Naotaka Okamoto1, Keisuke Yasumura1, Koji Yasumoto1, Akihiro Tanaka1, Naoki Mori1, Daisuke Nakamura1, Masamichi Yano1, Yasuyuki Egami1, Ryu Shutta1, Yasushi Sakata2, Jun Tanouchi1, and Masami Nishino1
1 Division of Cardiology, Osaka Rosai Hospital, Osaka, Japan
2 Department of Cardiovascular Medicine, Osaka University Graduate School of Medicine, Suita, Japan
*Corresponding Author:
Masami Nishino
Division of Cardiology, Osaka Rosai Hospital, 1179-3
Nagasone-Cho, Kita-Ku, Sakai City, Osaka 5918025, Japan.
Tel: 81722523561
Fax: +81722505492
E-mail: mnishino@osakah.johas.go.jp
Received Date: May 07, 2017; Accepted Date: June 01, 2017; Published Date: June 07, 2017
Citation: Okamoto N, Yasumura K, Yasumoto K, et al. Neointimal Characteristics after Everolimus-Eluting Stents Implantation for Chronic Total Occlusion: An Optical Coherence Tomographic Study. Interv Cardiol J 2017, 3:2. DOI: 10.21767/2471-8157.100055
Keywords
Chronic total occlusion; Coronary artery disease; Optical coherence tomography; Drug-eluting stents
Introduction
Current studies have shown that successful percutaneous coronary intervention (PCI) for chronic total occlusion (CTO) improves the quality of life, left ventricular ejection fraction and survival rate [1-8]. The development of devices such as guide wires, balloons and stents, and the improvement of operator techniques has increased the success rates of PCI for CTO lesions and decreased the complication rates over time [9,10]. Accordingly, PCI for CTO has been more generally performed. In addition, drug-eluting stents (DESs) for treatment of CTO lesions has improved angiographic and clinical long-term outcomes [11- 13]. However, even now, restenosis and reocclusion rate of PCI for CTO lesions has been high [14]. Subintimal tracking and reentry (STAR) technique is one of methods of PCI for CTO lesions. It is reported that STAR technique is associated with higher rate of restenosis and reocclusion than conventional antegrade CTO recanalization [15,16]. Therefore, we think the way of healing after stent implantation for CTO lesions may be different by the stent locations; subintimal stenting or intra true-lumen stenting. In this study, we investigated the difference of neointimal characteristics of everolimus-eluting stent (EES) among stent locations including subintimal stenting and intra true-lumen stenting for CTO lesions and stenting for non-CTO lesions.
Subjects and Methods
Patient selection and study design
We enrolled consecutive patients who underwent implantation of EES (XIENCE V™, Abbott Vascular, Santa Clara, CA, USA and PROMUS™, Boston Scientific Corp., Natick, MA, USA) from February 2010 to June 2011 at our hospital. Stent placement was guided by intra-vascular ultrasound systems (IVUS), iLab™ Ultrasound Imaging System (Boston Scientific Corp., Natick, MA, USA) and Eagle Eye™ (Volcano Corporation, Rancho Cordova, CA, USA). We excluded patients with in-stent restenosis, acute myocardial infarction, coronary bypass graft lesion, left main coronary, ostium of the right coronary artery, extremely tortuous lesions, and hemodialysis. We first divided our patients into a CTO group and non-CTO group. In this study, CTO was defined as a lesion whose occlusion duration was more than 3 months. In addition, we investigated IVUS date obtained when we performed PCI in detail. In non-CTO lesions, crossing wire passed through true lumen in all cases. On the other hand, in CTO lesions, some crossing wires passed through subintima (Figure 1), and some part of stents were implanted in subintima in such cases. We defined these cases with subintimal stenting as CTO subintima group. We defined the patients with crossing wire passing through true-lumen completely in CTO lesion as CTO true-lumen group. Therefore, we finally divided our patients into CTO subintima group, CTO true-lumen group and non-CTO group. We used the two OCT systems (TD-OCT and FD-OCT). OCT examination was performed at 8 (6 to 10, mean 7.8) months after EES implantation. We performed TD-OCT with a 0.014inch wiretype imaging catheter (Image Wire, Light Lab Imaging, Westford, MA, USA). The OCT catheter was inserted through a 6 Fr catheter into target coronary arteries beyond the implanted EES. After injecting nitro-glycerine, OCT image was acquired during inflating occlusion catheter (Helios; Light lab Imaging, Westford, MA, USA) and injecting ringer lactate to remove the blood cell from the field of view. An auto pull-back system at 1 mm/sec was used [16]. We performed FD-OCT with a 2.7 Fr OCT catheter (Dragonfly imaging catheter, Light Lab Imaging, Westford, MA, USA). During acquirement of imaging, we injected non-diluted iodine contrast or ringer lactate at rate of 3 ml/s to 5 ml/s to achieve blood clearance. Auto pull-back at rate of 20 mm/s was used [17].
