Research Article - (2018) Volume 2, Issue 1
Alanoud Alotaibi, Adriana Modesto and Alexandre R Vieira*
Departments of Oral Biology and Pediatric Dentistry, University of Pittsburgh School of Dental Medicine, USA
*Corresponding Author:
Alexandre R Vieira
Departments of Oral Biology and Pediatric Dentistry
University of Pittsburgh School of Dental Medicine, USA
Tel: +(412) 383-8972
E-mail: arv11@pitt.edu
Received Date: March 03, 2018; Accepted Date: March 14, 2018; Published Date: March 24, 2018
Citation: Alotaibi A, Modesto A, Vieira AR (2018) Non-Surgical Periodontal Therapy Impact on Progression of Chronic Periodontitis and Prevention of Further Tooth Loss. J Ora Med Vol.2 No.1:3
Objective: Prevention of tooth loss and maintaining favorable periodontal status are the ultimate goals of periodontal therapy. The aim of this study was to evaluate the effectiveness of non-surgical periodontal therapy and supportive periodontal care in arresting the progression of chronic periodontitis and in preventing tooth loss. Materials and methods: Periodontal charts, self-reported medical history, and two interleukin-1 (IL-1) polymorphism genotypes of 100 patients were obtained from the University of Pittsburgh School of Dental Medicine Dental Registry and DNA Repository after screening 4,825 subjects. We used tooth loss (TL) and clinical attachment loss (CAL) as outcomes of disease affection in our analysis. In our study, we have included third molars, teeth lost during active periodontal treatment (APT), and those lost during the supportive periodontal program. All subjects had at least 12 months of follow-up. Fisher’s exact test was used to investigate the association between tooth loss and different risk factors. Paired t-test was conducted to detect the difference in means of CAL at baseline and at final assessment. Results: There were 59 patients (36 males and 23 females with an average age of 52 years) that lost at least one tooth during supportive periodontal care. Tooth mortality rate declined by more than half in patients who enrolled in a supportive periodontal program for six years compared to those who dropped out after one year (0.52 vs. 3.4 teeth/patient/year, respectively). Increased risk of tooth loss was found to be associated with diabetes (p=0.01), as well as high blood pressure (p<0.0001). We did not find an association between tooth loss and polymorphisms in interleukin IL-1α/IL-1β (rs1800587, p=0.36 and rs1143634, p=0.51, respectively). During the first and the second year of supportive periodontal treatment, the clinical attachment loss decreased by 0.36 mm and 0.34 mm, respectively (p=0.025 and p=0.0697), when compared to baseline measurements. Moreover, a slight increase in CAL was found in the group of patients who attended regular periodontal maintenance program for six years (p=0.037, difference of 0.38 mm in comparison to the baseline measurement). Conclusion: Our findings suggested that supportive periodontal therapy is effective for the long-term preservation of teeth and the stability of periodontal tissue in patients with advanced chronic periodontitis.
Keywords
Supportive periodontal therapy; Tooth loss; Chronic periodontitis
Introduction
Periodontal disease (periodontitis) can be defined as a group of inflammatory diseases that affect the tooth-supporting tissues in response to microbial pathogens and can lead to tooth loss if left untreated. The clinical diagnosis of periodontitis is based on the presence of gingival inflammation, periodontal pockets, loss of clinical attachment and alveolar bone loss [1,2].
Loss of clinical attachment level (CAL) is considered the gold standard for the diagnosis and the measurement of past periodontal disease activity [3]. Moreover, CAL is found to be more accurate in measuring of periodontal disease progression compared to periodontal pocket depth [2].
Attachment loss and destructive periodontitis are more prevalent in males than females and are more prevalent in Blacks and Mexican Americans than Whites. The latest report on the prevalence of periodontitis in the United States revealed that 46% of US adults who were aged ≥30 years had periodontitis with 8.9% of them diagnosed with severe periodontitis [4].
Bacterial biofilm is the primary etiological factor for the initiation of gingivitis and subsequent destruction of periodontal tissues [5]. However, the indirect damage from periodontal diseases occurs as a result of the protective host response [6,7].
The effectiveness of non-surgical therapy regarding clinical parameters such as changes in clinical attachment level, probing pocket depth, and bleeding on probing for patients with chronic periodontitis has been discussed in many studies [8-10]. Chronic periodontal disease can be successfully treated, even in advanced stages by non-surgical or surgical periodontal therapy with adequate plaque control that can be maintained during the supportive care program [11,12].
