Keywords
Brain metastases; Surgical resection; Metastasectomy; Localized field radiation therapy
Abbreviations
BM: Brain metastasis; RPA: Recursive partitioning analysis; MRI: Magnetic resonance imaging; CT: Computed tomography; SRS: Stereotactic radiotherapy; KPS: Karnofsky performance status
Introduction
Brain metastases occur in 9-17% of cancer patients [1]; tumor cells metastasize through the blood either by crossing the bloodbrain barrier or by occupying a vascular space to reach the parenchyma [2]. Lung and breast primaries are the most common tumors that metastasize to the brain, which may be attributed to the high incidence of these diseases. However, melanoma is the third in frequency to metastasize to brain irrespective to its low incidence [1]. This selectivity may be attributed to the special microenvironment of the brain [3].
Patients with single brain metastasis have median survival of about 3 months without treatment, noteworthy this median survival likely increased with the use of enhanced screening which allows detection of small sized metastasis [4]. Limited metastatic brain lesions can be controlled by local excision and radiation therapy with acceptable local control rates, unfortunately, the patients with brain metastasis experienced a poor survival outcome due to associated visceral metastasis [5]. Historically, the blood brain barrier was accused to be the direct cause of the resistance to various types of systemic therapy [6,7]. The data supported the idea of interrupted blood brain barrier that related to metastatic brain tumors. This phenomenon referred to the coordination between the microenvironment of the brain and tumors’ cells [3,8].
Benefits in survival and neurological symptom can be achieved by meta-stasectomy of solitary accessible brain lesions that originate from solid tumors, in selected patients with absent or limited systemic metastasis [9,10]. However these benefits is lost with high tumor burden and multiple brain lesions [11,12].
Concerning surgical procedures, local recurrence rates significantly increased with piece meal excision due to violation of tumor capsule to express 1.7 fold recurrence rate compared to en bloc resection [13]. In certain situations, such as posterior fossa tumors en bloc resection is mandatory, to avoid increased rates of local recurrence due to easily seedling of cerebrospinal fluid in such place [14]. Yoo et al. evaluated the local recurrence rates at 12 months in tumors removed with microscopic total resection versus piece meal total resection, which were 29% and 59% respectively without adjuvant local therapy [15].
The optimal treatment for multiple brain metastases is whole brain radiotherapy as it increases the overall survival 2-6 months this in addition to symptomatic improvement of local symptoms in up to 83% of patients with disseminated brain metastases [16]. The addition of surgery to WBRT when feasible dramatically increases local control rates compared to surgery alone [17].
However, the major problem in patients treated with WBRT is the late neurocognitive dysfunction [18,19], which began to appear more obviously due to advancement in the era of systemic therapy [20-22]. Preserving the neurocognitive function by shrinking the irradiated area and using special techniques such as hippocampal-sparing radiation therapy are used to achieve this goal [23,24].
Local therapy to the tumor bed after surgical excision, including SRS and involved field radiation therapy [IFRT] significantly decrease local recurrence rates and minimize late neurocognitive dysfunction by irradiating less normal tissue [25-31]. The aim of the study is to evaluate local recurrence and distant failure for patients who treated with localized radiation therapy, as well as to analyse the clinical and pathological variables associated with local recurrence and distant failure.
Materials and Methods
Patient eligibility criteria
Between January 2011 and April 2015, 31 patients with solitary brain metastasis proved by MRI or CT (if MRI was contraindicated) at Clinical Oncology and Neurosurgery Department, Tanta University Hospital and Kasr El Aini Hospital; Cairo University were enrolled. Patients were followed up until December 2015. At the time of analysis, the median follow-up duration was 18 months (Range: 4-40 months).
Patients fulfilled the following criteria: age older than 18 years, well-controlled primary and extra-cranial metastasis, Karnofsky performance status (KPS) of ≥ 60, underwent surgical excision in order to relieve neurological manifestations, obtain histopathological diagnosis or relieve mass effect followed by involved field (localized field) radiation therapy.
Patients were ineligible for the study if they presented with multiple brain metastases, incomplete resection of metastatic lesions, uncontrolled primary or extra-cranial metastatic disease, pregnant patients, those with dementia, altered mentality, or any psychological conditions that may make the patient unable to understand or render an informed consent. Also, patients who had second primary or concurrent serious comorbid medical diseases (e.g., uncontrolled cardiac disease, immune-compromised states, or uncontrolled severe infection) were not eligible.
