Research Article - (2016) Volume 24, Issue 3
Marwa Ismail Abdelgawad*
M.D, Assiut University Faculty of Medicine, Assiut, Egypt
Ebtessam Elgezawy
Assistant Professor, Assiut University Faculty of Medicine, Assiut, Egypt
Samy Elgizawy
Assistant Professor, Assiut University Faculty of Medicine, Assiut, Egypt
Mohamed Zaineldeen
Assistant Professor, Assiut University Faculty of Medicine, Assiut, Egypt
Hanan Sharaf El-deen Mohammed
Lecturer, Assiut University Faculty of Medicine, Assiut, Egypt
Corresponding Author:
Marwa Ismail Abdelgawad
MD, Lecturer, Assiut University Faculty of Medicine, Clinical oncology, Assiut, 002088, Egypt
Tel: +201201777338
Fax: +20882333367
E-mail: esmailmarwa@yahoo.com
Submitted date: November 30, 2015; Accepted date: June 22, 2016; Published date: June 29, 2016
Lymphomas are heterogenous group of lymphoid disorders that have in common clonal expansion of malignant lymphocytes. Angiogenesis is important in the proliferation of hematopoietic neoplasms. Vascular endothelial growth factor (VEGF) plays an important role in the initiation of angiogenesis. Moreover, Lymphoma progression may be enhanced by angiogenesis. Physiologically, angiogenesis helps in regulation of reproduction and wound healing, but the unregulated angiogenesis may result in tumor growth as the growing tumor needs an extensive network of capillaries to provide nutrients and oxygen. Angiogenesis is a prerequisite for growth of tumors whether solid or liquid tumors. Aim of the work: In the current study we determine the serum levels of some known angiogenic stimulators as VEGF, copper and nitric oxide and serum level of zinc as one of the angiogenic inhibitors and determine copper/zinc ratio in newly diagnosed and relapsed patients with NHL to study the significance of the previous factors in relation to disease progression and staging. Methods: Serum levels of VEGF, Nitric oxide, Copper, Zinc and Copper/zinc ratio were determined in 114 patients of NHL classified into four groups according to the disease stage investigated before the start of chemotherapy and 40 healthy controls. Results: Patients in 1st group (stage I and II) showed significant elevation in serum levels of VEGF, copper and insignificant changes occurred in serum levels of zinc, nitric oxide and in copper/zinc ratio in comparison with the control group. On the other hand; patients in 2nd group (stage III) and 3rd group (stage IV) showed highly significant increase in serum levels of copper and copper/zinc ratio; while insignificant changes occurred in serum levels of VEGF and nitric oxide. Conversely, highly significant decrease occurred in serum levels of zinc in 2nd group only. Patients in the 4th group (relapsed cases) showed highly significant increase in serum levels of copper, significant increase in serum levels of VEGF and in copper/zinc ratio, while insignificant changes occurred in serum levels of zinc, and nitric oxide. The comparison between different patients groups revealed no significant differences in all special investigations except for zinc where there was a significant lower level of zinc in 2nd group than 1st group, and for copper and copper/zinc ratio; there were significant rise of each in 4thgroup in comparison to 1st group. Conclusion: Serum VEGF and copper levels play an important role in early detection of NHL as it increased significantly in early stages, the highest levels were found in advanced cases together with low zinc level suggesting their role in follow up of NHL together with consideration of copper/zinc ratio while limited role of nitric oxide had been observed.
Keywords
NHL; VEGF; Copper; Zinc; Nitric oxide
Introduction
Non-Hodgkin's lymphomas (NHLs) form a heterogeneous group of lymphoproliferative neoplasms with different presenting features, clinical course and response to treatment. NHLs constiute approximately 3% of all malignancies. According to geographic location, the incidence of lymphomas varies from 2 to 18 cases per 100.000 people per year. The course of disease depends on various biological and clinical parameters as well as therapeutic decisions at the beginning of treatment.1
Angiogenesis is defined as a new blood vessel sprouting from pre-existing vessels.2 Angiogenesis is critical in both physiological development and pathological processes such as tumor progression, wound healing, cardiovascular, inflammatory, ischemic and infectious diseases.3 Angiogenesis is regulated by a balance of various positive (e.g. VEGF) and negative regulatory factors (e.g. Zinc). Increased levels of positive regulatory molecules have been correlated with poor prognosis in patients with solid tumors.4
While many aspects of postnatal pathological angiogenesis have been extensively studied in the context of non-hematopoietic neoplasms, the precise role of these processes in lymphoma pathogenesis is under active investigation.5
VEGF is a key tumor-derived angiogenic factor that exerts multiple functions including stimulation of angiogenesis, vasculogenesis, inflammation and vascular permeability.6 The VEGF family of growth factors and its receptors constitute the most important signaling pathways in tumor angiogenesis.7 VEGF promotes tumor angiogenesis through several mechanisms including; enhanced endothelial cell proliferation and survival, increased migration and invasion of endothelial cells, increased permeability of existing vessels forming a lattice network for endothelial cell migration and enhanced chemotaxis and homing of bone marrow derived vascular precursor cells.8 In addition to having proangiogenic effects, VEGF has several important functions that are independent of vascular processes including autocrine effects on tumor cell function (survival, migration, invasion), immune suppression and homing of bone marrow progenitors to prepare an organ for subsequent metastasis.9,10