A twelve-year longitudinal study on evaluating the effectiveness of supportive periodontal therapy (SPT) on periodontal disease progression revealed that SPT can prevent tooth loss and maintain the stability of bone and attachment loss among subjects with normal susceptibility to periodontal disease (with mean overall attachment loss of 0.5 mm, i.e. 0.04 mm/tooth surface/year). On the other hand, highly susceptible patients who receive a similar SPT experienced some tooth loss and lost significant amounts of bone and attachment (CAL loss of 0.8 mm, i.e., 0.06 mm/tooth surface/year) [13].
It was reported that patients treated for advanced periodontitis continued to lose teeth despite maintenance care, and tooth loss was signifiÂcantly more prevalent among smokers [14]. Poorly compliant patients should be considered to be at a higher risk of periodontal disease progression and tooth loss (TL). A recent study has investigated the impact of irregular compliance to periodontal maintenance on tooth loss. It was found that individuals with irregular compliance exhibit a significantly higher rate of TL (0.36 teeth lost/year) compared with regular compliance individuals (0.12 teeth lost/year). Individuals that were > 55 years old, males, and smokers lost significantly more teeth in both groups under the SPT program for five years [15]. The effectiveness of SPT was investigated in many retrospective studies. One study reported that the mean tooth loss among SPT compliant patients for at least ten years was 0.07 teeth/year [16].
In this study, we assessed patient-related factors that contribute to tooth loss during supportive periodontal therapy. We also assessed the prevalence and the severity of clinical attachment loss of periodontal tissue and tested the changes in clinical attachment level (CAL) under maintenance care. Finally, we evaluated the association between tooth loss and I-L1 genotypes.
Subjects and Methods
The Dental Registry and DNA Repository is a project that was established in 2006, in which every patient that seeks treatment at the University of Pittsburgh, School of Dental Medicine receives an invitation to participate. Upon agreeing, all individuals sign a consent form authorizing the retrieval of information from their dental records and provide a saliva sample. The study is approved by University of Pittsburgh Institutional Review Board (IRB approval#0606091). For this study, subjects were recruited from the Dental Registry and DNA Repository and they were selected based on the following main criteria:
• Patients that have been diagnosed with moderate to severe chronic periodontitis (participants with 30% or more teeth with sites of clinical attachment loss of five millimeters or more).
• The participant should have received at least two periodontal assessments after completion of active periodontal therapy (APT).
• The patient must have been attended regular supportive periodontal therapy for a period of 12 months.
Upon the screening of 4,825 subjects, 100 participants met the selection criteria. Most patients in the registry come from patients receiving restorative treatments and very few come from specialty clinics, which explain the number of cases identified receiving periodontal treatment. The data included the number of missing teeth and clinical attachment loss, as well as demographic and health status information that was extracted from the dental records. Of all of the 100 enrolled patients, 69 were White, 29 were Black, and one patient was of Asian origin and another of Hispanic origin. There were 46 females and 54 males in the total sample. At the time of the initial examination, the average age was 53.07 years old and ranged from 20 to 91 years old. The mean follow-up period was three years. All subjects had at least a 12-month follow-up period. Twenty-six subjects had one year of follow-up, 13 subjects had two years, 17 subjects had three years, 20 subjects had four years, eight had five years, and 16 had six years of follow-up.
We used the case definition that was developed by the Centers for Disease Control and Prevention (CDC) Periodontal Disease Surveillance Workgroup. Severe periodontitis is defined as the presence of two or more interproximal sites with CAL ≥6 mm, not on the same tooth, and one or more interproximal sites with Probing Depth (PD) ≥5 mm.
Moderate periodontitis is defined as the presence of two or more interproximal sites with CAL ≥4 mm, not on the same tooth, or two or more interproximal sites with PD ≥5 mm, not on the same tooth [2]. Periodontal diagnosis was classified according to the American Academy of Periodontology guidelines [17]. The severity of periodontal disease is based on the amount of clinical attachment loss (CAL) and is designated as mild (1-2 mm CAL), moderate (3-4 mm CAL), or severe (>5 mm CAL). Periodontitis is classified as localized if 30% or less of periodontal tissue is affected and generalized if more than 30% of the tissue is affected. The clinical attachment level of all six points (the mesial, the mid, and the distal points of both buccal and lingual surfaces) of each standing tooth, including the third molars, was recorded for every periodontal evaluation the patients underwent.
Genomic DNA was extracted from unstimulated saliva according to previously published protocol [18] and genotypes were generated using Taqman chemistry [19]. The reactions were carried out with the use of standard conditions as suggested by the manufacturer.