This protocol was approved by The Ethics Committee in Faculty of Medicine at both Cairo and Tanta University, and all patients signed an informed consent before starting any treatment.
Study design and treatment Protocol
After the initial diagnosis of solitary brain metastasis, eligible patients were assigned for localized excision with involved field radiation therapy typically started 2-6 weeks post-surgery. Primary end-points were local recurrence and distant failure, which were calculated as time elapsed between date of surgery and MRI that demonstrated the recurrence.
Surgery
All patients underwent local excision. The extent of surgical excision was evaluated using post-operative computed tomography (CT) or magnetic resonance imaging (MRI) within 48 hours after surgery in order to confirm the extent of surgery.
Radiotherapy
Radiation therapy consisted of 3 D conformal fractionated radiation given at a daily dose of 2.67 Gy. Treatment continued over three weeks, 5 days a week to a total dose of 40.05 Gy. We used thermoplastic head mask for immobilization, CT simulation was done, and 1 mm axial CT cuts were obtained, then transferred to the Eclipse treatment planning system (Varian, Palo Alto, CA, USA) and registered with the pre-operative T1- weighted, pre and post-contrast and T2-weighted MRI to aid in target volume delineation. If there was any contraindication to MRI (e.g., claustrophobia or metallic implant) CT simulation images were fused with preoperative brain CT with and without contrast CTs. Clinical target volume (CTV) was identified as the resection cavity on CT cuts, and whereas, the planning target volume (PTV) was defined as CTV plus a 1 cm safety margin around it [30,31], with plan verification so that 95% of the dose was delivered to 100% of PTV.
Statistical analysis
SPSS 21.0 software version (SPSS, Inc.) was used for data analysis. Overall-survival (OS) rates were calculated from the time of brain metastasis diagnosis (date of meta-stasectomy) to the time of the last follow-up visit or death. Local recurrence (LR) was defined as the presence of newly developed enhancing lesion identified by MRI at the site of the resection cavity and distant failure (DF) was defined as enhancing lesion and/or lepto-meningeal metastasis outside resection cavity.
Kaplan-Meier method was used for estimating survival and log rank test was used to compare between the different prognostic factors. Mean and standard deviation were used to estimate the quantitative data. Stepwise multiple regression analyses were analysed by a Cox proportional hazards model, estimating the adjusted hazard ratio (HR) with 95% confidence intervals (CI) for age, sex, KPS, extent of tumor resection, maximal tumor diameter, tumor location, tumor depth and pathology of the primary tumor relative to the risk of death or disease progression. Statistical analyses used were two sided and the significance value was considered P ≤ 0.05.
Results
This study was conducted between January 2011 and April 2015 and recruited a total of 31 patients, including 18 females and 13 males, who had a median follow-up of 18 months (range, 4-40 months). Patient characteristics were summarized in Table 1. The mean age was 46.6 years (range, 26-73 years), and the mean KPS score was 82.9 ± SD 9.
| n (31) |
Characteristics |
% |
Age
Median (range)
Mean ± SD |
46 (26-73)
46.6 ± 13.68 |
|
Sex
Male female |
13
18 |
41.9
58.1 |
KPS
90
80
70
60
Median (range) |
14
12
3
2
90 (60-90) |
45.2
38.7
9.7
6.4 |
RPA
I
II III |
9
21
1 |
29
67.8
3.2 |
Extracranial metastasis
Present
Absent |
13
18 |
41.9
58.1 |
Primary tumor
NSCLC
Breast
Melanoma |
12
15
4 |
38.7
48.5
12.8 |
Tumor Size
Mean (range) |
3.1 (1.3-4.6) |
|
Tumor Site
Cerebellar
Frontal Parietal Temporal occipital |
12
6
7
4
2 |
38.7
19.4
22.6
12.9
6.4 |
Table 1: Patient and Tumor Characteristics.