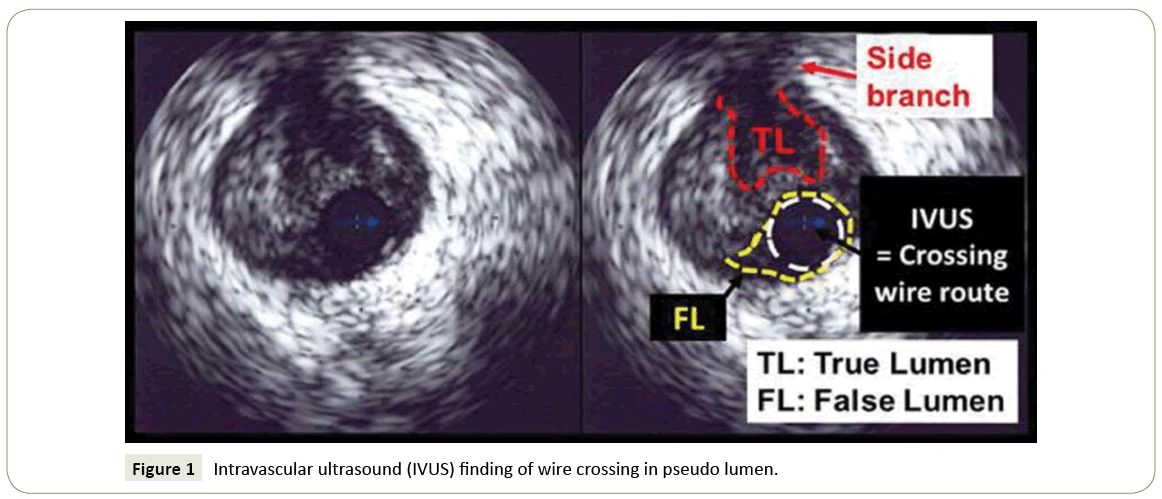
Figure 1: Intravascular ultrasound (IVUS) finding of wire crossing in pseudo lumen.
We analyzed cross-sectional OCT images at 1 mm intervals. We excluded the images that had some portion of the lesion out of screen, a side branch, or poor quality caused by residual blood cells, artifacts, or reverberation. OCT parameters including the percentage of neointimal hyperplasia area obstruction (%NHAO), mean neointimal hyperplasia (NIH) thickness, the proportion of uncovered struts, the proportion of malapposed struts at cross sectional level, NIH thickness, proportion of uncovered struts, and proportion of malapposed struts at strut level were measured. NIH cross-sectional area was calculated by the following formula: (stent cross-sectional area) – (lumen cross-sectional area). Percentage of neointimal hyperplasia area obstruction (%NHAO) was the percentage of NIH area and was calculated as NIH area / stent cross-sectional area × 100. NIH thickness was the distance between the luminal surface of the covering tissue and the luminal surface of the strut. Uncovered struts were defined as struts on which there was no definite neointima. Malapposed struts were defined as struts whose distance from the vessel wall was more than 99 μm [7,18-20]. We compared these OCT parameters among the three groups.
Medical ethics committee of Osaka Rosai Hospital approved this study and we obtained written informed consent from all study patients.
Statistical analysis
Categorical variables are presented as numbers and they were analyzed by either the chi-square test or Fisher's exact test. Continuous data in the normally distributed cases are presented as means ± standard deviation and the data between different groups are compared by ANOVA. Continuous date, that do not follow the normal distribution, are shown as median (25th percentile - 75th percentile), and the data between different groups are analyzed by Kruskal-Wallis test, if the distribution were skewed. Non-parametric multiple comparison was analyzed by Steel-Dwass test. A value of P<0.05 was considered to be statistically significant. Statistical analyses were performed using JMP version 11.0.0 (SAS Institute Inc., Cary, North Carolina, USA).
