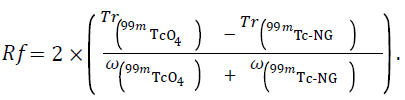Bérenger L1*, Anne-Claire D1,2, Nicolas A1,2 and Maia S1,2
1Radiopharmacy Unit, Hospital of Bretonneau, University Hospital Center of Tours, Boulevard Tonnellé, 37044 Tours, France
2National Institute of Health and Medical Research U930, 10 Boulevard Tonnelle, 37044 Tours, France
Corresponding Author:
Bérenger L
Radiopharmacy Unit, Hospital of Bretonneau
University Hospital Center of Tours, Boulevard Tonnellé
37044 Tours, France
Tel: +33-(0)2-47-47-97-07
E-mail: berenger.largeau@etu.univ-tours.fr
Received Date: June 12, 2017; Accepted Date: June 21, 2017; Published Date: June 30, 2017
Citation: Largeau B, Dupont AC, Guilloteau D, Nicolas A, Serge M. Optimized Method for the Radiochemical Purity Quality Control of 99mTc-MERTIATIDE – NEPHROMAG™. J Pharm Pharm Res. 2017, 1:7.
Copyright: © 2017 Largeau B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
99mTc-NephroMAG™ is a radiopharmaceutical which is used for dynamic renal imaging. The summary of product characteristics (SPC) specifies that the determination of radiochemical purity must be achieved via high pressure liquid chromatography or Sep-Pak™ C18 cartridge methods. To minimize the time required to test radiochemical purity, a method with radio-thin-layer chromatography (r-TLC) was used in radiopharmacy unit of Tours CHRU. However, this method on Whatman™ 3mm was too long and offered no optimized resolving parameters.
Methods: For each preparation (n=5), two strips r-TLC with Tec-Control™ #150-005 has been achieved, first with hydrophilic mobile phase (hMP) in order to highlight colloidal technetium (99mTcO2), second with lipophilic mobile (lMP) to separate 99mTcO4
Results: The method with two kinds of mobile phase offers a better resolving power for both impurities searched and was more rapid to perform.
Conclusion: The simplified method is a reasonable alternative to the registered SPC methods and to the method with Whatman™ 3 mm chromatographic paper.
Keywords
99mTc-NephroMAG™ ; Analytic method; Radiochemical purity; Radio-thin-layer; Chromatography
Introduction
99mTc-NephroMAG™ is a radiopharmaceutical which is used for dynamic renal imaging. Isotopic nephrogram indications with 99mTc-NephroMAG™ include the evaluation of obstructive uro-/ nephropathy, diagnosis of renovascular hypertension and the early follow-up of kidney graft quality [1,2]. Before patient injection, the radiochemical purity of the radiotracer must be determined. Analytical methods described in the summary of product characteristics (SPC) of 99mTc-NephroMAG™ (i.e. high pressure liquid chromatography or Sep-Pak™ C18 cartridge) are either too much time-consuming or too irradiating for operators. In radiopharmacy unit of Tours CHRU, a radio-thin-layer chromatography (r-TLC) method on Whatman™ 3 mm, with a length of 10 cm (Fisher Scientific) and with a mobile phase composed of CH3CN/water for injection (60:40, v/v) has been developed. Nevertheless, long duration of migration (# 30 min) and poor selectivity of the method to separate free technetium (99mTcO4-) on Whatman™ chromatography paper required optimization of chromatographic parameters of this quality control.
Materials and Methods
99mTc-NephroMAG™ preparation
All radiopharmaceutical preparations (n=5) were prepared with 99mTc-pertechnetate eluted from a 99mo-99mTc generator (Mallinckrodt). 99mTc-NephroMAG™ was prepared at 800 MBq in a 2 mL quantity of NaCl 0.9% after 15 minutes of incubation time then 2 mL of commercial buffer were added. All activities were measured with a Capintec CRC®-15R dose calibrator.