Surgical treatment
All patients were operated by gross total resection, where 14 patient's (45.2%) brain metastases removed with piece meal resection, while the other 17 patients (54.8%) their metastases removed en bloc excision. The most common tumor pathology was breast carcinoma in 15 patients (48.4%) and NSCLC in 12 patients (38.7%). About seventy seven percent (24 patients) had deep tumors (the lesion surrounded by brain parenchyma), while about only twenty three percent (7 patients) have superficial tumors reaching or abutting the meninges. Among patients, 16 of them (51.6%) had supra-tentorial lesions and the other 15 patients (48.4%) represented by infra-tentorial lesions. The mean maximal tumor dimensions determined by MRI were 3.1 cm ranging from 1.3 to 4.6 cm, with 58.1% having tumors ≥ 3 cm.
Radiation therapy
The median time to start radiation therapy was 4 weeks (range, 3-9 weeks). The surgical cavity was contoured after CT simulation using MRI fusion images as CTV, with the median volume of the excision cavity 19 cm3 (range, 5-75 cm3).
Chemotherapy
Most cases received systemic chemotherapy (90.3%); none of them received concurrent chemotherapy, while 18 cases (58.1%) received their chemotherapy prior to meta-stasectomy and the course of radiation therapy and 10 cases (32.3%) after completion of radiation therapy.
Local recurrence
Local control was achieved in 83.9%, local control rates at 1 and 2 years were reaching 89.4% and 83.8% respectively, 95%CI (10.8- 47.2) with median LR-FS 29 months (Figure 1). Univariate analysis using log rank test demonstrated that no statistically significant difference associated with tumors greater than 3 cm (p=0.88), superficial location (p=0.30), piecemeal excision (p=0.93), PS (p=0.29), RPA (P=0.70), tentorial relation (p=0.82) or pathological type (p=0.33) (Table 2).
| Prognostic factors |
|
Total
number |
Number of local
recurrence |
LRFS)%(
1 year 2 year |
P-value |
| Tumor size |
<3
≥ 3 |
13
18 |
2
3 |
92.3
87.4 |
79.1
87.4 |
0.88 |
| Tumor depth |
Superficial
Deep |
7
24 |
3
9 |
85.7
86.7 |
28.66
6.1 |
0.57 |
| Tumor excision |
Enbloc
Piece meal |
14
17 |
2
3 |
92.9
87.8 |
92.9
79.1 |
0.93 |
| Tentorial relation |
Supratento
-rial
Infratentor
-ial |
16
15 |
3
2 |
93.8
83.6 |
84.4
83.6 |
0.82 |
| Primary tumor |
NSCLC
Breast
Melanoma |
12
15
4 |
2
2
1 |
91.7
93.3
75 |
NR
93.3
75 |
0.33 |
| RPA class |
I
II
III |
9
21
1 |
1
4
0 |
100
84.3
100 |
83.3
84.3
100 |
0.70 |
| PS |
90
80
70
60 |
14
12
3
2 |
2
1
1
1 |
92.9
88.9
100
50 |
92.9
88.9
50
Not
reach ed |
0.29 |
Table 2: Association between prognostic factors and local recurrence, assessed by univariate analysis (log rank test).
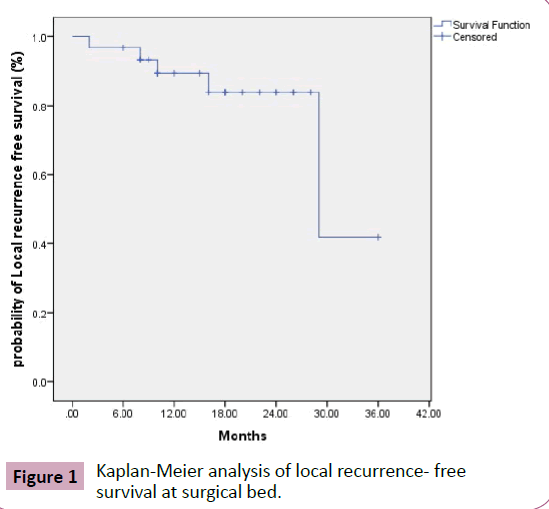
Figure 1 Kaplan-Meier analysis of local recurrence- free survival at surgical bed.