Results
Our patients comprised 28 patients with EES implantation. As a result, in our study, CTO subintima group consisted of 3 patients (5 EESs), CTO true-lumen group consisted of 2 patients (4 EESs), and non-CTO group consisted of 23 patients (35 EESs). We succeeded PCI by antegrade wiring approach in both 2 cases of CTO true-lumen group. In CTO subintima group, there were 2 cases succeeded by antegrade wiring approach and the other 1 case was succeeded by retrograde wiring approach. The number of observed cross sections and stent struts which we could evaluate were 59 cross sections and 584 struts in CTO subintima group, 102 cross sections and 997 struts in CTO true-lumen group, and 495 cross sections and 4747 struts in non-CTO group, respectively.
Patient characteristics are shown in Table 1. There were no significant differences of the patient characteristics among the three groups except age, and left ventricular ejection fraction. Lesion and stent characteristics are presented in Table 2. The vessel with target lesion was significantly different but the other lesion and stent characteristics were similar among the three groups. The OCT findings are shown in Figure 2 and 3. The results of comparison among the three groups at cross sectional analysis are shown in Figure 2. The %NHAO and mean NIH thickness were significantly lowest in CTO subintima group and highest in CTO true-lumen group among the three groups (%NHAO (%): CTO subintima vs. non-CTO: 7.4 (4.4-9.8) vs. 10.6 (7.3-15.8), p<0.0001; CTO true-lumen vs. non-CTO: 11.5 (7.9-19.2) vs. 10.6 (7.3-15.8), p=0.132; CTO subintima vs. CTO true-lumen: 7.4 (4.4-9.8) vs. 11.5 (7.9-19.2), p<0.0001, NIH thickness (μm): CTO subintima vs. non- CTO: 48 (36-71) vs. 74 (48-117) , p<0.0001; CTO true-lumen vs. non-CTO: 79 (55-159) vs. 74 (48-117) , p=0.099; CTO subintima vs. CTO true-lumen: 48 (36-71) vs. 79 (55-159), p<0.0001). On the contrary, the proportions of uncovered struts and malapposed struts were significantly highest in CTO subintima group and significantly lowest in CTO true-lumen group (uncovered struts (%): CTO subintima vs. non-CTO: 12.5 (0-28.6) vs. 0 (0-9.1), p<0.0001; CTO true-lumen vs. non-CTO: 0 (0-0) vs. 0 (0-9.1), p=0.041; CTO subintima vs. CTO true-lumen: 12.5 (0-28.6) vs. 0 (0-0), p<0.0001 and malapposed struts (%): CTO subintima vs. non-CTO: 0 (0-7.7) vs. 0 (0-0), p<0.0001; CTO true-lumen vs. non- CTO: 0 (0-0) vs. 0 (0-0), p=0.042; CTO subintima vs. CTO truelumen: 0 (0-7.7) vs. 0 (0-0), p<0.0001). The results of comparison among the three groups at strut-level analysis are shown in Figure 3. NIH thickness was significantly lowest in CTO subintima group and significantly highest in CTO true-lumen group (NIH (μm): CTO subintima vs. non-CTO: 40 (20-70) vs. 70 (40-110), p<0.0001; CTO true-lumen vs. non-CTO: 80 (40-150) vs. 70 (40- 110), p<0.0001; CTO subintima vs. CTO true-lumen, 40 (20-70) vs. 80 (40-150), p<0.0001). The proportion of uncovered struts and proportion of malapposed struts were significantly highest in CTO subintima group and significantly lowest in CTO truelumen group (uncovered struts (number (%)): CTO subintima vs. CTO true-lumen vs. non-CTO: 109 (18.7) vs. 47 (4.7) vs. 375 (7.9), p<0.0001 and malapposed struts (number (%)): CTO subintima vs. CTO true-lumen vs. non-CTO: 36 (6.2) vs. 0 (0) vs. 45 (0.95), p<0.0001).