All statistical analyses were performed using R programming language at 5% significance level and 95% confidence interval. Fisher’s exact test was used to determine the relationship between tooth loss and the different risk factors: age, sex, ethnicity, self-reported overall health, and smoking history (current, former, or never). Paired t-test was conducted to detect the difference between baseline and final CAL measurements (buccal, lingual, and full mouth CAL) in the patients at different time interval.
Results
Tooth loss and clinical attachment
In order to assess the long-term effectiveness of the non-surgical periodontal therapy, both tooth loss and clinical attachment loss levels were used as an outcome of periodontal disease. Based on the tooth loss category, subjects who lost one or more teeth were compared to individuals who maintained zero tooth loss. Third molars, teeth lost during active periodontal treatment (APT), and teeth lost during the supportive periodontal care program (SPC) were included in this study. Time interval or follow-up periods referred to the time from the baseline periodontal evaluation to the final periodontal evaluation. Out of 100 patients, 59 of the participants (36 males and 23 females, with an average age of 52 years of age) lost at least one tooth. On the other hand, the 41 subjects (18 males and 23 females, with an average age of 54 years of age) did not lose a tooth and were used as comparison. According to baseline periodontal evaluations, there were 2,482 present teeth and 718 missing teeth. Molars were the most frequently missing teeth, whereas canines were the least commonly missing teeth among all participants (Figure 1). Throughout the follow-up period, subjects who lost one or more teeth (59 patients) lost 188 teeth, which added 20.8% to the total number of missing teeth at the final periodontal evaluation. About 39.1% of the total missing teeth at the initial periodontal evaluation and 31% at the final periodontal evaluation came from the control group. During the active periodontal therapy (APT), 68 teeth were extracted (mean tooth loss of 0.68 teeth/patient). This number increased to 120 teeth during the supportive periodontal care program (mean tooth loss of 1.2 teeth/patient). Molars were lost at a higher frequency compared with incisors during APT as well as SPC (Figure 2). Tooth mortality rate per patient per year declined by more than half with increasing length of the supportive periodontal care. This can be explained by reduction of the rate of periodontal assessment per patient over time.
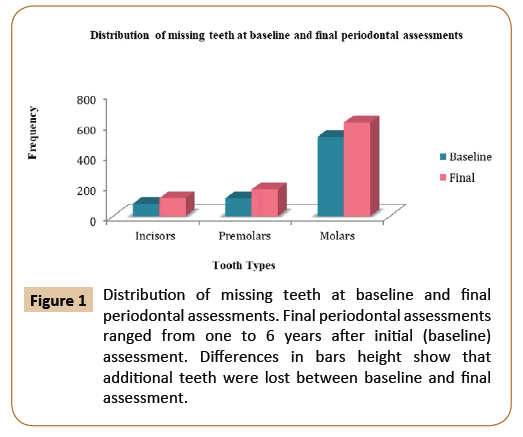
Figure 1: Distribution of missing teeth at baseline and final periodontal assessments. Final periodontal assessments ranged from one to 6 years after initial (baseline) assessment. Differences in bars height show that additional teeth were lost between baseline and final assessment.
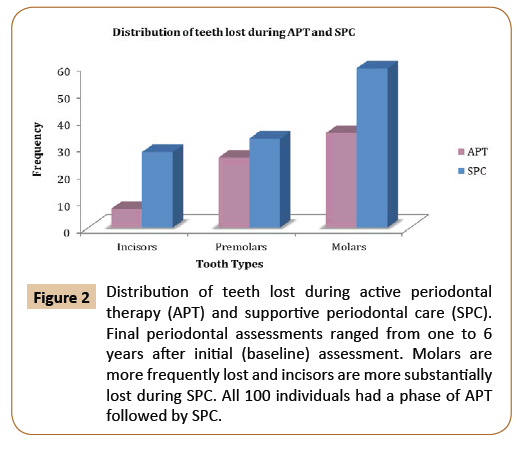
Figure 2: Distribution of teeth lost during active periodontal therapy (APT) and supportive periodontal care (SPC). Final periodontal assessments ranged from one to 6 years after initial (baseline) assessment. Molars are more frequently lost and incisors are more substantially lost during SPC. All 100 individuals had a phase of APT followed by SPC.
Overall Health Issues
Frequency of concomitant disease and having had or not tooth loss was compared by using the Fisher’s exact test. We found more tooth loss in individuals with diabetes and hypertension (p=0.014 and p=6.7 26e-07, respectively) (Table 1).