Distant failure
At final analysis relapse in the brain in sites other than tumor bed occurred in 38.7% of cases (12 patients); the actuarial rates of distant failure free survival were 72% and 54.9% at 12, 24 months respectively (Figure 2). The median DF-FS was 25 months, 95% CI (14.9-35.1). Univariate analysis demonstrated that clinical variables associated with direct impact on distant failure were supra-tentorial, tentorial tumors (p=0.02) and melanoma (p=0.002). However, only the primary tumor was still significant in multivariate analysis. Other variables such as, tumors greater than 3 cm (p=0.67), superficial location (p=0.26), piecemeal excision (p=0.55), PS (p=0.34) and RPA (P=0.57) were not associated with increased incidence of distant failure (Table 3).
| Prognostic factors |
|
Total
number |
Number of
distant failure |
DFFS )%(
1 year 2 year |
P-value |
| Tumor size |
<3
≥ 3 |
13
18 |
4
8 |
83.1
65.5 |
55.4
57.3 |
0.67 |
| Tumor depth |
Superficial
Deep |
7
24 |
4
8 |
51.4
77.6 |
34.3
60.5 |
0.26 |
| Tumor excision |
Enbloc
Piece meal |
14
17 |
6
6 |
76.2
69.7 |
47.6
61 |
0.55 |
| Tentorial relation |
Supratentorial
Infratentorial |
16
15 |
3
9 |
86.5
56.2 |
75.7
31.2 |
0.02 |
| Primary tumor |
NSCLC
Breast
Melanoma |
12
15
4 |
5
3
4 |
58.3
93.3
25 |
33.5
74.9
0 |
0.002 |
| RPA class |
I
II
III |
9
21
1 |
3
9
0 |
88.9
80.4
100 |
66.7
49.1
100 |
0.57 |
| PS |
90
80
70
60 |
14
12
3
2 |
6
3
1
2 |
77.9
68.8
66.7
50 |
55.7
68.8
66.7
NR |
0.34 |
NR: not reached
Table 3: Association between prognostic factors and distant failure, assessed by univariate analysis (log rank test).
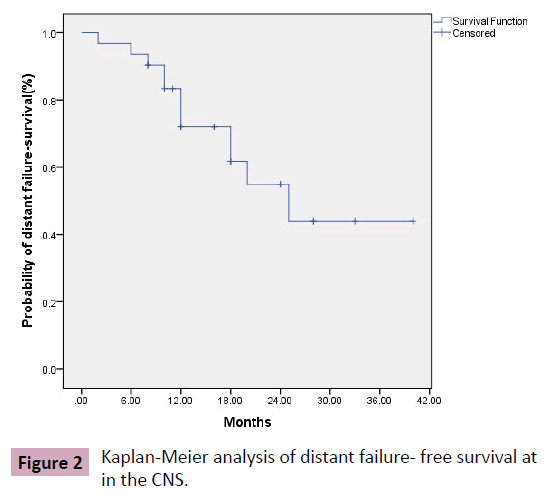
Figure 2 Kaplan-Meier analysis of distant failure- free survival at in the CNS.
Salvage therapy
Local recurrence was salvaged in four of five patients: 2 patients by surgical excision followed by SRS; 1 with surgery followed by WBRT; and 1 by SRS alone. Distant failure was salvaged in 10 of 12 patients: 5 patients with SRS and 5 by whole brain radiotherapy.
Overall survival
The median overall survival was 26 months {95%CI (17.7- 34.3)}. Overall survival at 12 and 24 months was 86.4% and 56.4% respectively (Figure 3). RPA class associated with survival (P=0.02). Among all cases, five cases died from local progression of primary tumor, four died due to neurological symptoms progression, one case died due to neurological disease not related to malignancy. One case died due to febrile neutropenia secondary to chemotherapy and one case due to unknown cause.
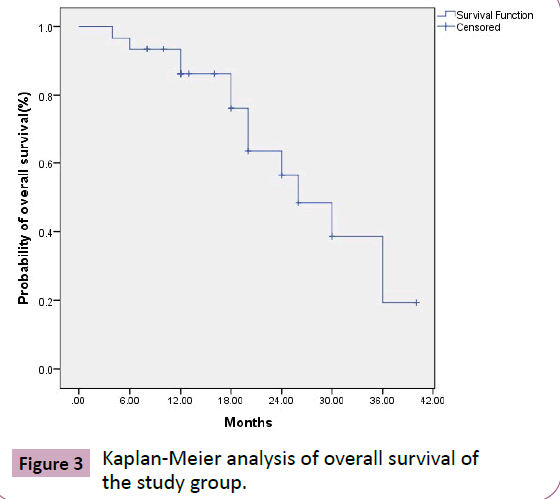
Figure 3 Kaplan-Meier analysis of overall survival of the study group.
