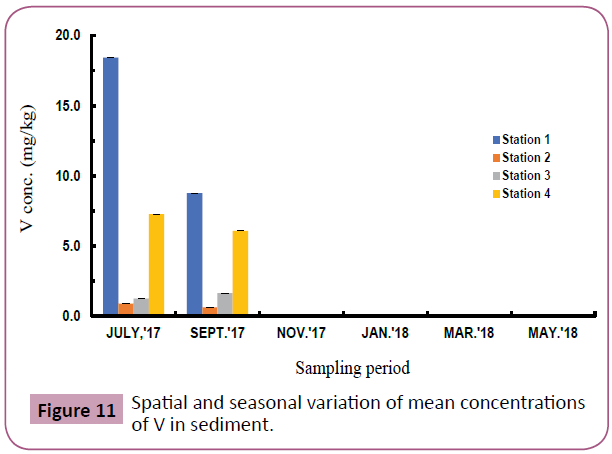
Figure 11: Spatial and seasonal variation of mean concentrations of V in sediment.
Sediment enrichment factor
Table 6 shows the heavy metal enrichment factor of selected metals in the study area, which were assessed through the application of Enrichment Factor using Iron (Fe) as a reference element. V and Al showed minimal enrichment, Pb was moderate and Cd showed extremely high enrichment.
| Metal |
Metal concentration (mg/kg) |
Metal background concentration |
Enrichment factor |
Implication |
| Cd |
2.16 |
0.15 |
137.28 |
Extremely high enrichment |
| Pb |
2.84 |
10 |
2.703 |
Moderate enrichment |
| Fe |
6608.63 |
63,000 |
1 |
Minimal enrichment |
| V |
1.9 |
190 |
0.111 |
Minimal enrichment |
| Al |
2.54 |
82,000 |
0.0003 |
Minimal enrichment |
EF<2 (Depletion to minimal enrichment), EF 2 ≤ 5 (Moderate enrichment), 5≤ EF<20 (Significant enrichment), 20 ≤ EF<40 (Very high enrichment), EF>40 (Extremely high enrichment) [16,17].
Table 6 Heavy metal enrichment factor (EF) of selected heavy metals in the study area.
Discussion
Heavy metals in surface water
Lead (Pb) is a typical nonessential metal. In this study, Pb values ranged from 0.03-2.06 mg/L with a mean concentration of 0.64 ± 0.11 mg/L which was higher than the standard permissible limit of 0.05 mg/L [19,20]. Similar results were reported for the choba section of the New-calabar River, in Eastern Niger-Delta, Lagos Lagoon and for Tombia and Gbarantoru axis of Nun River in Bayelsa State [26-28]. The high concentration of Pb may be attributed to the oil and gas pipelines, the degeneration of pipes from old jetties and embankment, household waste from Pb containing paints used for boats and houses and Pb used as weights for fishing nets [29]. This result is in consonance with reports from who identified sources of Pb contamination in the Nigerian environment to include batteries, metal plating, painting and mining industries and who reported that about 200 tonnes of Pb were lost annually from fishing gears at mouths of rivers in Sweden [30]. Cadmium (Cd) had a mean concentration of 0.02 ± 0.01 mg/Land was above the standard limits [19]. This high value is worrisome considering the fact that it is relatively water soluble, highly toxic at very low exposure levels, generally more bioavailable and tend to bio accumulate, worse still, it is not degradable in nature and will thus, once released to the environment, stay in circulation [31]. Furthermore, these values may be as a result of pipeline electroplating, waste batteries and plastics (one source of Cadmium) disposed within the Soku oil field. High levels of Cd have also been recorded in surface waters of Aguobiri community, in South Ijaw Local Government, Bayelsa State Nigeriaby and Nun River by [28,32]. Fe showed a mean concentration of 1.94 ± 0.49 mg/L far above the permissible limit of 0.3 mg/L and 0.05mg/L by [19,20]. Iron (Fe) is required as one of the essential metals in nature is and elaborate mechanisms are used for its adequate acquisition in biological processes but it is also a toxicant. Fe has been implicated to precipitate on gills of fish as ferric oxide thus impairing oxygen uptake nevertheless and reported similar results while reported lower levels of Fe in Nun River [28,32,33].