| |
CTO subintima
Group(n=3) |
CTO true-lumen group
(n=2) |
non-CTO group
(n=23) |
P value |
| Age |
63.7 ± 5.1 |
74.0 ± 2.8 |
70.4 ± 4.8 |
0.049 |
| Male (n (%)) |
3 (100) |
1 (50) |
16 (70) |
0.569 |
| Diabetes mellitus (n (%)) |
0 (0) |
1 (50) |
10 (43) |
0.361 |
| Hyper tension (n (%)) |
2 (67) |
2 (100) |
21 (91) |
0.459 |
| Dyslipidemia (n (%)) |
2 (67) |
1 (50) |
17 (74) |
0.763 |
| Chronic kidney disease (n (%)) |
0 (0) |
1 (50) |
3 (13) |
0.308 |
| Estimated glomerular filtration rate (ml/min/1.73m2) |
83.9 ± 7.7 |
59.2 ± 4.2 |
71.6 ± 14.1 |
0.143 |
| Left ventricular ejection function (%) |
70.0 ± 2.6 |
48.0 ± 2.8 |
67.7 ± 9.4 |
0.018 |
| Prior myocardial infarction (n (%)) |
1 (33) |
0 (0) |
3 (13) |
0.568 |
| Prior coronary artery bypass graft surgery (n (%)) |
0 (0) |
0 (0) |
0 (0) |
- |
| Prior percutaneous coronary intervention (n (%)) |
0 (0) |
0 (0) |
3 (13) |
1.000 |
| Aspirin (n (%)) |
3 (100) |
2 (100) |
23 (100) |
- |
| Clopidogrel (n (%)) |
3 (100) |
2 (100) |
23 (100) |
- |
| β-blocker (n (%)) |
2 (67) |
2 (100) |
11 (48) |
0.600 |
| Statin (n (%)) |
1 (33) |
2 (100) |
19 (83) |
0.144 |
| Angiotensin converting enzyme inhibitor (n (%)) |
0 (0) |
0 (0) |
0 (0) |
- |
| Angiotensin receptor blocker (n (%)) |
2 (67) |
2 (100) |
12 (52) |
0.613 |
| Number of disease vessels |
1.7 ± 0.6 |
1.0 ± 0.0 |
1.8 ± 0.8 |
0.335 |
CTO, chronic total occlusion
Table 1: Patient characteristics.
| |
CTO subintima group
(n=5) |
CTO true lumen group
(n=4) |
non-CTO group
(n=35) |
P value |
| Stent diameter (mm) |
3.00 ± 0.41 |
2.75 ± 0.25 |
2.84 ± 0.33 |
0.531 |
| Stent length (mm) |
22.8 ± 7.5 |
26.0 ± 4.5 |
21.4 ± 5.0 |
0.178 |
| Max inflation pressure (atm) |
16.3 ± 6.8 |
14.0 ± 6.4 |
16.2 ± 5.2 |
0.697 |
| Time to OCT examination (months) |
8 (8-8) |
8 (8-8) |
8 (7-8) |
0.576 |
| Lesion |
|
0.028 |
| Left anterior descending artery (n (%)) |
0 (0) |
2 (40) |
16 (46) |
|
| Left circumflex (n (%)) |
1 (25) |
0 (0) |
11 (31) |
|
| Right coronary artery (n (%)) |
3 (75) |
3 (60) |
8 (23) |
|
| Type of OCT |
|
0.147 |
| Time domain-OCT (n (%)) |
4 (100) |
3 (60) |
32 (91) |
|
| Frequency domain-OCT (n (%)) |
0 (0) |
2 (40) |
3 (9) |
|
CTO, chronic total occlusion
OCT, optical coherence tomography
Table 2: Lesion and stent characteristics.