Table 1 Frequency of tooth loss and history of overall health issues.
| Variable | Tooth Loss=0 | Tooth Loss>0 | Fisher’s exact test |
|---|---|---|---|
| With Diabetes | 9 | 3 | 0.01 |
| Without Diabetes | 32 | 56 | |
| Healthy | 9 | 14 | 1 |
| Systemic disease | 32 | 45 | |
| With epilepsy | 0 | 3 | 0.27 |
| Without epilepsy | 41 | 56 | |
| With stroke | 2 | 2 | 1 |
| Without stroke | 39 | 57 | |
| With asthma | 7 | 8 | 0.78 |
| Without asthma | 34 | 51 | |
| With tuberculosis | 2 | 2 | 1 |
| Without tuberculosis | 39 | 57 | |
| With sickle cell anemia | 2 | 2 | 1 |
| Without sickle cell anemia | 39 | 57 | |
| With hepatitis | 2 | 2 | 1 |
| Without hepatitis | 39 | 57 | |
| With high blood pressure | 18 | 2 | 6.7 26e-07 |
| Without high blood pressure | 23 | 57 | |
| With cancer | 5 | 8 | 1 |
| Without cancer | 36 | 51 | |
| Female | 23 | 23 | 0.11 |
| Male | 18 | 36 | |
| Black | 14 | 15 | 0.14 |
| Other | 2 | 0 | |
| White | 25 | 44 | |
| Smoker | 7 | 15 | 0.5 |
| Non-smoker | 29 | 35 | |
| Former smoker | 5 | 9 |
Interleukin 1 (IL-1) genetic variants
IL-1 genotype frequencies were comparing in subjects who lost one or more teeth with individuals who maintained zero tooth loss using PLINK Whole genome association analysis software version 1.9. No significant difference in distribution of alleles and genotypes were observed between any of the IL-1 SNPs and tooth loss (rs1800587, p=0.3641; and rs1143634, p=0.5132). These findings were confirmed by using Fisher’s exact test to test for association under a dominant model between tooth loss and the genotypes containing the minor allele A in both SNPs (Table 2).
Table 2 Comparisons for IL-1 genotypes.
| SNP/genotypes | Test/model | At least one tooth lost | No tooth loss | Fisher’s exact test |
|---|---|---|---|---|
| IL1-A (rs1800587) | ||||
| AA/AG/GG | Genotype | 7/30/21 | 4/18/17 | 0.76 |
| AA + AG vs. GG | Dominant model | 37/21 | 22/17 | 0.53 |
| AA vs. AG + GG | Recessive model | 7/51 | 4/35 | 1.0 |
| IL1-B (rs1143634) | ||||
| AA/AG/GG | Genotype | 4/21/33 | 2/12/27 | 0.67 |
| AA + AG vs. GG | Dominant model | 25/33 | 14/27 | 0.41 |
| AA vs. AG + GG | Recessive model | 4/54 | 2/39 | 1.0 |
Analyses by age
Of all participants, 18% were aged 20 - 39 years old (younger group) and 82% were aged 40- 91 years old (older group). Comparing situations at the baseline between the two groups, the number of missing teeth was higher in the older group (645 missing teeth in the older group compared with 73 in the younger group). The number of missing teeth continued to be larger for the older group at the final periodontal evaluation and the total tooth loss over the entire follow up period was 167 in the older group compared to 13 in younger group (Figure 3).
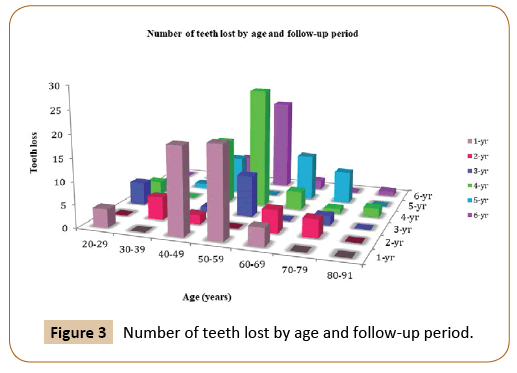
Figure 3: Number of teeth lost by age and follow-up period.
Analyses based on disease progression
Of the 14,527 recorded measurements (missing sites were excluded), 196 (1.35%) sites were classified as healthy, having no active periodontitis, yet 6,979 (48.04%), 5,762 (39.7%), and 1,590 sites (10.9%) were included in the study defined with mild, moderate, or severe periodontitis, respectively.
Based on the differences in clinical attachment (final CALinitial CAL), the patients were categorized into three groups: progressive, regressive, and stable. The progressive group included the patients who showed an increase in CAL during SPC (CAL difference ≥+1). The regressive groups consisted of patients who showed a decrease in CAL (CAL difference ≤-1). The patients who showed CAL difference of zero belonged to the stable group. There were 35 subjects who showed progressive clinical attachment loss, 29 subjects showed regressive clinical attachment loss, and 36 subjects were in the stable group.