Aluminium(Al) and Vanadium(V) concentrations ranged from 0.00-6.54 mg/L and 0.001-0.410 mg/L respectively. Most studies on heavy metals in the Niger Delta rarely investigate the levels of Aluminum in surface waters, but some have reported concentrations of 0.001 to 0.07 mg/L for Vanadium [32]. The concentrations of Al in the dry season were below detectable limits at all sites probably due to a reduction in illegal oil activities. The mean value of 0.88 ± 0.34 mg/L recorded for Al showed that it was within the standard limit of 1.00 mg/L and the 0.08 ± 0.03 mg/L of V in surface water is indicative of V pollution as it was higher than the standard limit of 0.009mg/L [19,22]. Al’s importance in toxicology has mostly been associated with environmental acidification. At high pH values (pH>7) that is in alkaline medium, it forms mostly insoluble hydroxides, at intermediate pH (5-7), sparingly soluble hydroxides are formed and at low pH values (pH<5), it forms the Al3+ ion [34].This ion is an osmoregulatory toxicant whereas the hydroxides may precipitate on gills of fish, hindering oxygen uptake. Thus, Al is considered to be an osmoregulatory toxicant at low pH values but a respiratory toxicant at intermediary pH values. The pH of the study area was between 6.65-6.83 indicating the possible formation of soluble hydroxides of Al [34]. The high V could be attributed to the spills of petroleum products from illegal refining of crude oil, out board engines and from bitumen used as adhesive and sealcoat in local wooden boat construction and also as a result of spill from illegal bunkers intercepted by military agencies during the wet season.
Heavy metals in sediment
Sediments have been reported to form the major repository of heavy metals in aquatic system [35]. Studies further showed that in the distribution of heavy metals in aquatic system, the levels in the bottom sediment are usually higher than in the water column which confirms that sediments acts as sink for heavy metals [36]. This concentration pattern was in agreement with report from Abonnema River by [37]. Mean concentration of Cd(2.16 mg/kg) in sediment from Soku oil field/aquatic environment was above the Federal Ministry of Environment standard limit of 0.03-0.3 mg/kg. However, this was lower than the value of 12.80 mg/kg recorded from the sediment of the lower reaches of the New Calabar river of Niger Delta [38]. The present study showed that the concentration of Pb in Soku oil field environment ranged from 0.06-8.76 mg/kg (mean of 2.84 mg/kg) which is low when compared with some previous investigations in the Niger Delta. Obunwo, reported sediment Cd of 3.96-37.14 mg/kg and recorded Cd concentration of 6.22 mg/kg from the sediment of Owudu creek, Niger Delta [39,40]. Fe concentration in the sediment was higher than those recorded in the surface water by three times order of magnitude. This difference is consistent with the report of who recorded two times order of magnitude in sediment to surface water [41]. The high value recorded is an indication of high rate of deposition of iron in the sediment. The observed concentration of Fe in the sediment was quite higher than recommended limit [20]. Higher concentrations of the heavy metals (Cd, Pb, V, Fe and Al) were obtained in the sediment than surface water and this was expected suggesting overtime pollution of the rivers and the property of sediment as repository to heavy metals pollution. The mean concentration of Al in sediment of Soku oil field environment was 2.54 mg/ kg with a range of 0.003-10.79 mg/kg which was above 1 mg/ kg recommended limit [19,24,42]. V was observed to be high with a mean value of 1.90 mg/kg. The higher values observed in the wet season indicative of spills from out board engines and illegal refining of crude oil, also from the use of bitumen in wooden boats construction. This result was consistent with the report from Oturuba creek of the same Niger Delta area by who reported V range of 1.21-5.24 mg/kg [43].
The Enrichment Factor (EF) of the sediments calculated using Fe as a reference element recorded the following mean concentrations with respective background level or abundance in earth’s crust in parenthesis for the various heavy metals investigated in this study [18]. The EF values (mg/kg) were 0.0003, 0.111, 2.703, 137.28 and 1.00 for Al, V, Pb, Cd and Fe respectively, which was indicative of minimal enrichment of Al, V and Fe in the sediment. While 2.703 mg/kg Pb classified the sediment as moderately contaminated with Pb while 137.28 mg/kg (Cd) was indicative of extremely high Cd contamination of the sediment of Soku oil Field.