Discussion
Single brain metastasis is reported in nearly 40%–47% of all patients who have brain metastases [32]. Historically, WBRT as a single modality of treatment remained the optimal treatment for these patients. Two prospective randomized trials IN 1990s, confirmed meta-stasectomy followed by WBRT resulted in better neurological outcomes combined with improved overall survival compared with WBRT alone [33,34].
In a subsequent randomized trial by Patchell et al. The omission of WBRT after surgical resection did not influence overall survival [17]. However, Postoperative WBRT results in improved local control of disease, both at the excision cavity and distant brain sites [35]. WBRT also reduced deaths from neurological causes [33].
Few series addressed the management of solitary brain metastases after surgical excision by IFRT. Chin et al. enrolled 56 patients; the median follow up was 19.1 months. These series was an extension from their previous study including 33 patients [30,31].
In our series, one year local control rates were 89.4%, this is in agreement with the results reported by the Patchell et al. 90% [30], and Shin et al. 91.4% [31], but higher than that reported by some other trials of local treatments with local control rates at one year ranked from 74 to 94% [25-29,37-42] (Table 4). In our series, the one year DF rates were 30.6%, which is in agreement with other series in which DF ranged from 28 to 69% [25-29,36-41] (Table 2). These favourable results may attribute to the progress in systemic treatment in addition to proper selection of patients with good performance and well–controlled systemic disease.
| Author |
N |
GTR
% |
Local
therapy |
Medi
an dose |
Local
contr ol 1 year
% |
Local
contr ol
2year
% |
Distant
failure
1 year
% |
Media
n surviva l
months |
Media
n FU
months |
Radiatio
n necrosis |
| Chin et al. [31] |
56 |
100 |
IFRT |
40.0
5 |
91 |
85 |
33 |
19 |
19 |
4 |
Connolly et al.
[30] |
33 |
100 |
IFRT |
40.0
5 |
90.3 |
85.8 |
39.3 |
16 |
16 |
0 |
Brennan et al.
[25] |
39 |
92 |
SRS |
18 |
85 |
a |
44 |
15 |
12 |
18 |
| Choi et al. [38] |
112 |
90 |
SRS |
20 |
91 |
88 |
54 |
17 |
a |
7 |
Robbins et al.
[40] |
85 |
68 |
SRS |
16 |
81 |
76 |
58 |
12 |
11 |
8 |
| Kelly et al. [39] |
18 |
94 |
SRS |
18 |
89 |
a |
35 |
Not
reache
d |
13 |
0 |
Karlovits et al.
[26] |
52 |
92 |
SRS |
15 |
92 |
a |
44 |
15 |
13 |
0 |
| Soltys et al. [37] |
72 |
85 |
SRS |
19 |
79 |
a |
47 |
15 |
8 |
10 |
| Quigely et al. [28] |
32 |
94 |
SRS |
14 |
94 |
a |
28 |
16 |
14 |
0 |
Mathieu et al.
[29] |
40 |
80 |
SRS |
16 |
74 |
a |
54 |
13 |
13 |
5 |
| Ling et al. [42] |
100 |
81 |
SRT |
22 |
72 |
a |
64 |
13 |
12 |
9 |
| Ahmed et al. [44] |
65 |
97 |
SRT |
20-
30 |
87 |
70 |
51 |
10 |
9 |
2 |
| Minniti et al. [45] |
101 |
72 |
SRT |
27 |
93 |
84 |
50 |
17 |
16 |
9 |
| Petr et al. [27] |
72 |
100 |
I-125
seeds |
b |
93 |
a |
32 |
14 |
16 |
0 |
| Patchel et al. [33] |
23 |
100 |
WBRT |
36 |
80 |
a |
a |
10 |
18 |
a |
| Patchel et al. [35] |
49 |
100 |
WBRT |
50 |
90
Day |
a |
14 Day |
12 |
12 |
a |
| Patchel et al. [35] |
46 |
100 |
None
(oberva- tion group) |
|
54
Day |
a |
37 Day |
11 |
11 |
a |
Table 4: Outcomes of different local treatment modalities after surgical resection of brain metastasis.