Paired t-test was used to measure the difference between the clinical attachment loss before and after the treatment at different time intervals. The mean difference of clinical attachment level at one, two and six-year intervals were 0.36, 0.343, and -0.38 respectively, and they were found to be significant only at those time interval (p=0.025, p=0.0697, and p=0.0373). By comparing the mean difference of buccal CAL at the baseline and final periodontal evaluation, there was a significant difference at one, two and six-year intervals, although the mean difference is small (p=0.0042, p=0.0598 and p=0.0816, respectively). On the other hand, lingual CAL showed close to significant difference in mean value among those who have been followed-up for 5 years (p=0.0586) (Figures 4 and 5).
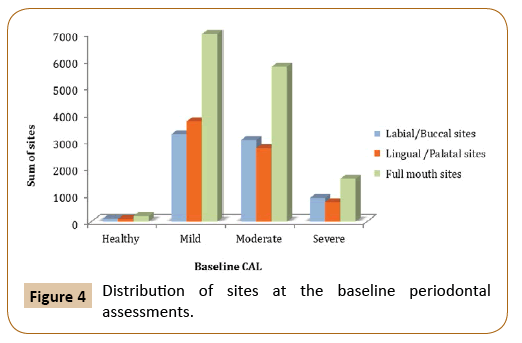
Figure 4: Distribution of sites at the baseline periodontal assessments.
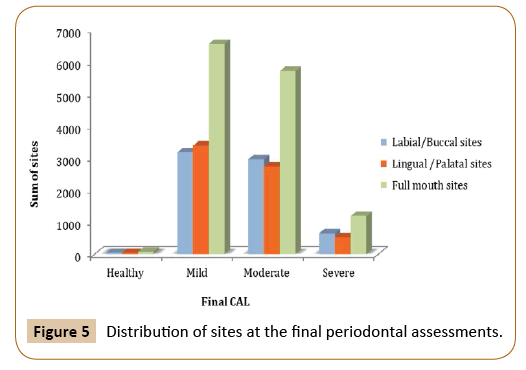
Figure 5: Distribution of sites at the final periodontal assessments.
Discussion
It has been established that regular, professional prevention programs can be effective in the prevention of caries and periodontal disease and subsequent tooth loss in adults. The assessment of the risk level for periodontal disease progression in patients would help the dental practitioners to determine the frequency of SPT visits and the extent of professional support necessary for each patient. It has been suggested that patients with advanced periodontitis may need an SPT visit at short time interval (3-4 months). While for mild-to-moderate forms of periodontitis, one annual visit may be enough to prevent further clinical attachment loss [20].
In this study, we have used tooth loss as a measurement of the effectiveness of supportive periodontal therapy. Tooth loss is considered a true clinical end point and that makes it the most relevant parameter to evaluate the efficacy of dental treatment. In order to make a precise measurement of incidence of tooth loss, all teeth that had been lost, including those that were extracted during the active phase of treatment, were included in this study. Many studies have investigated the association between periodontal disease, tooth loss, and several systemic diseases, including diabetes mellitus, cancer, and cardiovascular disease [12].
The study was designed to assess the effectiveness of nonsurgical periodontal therapy taking into consideration risk factors that contribute in the modulation of the host response in patients with periodontitis. The data showed that tooth loss is more likely to happen in diabetes as well as hypertension. These findings are consistent with previous studies [21,22].
Non-surgical periodontal therapy induces beneficial changes to periodontal tissues and does not appear to depend on genetic variation in IL-1. Improvement of clinical parameters can be seen as the reduction of gingival inflammation, reduction of periodontal pocket depth, and gain in clinical attachment level. Clinical attachment loss is a useful clinical parameter that indicates the presence of periodontitis, but not necessarily the activity of the diseases.
In the present study, the difference in means of CAL at baseline and at the final periodontal evaluation was found to be statistically significant among the subjects in the first two years of supportive treatment (p=0.025 and p=0.0697). However, the clinical changes in CAL were minimal (0.36 mm and 0.343 mm gain in CAL). Patients who underwent periodontal maintenance for six years showed a statistically significant difference in CAL (p=0.0373) with 0.38 mm progression in CAL. This change can be attributed to the interaction between local factors or systemic disease that can modify the rate of periodontitis progression.
Conclusion
The study showed that non-surgical periodontal therapy and supportive care programs alone could be efficient for long-term management of periodontal stability in high-risk patients.
Acknowledgements
Data used here was obtained from the University of Pittsburgh School of Dental Medicine Dental Registry and DNA Repository.