Conclusion
The Soku oil field was a host of influences of illegal refining of crude oil, outboard engine spills of petroleum oil and exhaust release, industrial and human activities. Heavy metals were generally high in the dry season depicting heavy metal pollution and suggestive of high dilutions by heavy rain fall in the wet season. Higher concentrations of heavy metals in the sediment as against the surface water were indicative of overtime pollution of the river and confirmation of the sediment as a sink for heavy metals. Enrichment factors calculated showed a high Cd and Pb pollution of the environment becoming a potential threat to aquatic and human lives. The Enrichment factor of the other heavy metals (Fe, Al and V) studied were less than and equals One (≤ 1) indicative of minimal contamination. The determination of Al levels in water and sediment was a first in the area and with a pH between 6-7, presence of soluble hydroxides of Al as a respiratory toxicant for fish.
Competing Interests
Authors declare no competing interest exists
References
- International Union for Conservation-Niger (IUCN) (2013) Sustainable remediation and rehabilitation of biodiversity and habitats of oil spill sites in the Niger Delta: A report by the independent IUCN-Niger Delta Panel (IUCN-NDP) to the Shell Petroleum Development Company of Nigeria (SPDC). IUCN, Gland, Switzerland.
- United Nations Development Programme (UNDP) (2006) Niger delta human development report. Nigeria.
- Gibbs PJ, Miskiewics AZ (1995) Heavy metals in fish near major primary treatment sewage plant out fall. Mar Pollut Bull 30: 667-674.
- Elder JF (1988) Metal biogeochemistry in surface water systems: A review of principles and concepts. United States Geochemical Survey Circular 1013.
- Schuwerack PMM, Lewis JW, Jones PW (2009) The dynamics of protein and metal Metabolism in acclimated and Cd-exposed fresh water crab (Potamonautes warren). Ecotoxicol Environ Saf 72: 1220-1227.
- Turekian KK (1971) Impingement of man on the oceans. In: Hood DW (edtr), Wiley Interscience 1971: 9-73.
- Young EG, Conway CF (1942) On the estimation of allantoin by the rimini-schryver reaction. J Biol Chem 142: 839.
- Agbozu IE, Ekweozor IKE (2001) Heavy metals in a non-tidal fresh water swamp in the Niger Delta area of Nigeria. Afr J Sci 2: 175-182.
- Weis JS, Weis P (2002) Contamination of saltmarsh sediments and biota by CCA treated wood walkways. Mar Pollut Bull 44: 504-510.
- Akpoveta OV, Osakwe SA (2014) Determination of heavy metal content in refined petroleum product. J Appl Chem 7: 10-20.
- Cappo M, Alongi M, Williams D, Duke N (1995) A review and synthesis of Australian fisheries habitat research: Major threats, issues and gaps in knowledge of coastal and marine fisheries habitats-A prospectus of opportunities for the FRDC 'ecosystem protection program'. Australian Institute of Marine Science. Pp: 53.
- Harbison P (1986) Mangrove muds-a sink and a source for trace metals. Mar Pollut Bull 17: 246-250.
- Lee SV, Cundy AB (2001) Heavy metal contamination and mixing processes in sediments from the Humber Estuary, Eastern England. Estuar Coast Shelf Sci 53: 619-636.
- Chenall BE, Yassini I, Jones BG (1992) Heavy metal concentrations in lagoonal saltmarsh species, Illawarra region, South eastern Australia. Sci Total Environ 125: 203-225.
- American Public Health Association (1998) Standard methods for the examination of water and waste water. 14th edition, Washington DC. Pp: 1007-1157.
- Buat-Menard P, Chesselet R (1979) Variables influence of atmospheric flux on the trace metal chemistry of oceanic suspended matter. Earth Planet Sci Lett 42: 398-411.
- Likuku AS, Mmolawa KB, Gaboutloeloe GK (2013) Assessment of Heavy metal enrichment and degree of contamination around the copper-nickel mine in the Selebi Phikwe region, Eastern Botswana. Environ Ecol Res 1: 32-40.
- https://en.wikipedia.org/wiki/Enrichment_factor
- United States Environmental Protection Agency (USEPA) (2018) Risk assessment, Regional Screening Levels (RSLs) for chemical contaminants at superfund sites-User’s guide and summary generic tables.
- FMEnv (2011) National Guidelines and standards for water quality in Nigeria. Publication Federal Ministry of Environment (FMENV), Lagos, Nigeria. Pp: 114.
- https://www.epa.gov/sites/production/files/2016-09/documents/lead-compounds.pdf
- Integrated Risk Information System (IRIS) (2012) Chemical assessment summary national center for environmental assessment: United State Environmental Protection Agency (USEPA).
- Food and Agriculture Organization/World Health Organization (FAO/WHO) (2013) Guidelines for the safe use of waste water and food stuff: Waste water use in Agriculture. World Health Organization, Geneva. 2: 988.