Localized field radiation therapy has disadvantages in comparison to other local control measures such as brachytherapy and SRS due to longer overall treatment time. In our series, radiation therapy was delivered over 3 weeks, brachytherapy and SRS may be delivered in one day, however, the control rate is comparable. Other Local measures also have some disadvantages, Brachytherapy increases the risk of infection, not available in many centres and need operator experience [42]. If collapse of surgical cavity occurred after surgery, the SRS can't be delineated properly. Additionally, the tumor bed of larger tumors (≥ 3 cm) may be too large for safe irradiation with single-fraction SRS [25,29,36,37,40].
Advantage of localized field radiation therapy includes the ability of proper defining PTV, easy reproducibility of treatment planning, and the radiation delivery is safe for tumors with larger resection cavities unsuitable for single-fraction SRS. Alternatively, multi- fraction SRS may be another option for tumors with large cavities (≥ 3 cm) after resection; however, this may be at the expense of either more adverse effects of less tumor local control rates. Minniti et al. concluded that, patients treated with multiple fractions SRS regimen of 27Gy over three fractions (median surgical cavity volume of 17.5 cm3), experienced a 9% risk of radiation necrosis or adverse radiation effect [42]. The median volume of surgical cavity in our study using localized field radiation therapy was 19 cm3 and the local control rate was 83.8% at 24 months, with low events of radiation necrosis or adverse effects (3.2%). Patients with larger tumor cavities after surgery will, therefore, be selected for treatment with localized field radiation therapy instead of multiple fractions SRT to avoid increased risk of radiation toxicity [30,31].
Concerning univariate analysis, risk factors including larger tumor size, meningeal infiltration, and surgical technique (piecemeal versus en bloc) did not adversely affect the local control rate, in agreement with that reported by Connolly et al. and Shin et al. [30,31]. However, in another phase II study, Brennan et al. reported that tumor size ≥ 3 cm, superficial tumors and meningeal infiltration were associated with increased LR rates in patients treated with SRS following surgery [25]. This difference may be due to larger margins around (10 mm) PTV used in our series in comparison to the margins (2-3 mm) used in the phase II study.
Comparable to the results reported by other trials, primary tumors (melanoma histology) and tentorial relation (infratentorial tumors) had significant correlation with distant failure [25,30,31]. The possible explanation of increased rates of DF in patients with infratentorial lesions is the nearby location to cerebellar cisterns which facilitate seedling of cerebrospinal fluid. The direct association between melanoma histology proved in many series but the cause still unclear [25,31].
Salvage therapy has an important role in order to address local recurrence and distant failure; in our series, we utilized various salvage approaches according to individualized clinical data for every patient such as SRS and whole brain irradiation with or without exclusion of involved field [43,44].
Treatment was well tolerated with acceptable early and late toxicity our study aiming to decrease volume of irradiated normal brain tissue and consecutively neurocognitive toxicity. depending on the data from previous studied which postulated that decline in the level of neurocognitive function directly related to the volume of brain tissue irradiated following SRS alone in comparison to combined SRS and WBRT using Hopkins Verbal Learning Test-Revised [20,22].
As there is theoretical and retrospective evidence supporting that reducing irradiation to normal brain tissue is a worthwhile clinical goal in order to avoid neurocognitive dysfunction, direct comparison of IFRT with WBRT will be difficult to be done in a randomized trial for patients who had surgically resected solitary brain metastases. However, it is reasonable to evaluate the toxicity and control rate of SRS versus IFRT in those patients. In conclusion, our study demonstrated the safety and feasibility of IFRT following surgical excision of single BM for patients with good performance status if primary disease is well controlled. Also, localized field radiation therapy can be considered as an alternative treatment approach to SRS for patients with excised metastatic tumors and surgical resection cavities ≥ 3 cm, and an alternative option to WBRT for those patients with considerable close follow-up.
Conflict of Interests
All authors report no conflict of interests.
Authors’ Contribution
All authors designed the study, analysed the data, performed the statistical analysis, drafted the paper, contributed to patient treatment and follow-up and critically reviewed the paper.
Funding
We did not receive any funding for this research.