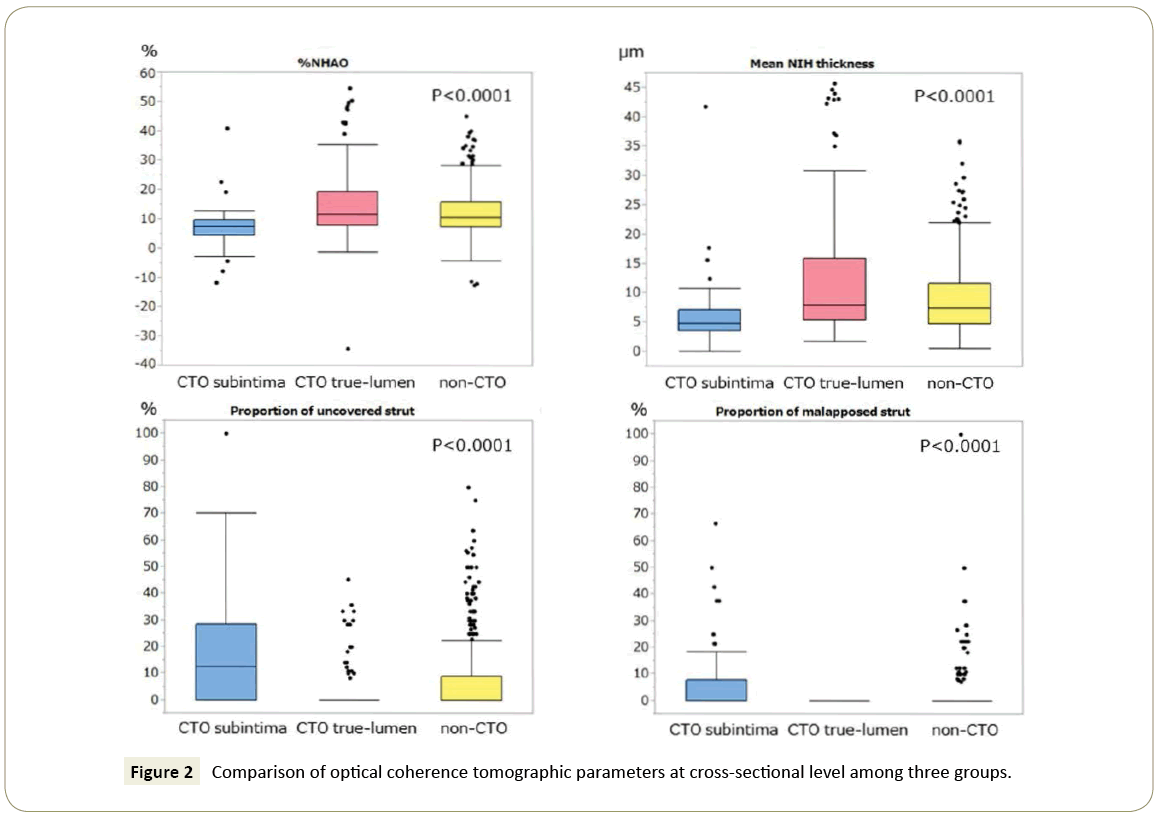
Figure 2: Comparison of optical coherence tomographic parameters at cross-sectional level among three groups.
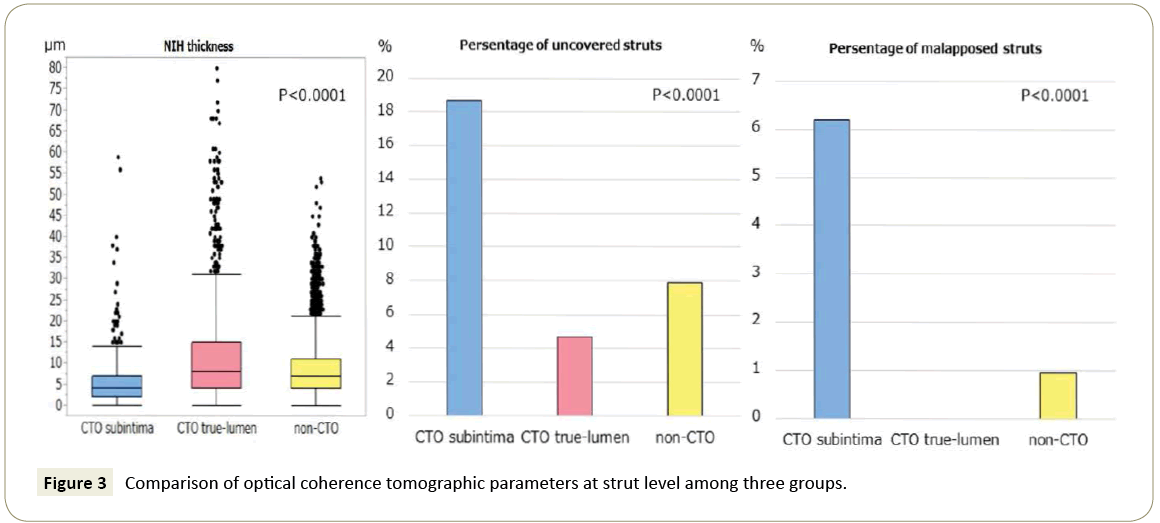
Figure 3: Comparison of optical coherence tomographic parameters at strut level among three groups.