- Food and Agriculture Organization/World Health Organization (FAO/WHO) (2007) Joint FAO/WHO food standard program Codex Alimentarius Commission, 13th edition.
- Department of Petroleum Resources (DPR) (2002) Environmental guidelines and standards for the petroleum industry in Nigeria (EGASPIN).
- Nwankwoala HO, Angaya YB (2017) An evaluation of heavy metals concentration in the Choba section of the New-Calabar River, Eastern Niger Delta. Biodivers Int J 1: 62-68.
- Aderinola OJ, Clarke EO, Olarinmoye OM, Kusemiju V, Anatekhai MA (2009) Heavy metals in surface water, sediments, fish and periwinkles of Lagos Lagoon. Am Eurasian J Agric Environ Sci 5: 609-661.
- Ogaga AA, Faith AM, Sylvester CI (2018) Impacts of anthropogenic activities on heavy metal levels in surface water of Nun River around Gbarantoru and Tombia towns, Bayelsa State, Nigeria. Ann Ecol Environ Sci 2: 1-8.
- Aroke UO, Osha OA, Aliyu UM, Shuaibu U (2016) Removal of Pb2+ onto HDTMA-Br modified kaolinite clay as function of pH: Batch sorption, isotherms and kinectics. J Environ Sci Toxicol Food Technol 10: 2319-2399.
- Gunner J, Bystrom M, Johansson L (2001) Lead emissions from lost fishing sinkers. Boreal Environ Res 6: 232-236.
- NCM Integrating fisheries and environment policies, The Nordic Experiences, The CFP (2003) Report prepared for the Nordic Council of Ministers. The Fisheries-Environment Strategy programme. Pp: 196.
- Imasuen OI, Egai AO (2013) Concentration and environmental implication of heavy metals in surface water in Aguobiri Community, Southern Ijaw Local Government Area, Bayelsa State, Nigeria. J Appl Sci Environ Manage 17: 467-472.
- Olowu RA, Ayejujo OO, Adewuyi GO, AdejoroIA, Denloye AAB, et al. (2010) Determination of heavy metals in fish tisues, water and sediment from Epe and Badagry Lagoons, Lagos , Nigeria. E-J Chem: 7: 215-221.
- Nikinmaa M (2014) An introduction to aquatic toxicology. 1st edition, Academic Press. Pp: 25.
- Adeniyi AA, Yusuf KA (2007) Determination of heavy metals in fish tissues, water and bottom sediments from Epe and Badagry Lagoons, Lagos, Nigeria. Environ Monit Assess 37: 451-458.
- Atta MB, Els-Sebale LA, Naoman MA, Kassab H (1997) The effect of cooking on the content of heavy metals in fish (Tilapia nilotica). Food Chem 58: 1-4.
- Ideriah TJK, David-Omiema S, Ogbonna DN (2012) Distribution of heavy metals in water and sediment along Abonnema Shoreline, Nigeria. Resour Environ 2:33-40.
- Horsfall M, Spiff AL (2002) Distribution and partitioning of trace metals in sediments of the lower reaches of the New Calabar River, Port Harcourt, Nigeria. Environ Monit Assess 78: 309-326.
- Obunwo CC, Braide SA, Izonfuo WAI,Chindah AC (2004) Influence of urban activities on the water quality of a fresh water stream in the Niger Delta, Nigeria. J Nigerian Environ Soc 2: 196-209.
- Friday UO, Wokoma AF, Edoghotu AJ (2013) Levels of bioaccumulation of some heavy metals in fish (Tilapia zilli) and their concentration in water and sediment of Owudu creek, Niger Delta, Nigeria. Resour Environ 3: 59-64.
- Ugbomeh AP (2013) Heavy metal concentration in two decapod estuarine crabs, Callinectes pallidus and Uca tangeri from the Azuabie Creek, Rivers State, Nigeria. PhD. desertation, Rivers State University of Science and Technology, Port Harcourt, Nigeria.
- Joint Food and Agriculture Organization/World Health Organization, Expert Committee on Food Additives (ECFA) (2006) Evaluation of certain food additives and contaminants: Sixty-seventh report of the Joint FAO/WHO expert committee on food additives. Rome, Italy. WHO Technical Report Series.
- Ezekwe CI, Utong IC (2017) Hydrocarbon pollution and potential ecological risk of heavy metals in the sediments of the Oturuba creek, Niger Delta, Nigeria. J Environ Geogr 10: 1-10.