References
- Nayak L, Lee EQ, Wen PY (2012) Epidemiology of brain metastases. Curr Oncol Rep 14:48-54.
- Pantel K, Brakenhoff RH (2004) Dissecting the metastatic cascade. Nat Rev Cancer 4:448-456.
- Ramakrishna R, Rostomily R (2013) Seed, soil, and beyond: The basic biology of brain metastasis. SurgNeurolInt 4:256-264.
- Nussbaum ES, Djalilian HR, Cho KH, Hall WA (1996) Brain metastases. Histology, multiplicity, surgery, and survival. Cancer 78:1781-1788.
- Johnson JD, Young B (1996) Demographics of brain metastasis. NeurosurgClin N Am 7:337-44.
- Alonso A (2015) Ultrasound-Induced blood-brain barrier opening for drug delivery. Front Neurol Neurosci 36:106-115.
- Barani IJ, Larson DA, Berger MS (2013) Future directions in treatment of brain metastases. Surg Neurol Int 4: 220-230.
- Meng F, Wu G (2012) The rejuvenated scenario of epithelial-mesenchymal transition (EMT) and cancer metastasis. Cancer Metastasis Rev 31:455-467.
- Yaeger KA, Nair MN (2013) Surgery for brain metastases. Surg Neurol Int 4:203-208.
- Patchell RA (2003) The management of brain metastases. Cancer Treat Rev 29:533-540.
- Galluzzi S, Payne PM (1956) Brain metastases from primary bronchial carcinoma: a statistical study of 741 necropsies. Br J Cancer10:408-414.
- Takakura K (1990) Metastatic brain tumors—recent trends. GanTo Kagaku Ryoho17:768-770.
- Patel TR, Knisely JP, Chiang VL (2012) Management of brain metastases: surgery, radiation, or both? Hematol Oncol Clin North Am 26:933-947.
- Suki D, Hatiboglu MA, Patel AJ, Weinberg JS, Groves MD (2009) Comparative risk of leptomeningeal dissemination of cancer after surgery or stereotactic radiosurgery for a single supratentorial solid tumor metastasis. Neurosurgery 64: 674-676.
- Yoo H, Kim YZ, Nam BH, Shin SH, Yang HS, Lee JS, et al. (2009) Reduced local recurrence of a single brain metastasis through microscopic total resection. J Neurosurg 110:730-736.
- Gaspar L, Scott C, Rotman M, Asbell S, Phillips T, et al.(1997) Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys 37:745-751.
- Patchell RA, Tibbs PA, Regine WF, Dempsey RJ, Mohiuiddin M, et al. (1998) Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA 280: 1485-1489.
- Cross NE, Glantz MJ (2003) Neurologic complications of radiation therapy. Neurol Clin. 21:249-277.
- Herrmann T, Knorr A, Dorner K (1987) The RTOG/EORTC classification criteria for early and late radiation reactions. Radiobiol Radiother (Berl) 28:519-528.
- De Angelis LM, Delattre JY, Posner JB (1989) Radiation-induced dementia in patients cured of brain metastases. Neurology 39:789-796.
- Packer RJ, Sutton LN, Atkins TE, Radcliffe J, Bunin GR, et al.(1989) A prospective study of cognitive function in children receiving whole-brain radiotherapy and chemotherapy: 2-year results. J Neurosurg 70:707-713.
- Chang EL, WefelJS, Hess KR, Allen PK, Lang FF, et al. (2009) Neuro recognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: A randomized controlled trial. Lancet Oncol 10:1037-1044.
- Brown PD, Pugh S, Laack NN, Wefel JS, Khuntia D, et al. (2013) Meman- tine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: A randomized, double-blind, placebo-controlled trial. Neuro Oncol 15:1429-1437.
- Gondi V, Tolakanahalli R, Mehta MP, Tewatia D, Rowley H, et al. (2013) Hippocampal-sparing whole-brain radiotherapy: a “how-to "technique using helical tomotherapy and near accelerator-based intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys 78:1244-1252.
- Brennan C,Yang TJ, Hilden P, Zhang Z, Chan K, et al. (2014) A phase 2 trial of stereotactic radiosurgery boost after surgical resection for brain metastases. Int J Radiat Oncol Biol Phys 88:130-136.