Discussion
DESs implanted in CTO subintima group were most likely to show poor apposition and thin neointima, and to remain uncovered by neointima among the three groups. Accordingly, neointimal coverage of DESs in subintima may be most delayed. Hong et al. reported that subintimal passage of guidewire and stenting of pseudo lumen caused stent malapposition [21]. Recently, Jia et al. also reported that CTO is associated with a higher incidence of malapposition and uncovered stent struts according to OCT findings at 6 months following DES implantation [22]. These two-data corresponded to our results. On the other hand, DESs in CTO true-lumen group revealed best apposition, fewest uncovered struts and thickest neointima among the three groups. Therefore, neointima in CTO true-lumen may grow most rapidly. Stent implantation leads to mechanical injury with vascular inflammation, which involves complex interactions between endothelial cells, smooth muscle cells, platelets, and inflammatory cells [23]. These cells produce signalling molecules, which stimulate mobilization of bone marrow-derived endothelial progenitor cells, and smooth muscle progenitor cell. The cellular and molecular processes control vascular healing after stent implantation in non-CTO lesion [23]. It has been reported that CTO lesions develop from a total luminal obstruction of coronary arteries by a thrombus, subsequently are organized by inflammatory cells followed by infiltrating smooth muscle cells, and generate proteoglycan matrix as well as calcification [24,25]. Thus, true-lumen in CTO lesion is pathologically different from true-lumen in non-CTO lesion. Subintima has completely different pathological feature from intima. Most importantly, endothelial cells do not exist in subintima. The difference of neointimal coverage of stents might be caused by the different environment where stent implanted, that is to say, subintimal stenting or intra true-lumen stenting for CTO lesion or stenting for non-CTO lesions. The delayed neointimal coverage of DES in subintima leads to more uncovered and malapposed struts, which are reported to cause stent thrombosis [26,27]. On the other hand, the thicker neointimal coverage of DES in truelumen in CTO lesions might lead to in-stent restenosis. Heeger et al. reported that significantly delayed DES coverage after CTOPCI was observed, but they did not evaluate whether DES was implanted in subintima or in true-lumen using IVUS or OCT [28]. In addition, their study used different type DESs while we used only EES. Thus, we believe our study can reveal pure effect of stenting for CTO lesion on neointimal growth including difference between subintimal stenting and intra true-lumen stenting. According to our results, neointimal coverage of DES (EES) implanted for CTO lesions showed more heterogeneous patterns depending on subintimal stenting or intra true-lumen stenting as compared to non-CTO lesions.
Recently, bio resorbable vascular scaffolds (BVS) can be available in clinical practice. The real-world outcomes of everolimussluting BVS showed acceptable rates of target lesion failure at 6 months and low incidences of cardiac death and reinfarction [29]. In addition, OCT revealed BVS had disappeared and had been replaced by connective proteoglycans and occasional smooth muscle cells with the restoration of normal vasomotor tone for 2 years after stenting, which is called ‘vascular reparative therapy’ [30]. Thus, although clinical trials on the effectiveness of everolimus-sluting BVS in patients with complex coronary artery disease including CTO have not yet to be completed, BVS may be a therapeutic option in patients with CTO [31].
It has been reported that EES is one of the best stents among currently used DESs, showing reduced restenosis and stent thrombosis [32,33]. However, even using EES, a good clinical course after stent implantation for CTO lesions is not guaranteed. To best of our knowledge, this is the first report describing different neointimal characteristics of stent position including subintimal stenting and intra true-lumen stenting for CTO lesions and stenting for non-CTO lesions.
Limitations
The CTO group is limited to only 5 patients and 9 stents. The number of patients and stents is small. However, the number of struts which could be evaluated by OCT may be enough to induce a kind of trend of difference between CTO lesions and non-CTO lesions. We analysed 1581 struts in CTO group and 4747 struts in non-CTO group. Post-hoc sample size calculation revealed 444 samples to reach the significance. Thus, we believe the sample size of our study may be acceptable when we use the strutsanalysis. Second, in CTO subintima group, all parts of stents were no implanted in subintima; some parts of stents were implanted in subintima, but the other parts of stents were implanted in true lumen. We could not distinguish whether the cross section of the stents were in subintima or true lumen by OCT examination at the follow-up angiography.
Conclusions
Observation of EES stents by OCT revealed that stents implanted in subintima in CTO lesion may have risks related to stent thrombosis such as malapposed struts and uncovered struts, while stents implanted in true-lumen in CTO lesion may have thick neointimal coverage that might occur restenosis. This might be the one reason of high frequency of stent reocclusion and restenosis in CTO lesion after stent implantation.