Radio-thin-layer chromatographic assays
All chromatographic solvents were obtained from Sigma- Aldrich. Quality control of radiochemical purity of 99mTc- NephroMAG™ was completed by r-TLC with Mini QC-Scan™ (Bioscan) with 0.25 mm.sec-1 speed of plate during 5 minutes, detection cut-off was 2 Mega shots per minute. Quantitative analysis of chromatograms (Tr=retention time; ω=width at the base of the peak) was completed by Chromoleon™ software. For each preparation, two strips r-TLC has been achieved, first with hydrophilic mobile phase (hMP) in order to highlight colloidal technetium (99mTcO2), second with lipophilic mobile phase (lMP) to separate 99mTcO4 -. The stationary phase was Tec-Control™ #150-005 systems (Biodex), with a length of 5.5 cm. hMP was sodium citrate. lMP used were ethyl acetate (EA), methyl ethyl ketone (MEK), mix EA/CH3CN and EA/MeOH (60:40, v/v). In lMP, 2 μL of a mix of 99mTc-NephroMAG™ (99mTc-NG) with 99mTc- pertechnetate (99mTcO4 -) was extemporaneously prepared in order to bring out the resolving power of the method. Selectivity was calculated according to the equation (i)
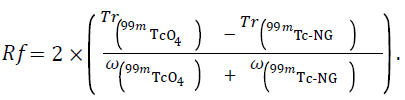
Separation was considered acceptable based on resolving factor (Rf) greater than 2 and peak profile fitted with a Gaussianlike curve. Statistical results were expressed in terms of 95% confidence interval.
Results
With sodium citrate on Tec-Control™ chromatography system the mean Tr and the mean ω 99mTc-NG were 3.74 ± 0.16 and 1.31 min ± 0.05, respectively (Figure 1).
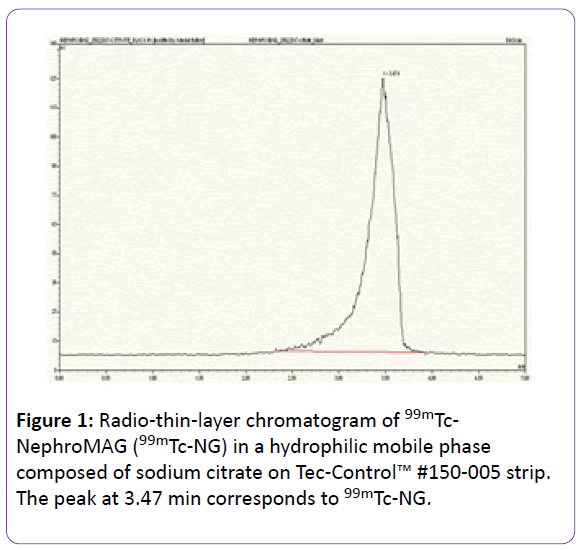
Figure 1: Radio-thin-layer chromatogram of 99mTc-NephroMAG (99mTc-NG) in a hydrophilic mobile phase composed of sodium citrate on Tec-Control™ #150-005 strip. The peak at 3.47 min corresponds to 99mTc-NG.
From prior work, in this chromatographic conditions 99mTcO2 showed mean Tr and 99mTcO2 mean ω of 0.94 min ± 0.02 and 0.8 ± 0.02 min, respectively.
In lMP, only the mixture EA/CH3CN (60:40, v/v) provided satisfactory results. Indeed, the 99mTc-NG profile peak was not a Gaussian-like curve with the following lMP: MEK, mixture EA/ MeOH (60:40, v/v). When EA was used like lMP, 99mTcO4 - exhibited a double-shouldered peak. Whereas in the mixture EA/CH3CN (60:40, v/v), the 99mTc-NG and 99mTcO4 - peak respected a Gaussian curve with mean Tr 1.05 ± 0.12 et 4.07 ± 0.13 min, respectively (Figure 2).