- Karlovits BJ, Quigley MR, Karlovits SM, Miller L, Johnson M, et al.(2009) Stereotactic radiosurgery boost to the resection bed for oligometastatic brain disease: Challenging the tradition of adjuvant whole-brain radiotherapy. Neuro- surg Focus 27:E7.
- Petr MJ, McPherson CM, Breneman JC, Warnick RE (2009) Management of newly diagnosed single brain metastasis with surgical resection and permanent i- 125 seeds without upfront whole brain radiotherapy. J Neurooncol 92:393-400.
- Quigley MR, Fuhrer R, Karlovits S, Karlovits B, Johnson M (2009) Single session stereotactic radiosurgery boost to the post-operative site in lieu of whole brain radiation in metastatic brain disease. J Neurooncol 87:327-332.
- Mathieu D, Kondziolka D, Flickinger JC, Fortin D, Kenny B, et al. (2008) Tumor bed radiosurgery after resection of cerebral metastases. Neurosurgery 62:817-823.
- Connolly EP, Mathew M, Tam M, King JV, Kunnakkat SD, et al.(2013)Involved field radiation therapy after surgical resection of solitary brain metastases-mature results. Neuro Oncol 15:589-594.
- Shin SM, Vatner RE, Tam M, Golfinos JG, Narayana A, et al.(2015) Resection followed by involved-field fractionated radiotherapy in the management of single brain metastasis. Front. Oncol. 5:206.
- Gavrilovic I, Posner J (2005) Brain metastases: Epidemiology and pathophysiology.J Neuro-Oncol. 75:5-14.
- Patchell RA, Tibbs PA, Walsh JW (1999)A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med 322:494-500.
- Vecht CJ,Haaxma-Reiche H, Noordijk EM (1993) Treatment of single brain metastasis: radiotherapy alone or combined with neurosurgery. Ann Neurol 33:583-590.
- Kocher M, Soffietti R (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: Results of the EORTC 22952-26001 study. J Clin Oncol 29:134-141.
- Soltys SG, Adler JR, Lipani JD, Jackson PS, Choi CY, et al.(2008) Stereotactic radiosurgery of the postoperative resection cavity for brain metastases. Int J Radiat Oncol Biol Phys70:187-193.
- Choi CY, Chang SD, Gibbs IC, Adler JR, Harsh GR, et al. (2012) Stereotactic radiosurgery of the post-operative resection cavity for brain metastases: prospective evaluation of target margin on tumor control. Int J Radiat Oncol Biol Phys 84:336-342.
- Kelly PJ, Lin YB, Yu AY, Alexander BM, Hacker F, et al.(2012) Stereotactic irradiation of the post-operative resection cavity for brain metastasis: a frameless linear accelerator based case series and review of the technique. Int J Radiat Oncol Biol Phys 82:95-101.
- Robbins JR, Ryu S, Kalkanis S, Cogan C, Rock J, et al.(2012) Radiosurgery to the surgical cavity as adjuvant therapy for resected brain metastasis. Neuro-surgery 71: 937-943.
- Do L, Pezner R, Radany E, Liu A, Staud C, et al. (2009) Resection followed by stereotactic radiosurgery to resection cavity for intracranial metastases. Int J Radiat Oncol Biol Phys 73:486-491.
- Ling DC,Vargo JA, Wegner RE, Flickinger JC, Burton SA, et al.(2015) Postoperative stereotactic radiosurgery to the resection cavity for large brain metastases: clinical outcomes, predictors of intracranial failure, and implications for optimal patient selection. Neurosurgery 76:150-156.
- Chan TA, Weingart JD, Parisi M, Hughes MA, Olivi A, et al.(2005) Treatment of recurrent glioblastomamultiforme with gliasite brachytherapy. Int J Radiat Oncol Biol Phys 62:1133-1139.
- Ahmed KA, Freilich JM, Abuodeh Y, Figura N, Pate lN, et al.(2014) Fractionated stereotactic radiotherapy to the post-operative cavity for radioresistant and radiosensitive brain metastases. J Neuro Oncol 118:179-186.
- Minniti G, Esposito V, Clarke E, Scaringi C, Lanzetta G, et al.(2013) Multi dose stereotactic radiosurgery (9 gy x 3) of the postoperative resection cavity for treatment of large brain metastases. Int J Radiat Oncol Biol Phys 86:623-629.