References
- George S, Cockburn J, Clayton TC, Ludman P, Cotton J, et al. (2014) Long-term follow-up of elective chronic total coronary occlusion angioplasty: Analysis from the UK central cardiac audit database. J Am Coll Cardiol 64:235-243.
- Valenti R, Migliorini A, Signorini U, Vergara R, Parodi G, et al. (2008) Impact of complete revascularization with percutaneous coronary intervention on survival in patients with at least one chronic total occlusion. Eur Heart J 29:2336-2342.
- Hoye A, Tanabe K, Lemos PA, Aoki J, Saia F, et al. (2004) Significant reduction in restenosis after the use of sirolimus-eluting stents in the treatment of chronic total occlusions. J Am Coll Cardiol 43:1954-1958.
- Suero JA, Marso SP, Jones PG, Laster SB, Huber KC, et al. (2001) Procedural outcomes and long-term survival among patients undergoing percutaneous coronary intervention of a chronic total occlusion in native coronary arteries: A 20-year experience. J Am Coll Cardiol 38:409-414.
- Noguchi T, Miyazaki MS, Morii I, Daikoku S, Goto Y, et al. (2000) Percutaneous transluminal coronary angioplasty of chronic total occlusions. Determinants of primary success and long-term clinical outcome. Catheter Cardiovasc Interv 49:258-264.
- Michael TT, Mogabgab O, Alomar M, Kotsia A, Christopoulos G, et al. (2014) Long-term outcomes of successful chronic total occlusion percutaneous coronary interventions using the antegrade and retrograde approach. J Interv Cardiol 27:465-471.
- Takano M, Murakami D, Yamamoto M, Kurihara O, Murai K, et al. (2013) Six-month follow-up evaluation for everolimus-eluting stents by intracoronary optical coherence tomography: Comparison with paclitaxel-eluting stents. Int J Cardiol 166:181-186.
- Gao L, Wang Y, Liu Y, Cao F, Chen Y (2017) Long-term clinical outcomes of successful revascularization with drug-eluting stents for chronic total occlusions: A systematic review and meta-analysis. Catheter Cardiovasc Interv7:574-581
- Patel VG, Brayton KM, Tamayo A, Kiemeneij F, Bal ET, et al. (2013) Angiographic success and procedural complications in patients undergoing percutaneous coronary chronic total occlusion interventions: A weighted meta-analysis of 18,061 patients from 65 studies. JACC Cardiovasc Interv 6:128-136.
- Morino Y, Kimura T, Hayashi Y, Muramatsu T, Ochiai M, et al. (2010) In-hospital outcomes of contemporary percutaneous coronary intervention in patients with chronic total occlusion insights from the J-CTO Registry (Multicenter CTO Registry in Japan). JACC Cardiovasc Interv 3:143-1451.
- Rahel BM, Laarman GJ, Kelder JC, Ten Berg JM, Suttorp MJ (2009) Three-year clinical outcome after primary stenting of totally occluded native coronary arteries: A randomized comparison of bare-metal stent implantation with sirolimus-eluting stent implantation for the treatment of total coronary occlusions (Primary Stenting of Totally Occluded Native Coronary Arteries [PRISON] II study). Am Heart J 157:149-155.
- De Felice F, Fiorilli R, Parma A, Nazzaro M, Musto C, et al. (2009) 3-year clinical outcome of patients with chronic total occlusion treated with drug-eluting stents. JACC Cardiovasc Interv 2:1260-1265.
- Lee SP, Kim SY, Park KW, DongHo S, Hyun-Jae K,et al. (2010) Long-term clinical outcome of chronic total occlusive lesions treated with drug-eluting stents. Circ J 74:693-700.
- Garcia S, Abdullah S, Banerjee S, Brilakis ES (2013) Chronic total occlusions: Patient selection and overview of advanced techniques. Curr Cardiol Rep 15:334.
- Godino C, Latib A, Economou FI, Al-Lamee R, Ielasi A, et al. (2012) Coronary chronic total occlusions: Mid-term comparison of clinical outcome following the use of the guided-STAR technique and conventional anterograde approaches. Catheter Cardiovasc Interv 79:20-27.
- Valenti R, Vergara R, Migliorini A, Parodi G, Carrabba, N. et al. (2013) Predictors of reocclusion after successful drug-eluting stent-supported percutaneous coronary intervention of chronic total occlusion. J Am Coll Cardiol 61:545-550.