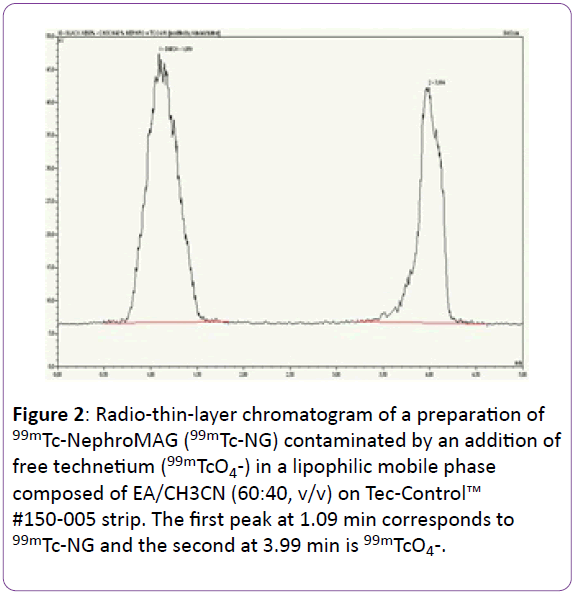
Figure 2: Radio-thin-layer chromatogram of a preparation of 99mTc-NephroMAG (99mTc-NG) contaminated by an addition of free technetium (99mTcO4-) in a lipophilic mobile phase composed of EA/CH3CN (60:40, v/v) on Tec-Control™ #150-005 strip. The first peak at 1.09 min corresponds to 99mTc-NG and the second at 3.99 min is 99mTcO4-.
According to the equation (i), the mean Rf was 3,23 ± 0.24 min. Concerning the time of quality control (migration + analysis), it was 27 ± 0.8 min for the method on Whatman™ 3 mm against only 15 ± 0.6 min for method on Tec-Control™ strip.
Table 1 summarizes and compares chromatographic conditions and results between quality control method on Whatman™ 3 mm paper and Tec-Control™ #150-005 system.
| |
Whatman™ 3 mm |
Tec-Control™ #150-005 |
| Stationary phase |
Cellulose |
Cellulose |
| Mobile phase |
CH3CN/EPPI (60:40, v/v) |
- Sodium citrate |
| - EA/CH3CN (60:40, v/v) |
| Migration time |
# 20 min |
# 5 min |
| Analysis time |
# 7 min |
# 5 min (x2) |
| Rf (99mTc-NG &99mTcO2) |
>>2 |
>>2 (hMP) |
| Rf (99mTc-NG &99mTcO4-) |
0.92 ± 0.09 |
3.23 ± 0.24 (lMP) |
Table 1: Chromatographic conditions and results of the 99mTc-NephroMAG™ quality control with the two r-TLC methods. Rf: resolution factor; hMP: hydrophilic mobile phase; lMP, lipophilic mobile phase.
Discussion/Conclusion
At the opposite of 99mTc-MAG3™, another radiopharmaceutical used for dynamic renal scintigraphy, where analytical methods for determining radiochemical purity were published [3]; to our knowledge it is the first study that reported a r-TLC quality control method of radiochemical purity of 99mTc- NephroMAG™. Technescan MAG3™ is composed of betiatide and its preparation is more time-consuming than NephroMAG™ due to the radiolabeling requires heating. Concerning the determination of radiochemical purity of 99mTc-MAG3™ both techniques, with modified solid phase extraction cartridge procedure and instant thin-layer chromatography method has been developed [3]. The quality control of the radiochemical purity of 99mTc-NephroMAG™ via r-TLC with both mobile phase, hydrophilic (sodium citrate) and lipophilic (EA/CH3CN (60:40, v/v)) thanks to Tec-Control™ strip, is an interesting alternative to the method on Whatman™ 3 mm chromatographic paper. On the one hand, this new analytical method offers a better resolving power for both impurities searched. On the other hand, these chromatographic parameters allow reducing quality control time by half, making it easier to realize a discharge control of 99mTc-NephroMAG™.
References
- Sfakianakis GN, Sfakianaki E, Georgiou M, Serafini A, Ezuddin S, et al. (2009) A renal protocol for all ages and all indications: mercapto-acetyl-triglycine (MAG3) with simultaneous injection of furosemide (MAG3-F0): a 17-year experience. SeminNucl Med 39:156–173.
- Taylor AT, Folks RD, Rahman AKMF, Polsani A, Dubovsky EV, et al. (2017) (99m)Tc-MAG3: Image Wisely. Radiology 15: 2311.
- Seetharaman S, Sosabowski M, Ballinger J (2007) Validation of alternative methods of preparing 99mTc-MAG3. Applied Radiation and Isotopes 65:1240–1243.