- Ishigami K, Uemura S, Morikawa Y, Soeda T, Okayama S, et al. (2009) Long-term follow-up of neointimal coverage of sirolimus-eluting stents: Evaluation with optical coherence tomography. Circ J 73:2300-2307.
- Gonzalo N, Tearney GJ, Serruys PW, van Soest G, Okamura T, et al. (2010) Second-generation optical coherence tomography in clinical practice. High-speed data acquisition is highly reproducible in patients undergoing percutaneous coronary intervention. Rev Esp Cardiol 63:893-903.
- Tearney GJ, Regar E, Akasaka T, Adriaenssens T, Barlis P, et al. (2012) Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International working group for intravascular optical coherence tomography standardization and validation. J Am Coll Cardiol 59:1058-1072.
- Inoue T, Shite J, Yoon J, Shinke T, Otake H, et al. (2011) Optical coherence evaluation of everolimus-eluting stents 8 months after implantation. Heart 97:1379-1384.
- Hong MK, Mintz GS, Lee CW, Park DW, Park KM, et al. (2006) Late stent malapposition after drug-eluting stent implantation: an intravascular ultrasound analysis with long-term follow-up. Circulation 113:414-419.
- Jia H, Hu S, Liu H, Zhu Y, Zhe CY, et al. (2017) Chronic total occlusion is associated with a higher incidence of malapposition and uncovered stent struts: Oct findings at 6 months following des implantation. Catheter Cardiovasc Interv 89:582-591.
- Inoue T, Croce K, Morooka T, Sakuma M, Node K, et al. (2011) Vascular inflammation and repair: Implications for re-endothelialization, restenosis, and stent thrombosis. JACC Cardiovasc Interv 4:1057-1066.
- Munce NR, Strauss BH, Qi X, Weisbrod MJ, Anderson KJ, et al. (2010) Intravascular and extravascular microvessel formation in chronic total occlusions a micro-CT imaging study. JACC Cardiovasc Imaging 3:797-805.
- Finn AV, Kolodgie FD, Nakano M, Virmani R (2010) The differences between neovascularization of chronic total occlusion and intraplaque angiogenesis. JACC Cardiovasc Imaging 3:806-810.
- Finn AV, Joner M, Nakazawa G, Kolodgie F, Newell J, et al. (2007) Pathological correlates of late drug-eluting stent thrombosis: strut coverage as a marker of endothelialization. Circulation 115:2435-2441.
- Cook S, Wenaweser P, Togni M, Billinger M, Morger C, et al. (2007) Incomplete stent apposition and very late stent thrombosis after drug-eluting stent implantation. Circulation 115:2426-2434.
- Heeger CH, Busjahn A, Hildebrand L,Fenski M, Lesche F, et al. (2016) Delayed coverage of drug-eluting stents after interventional revascularisation of chronic total occlusions assessed by optical coherence tomography: The ALSTER-OCT-CTO registry. EuroIntervention 11:1004-1012.
- Capodanno D, Gori T, Nef H, Latib A, Mehilli J, et al. (2015) Percutaneous coronary intervention with everolimus-eluting bioresorbable vascular scaffolds in routine clinical practice: Early and midterm outcomes from the European multicentre ghost-euregistry. EuroIntervention10:1144-1153
- Onuma Y, Serruys PW, Perkins LE, Okamura T, Gonzalo N, et al. (2010) Intracoronary optical coherence tomography and histology at 1 month and 2, 3, and 4 years after implantation of everolimus-eluting bioresorbable vascular scaffolds in a porcine coronary artery model: An attempt to decipher the human optical coherence tomography images in the absorb trial. Circulation 122:2288-2300.
- Devito F, Zito A, Dachille A, Carbonara R, Giardinelli F, et al. (2016) Bioresorbable vascular scaffolds: Design, clinical trials, and current applications. Coron Artery Dis 27:151-158
- Sabate M, Cequier A, Iniguez A, Serra A, Hernandez-Antolin R, et al. (2012) Everolimus-eluting stent versus bare-metal stent in ST-segment elevation myocardial infarction (EXAMINATION): 1 year results of a randomised controlled trial. Lancet 380:1482-1490.
- Palmerini T, Biondi-Zoccai G, Della Riva D, Stettler C, Sangiorgi D, et al. (2012) Stent thrombosis with drug-eluting and bare-metal stents: evidence from a comprehensive network meta-analysis. Lancet 379:1393-1402.