Copper is one of the trace elements found in the serum in a concentration of 70-140 μg/dl in adult men and 80-155 μg/dl in women. It acts as an obligatory cofactor and is permissive to the angiogenic activator. Copper is an integral component of many metallo-enzymes which involved in oxidation–reduction reactions including; ceruloplasmin, cytochrome C oxidase, superoxide dismutase, etc.11
Moreover; VEGF expression is sensitive to copper and that the angiogenic potential of copper may be harnessed to accelerate dermal wound contraction and closure. At physiologically relevant concentrations, copper sulfate induced VEGF expression in primary as well as transformed human keratinocytes. Copper shared some of the pathways utilized by hypoxia to regulate VEGF expression. Topical copper sulfate accelerated closure of excisional murine dermal wound allowed to heal by secondary intention. Copper-sensitive pathways regulate key mediators of wound healing such as angiogenesis and extracellular matrix remodeling. Copper-based therapeutics represents a feasible approach to promote dermal wound healing.12
Serum copper level can be used as prognostic indicator for monitoring disease activity and response to treatment in malignant lymphoma. They also can be used as predictors of successful treatment in pediatric patients with lymphoma.13
Zinc intracellular concentration is correlated to cell proliferation, differentiation or apoptosis. Red blood cells contain 6 to 8 times the amount of zinc as blood plasma, which is around 100 μg/dl. White blood cells contain up to 25 times the amount of zinc in the serum. Mast cells and basophils contain extremely high amounts of zinc, being found in granules with histamine.14
Copper/zinc ratio has a diagnostic and prognostic value in solid tumors as well as hematological tumors. Copper/Zinc ratio is higher in patients with lymphoma or acute and chronic leukemias compared to gender and age–matched control subjects. Both the serum copper level and copper/zinc ratio were correlated to histopathological changes, clinical stage, and prognosis of HD.15
Nitric oxide plays a central role in angiogenesis as a mediator of signaling by VEGF and other angiogenic factors as well as acting as a downstream effector of VEGF action. There is evidence that NO is involved in both tumor angiogenesis and in immune cell killing of tumor cells and, therefore, can have positive and negative effects on tumor progression. At a relatively low concentration, NO can protect tumor cells from apoptosis, stimulate tumor angiogenesis and increase tumor blood flow thus promoting tumor growth. At higher levels, it can induce apoptosis and arrest tumor growth.16
Aim of the Work
In the current study we determine the serum levels of some known angiogenic stimulators as VEGF, copper and nitric oxide and serum level of zinc as one of the angiogenic inhibitors and determine copper/zinc ratio in newly diagnosed and relapsed patients with NHL to study the significance of the previous factors in relation to disease progression and staging.
Materials and Methods
The present study was performed on 114 patients with NHL attending both of South Egypt Cancer institute and Internal Medicine Department in Assiut University hospital in the period of June 2011 to December 2014 and 40 apparently healthy controls.
–– Careful clinical history including staging of lymphoma using Ann Arbor staging system according to Carbone et al. was done in patient group.17
–– Investigations including; [Complete blood picture, Liver function tests, Renal function tests, Uric acid and Lactate dehydrogenase (LDH)] were carried out on all patients and controls.
–– Investigations were done in the form of: Chest X–ray, abdominal ultrasound, Computerized tomography (CT), Bone marrow aspiration and/or biopsy were done for all cases, lymph node biopsy or splenic aspirate if needed in patient group.
–– Special investigations were done included assay of serum level of: VEGF [by enzyme linked immunosorbent assay (ELISA) technique using ChemiKine sandwich (Chemicon VEGF- ELISA kit–UK], Copper and Zinc [by Colorimetric technique using (Spinreact kits- Spain) Prime semi automated colorimeter], and Nitric oxide [by colorimetric technique micro method using Stat Fax ELISA reader] in patients and controls.
Statistics
This study is a case control study. Data was entered and analyzed using SPSS version 13. Numeric data was expressed as mean and standard deviation (SD). Statistical significance was calculated using t-test for numeric data. P value less than 0.05 was considered significant while p value less than 0.001 was considered highly significant. Categorical variables were expressed as absolute numbers and percentages.
Results
This study comprised (114) patients with NHL [88 (77%) were newly diagnosed cases and 26 (23%) were relapsed cases (after 6 months interval of complete remission)] classified into four groups: group 1 consisted of 18 (16%) patients in stage I or II, group 2 consisted of 34 (30%) patients in stage III, group 3 consisted of 36 (31%) patients in stage IV and 26 (23%) relapsed cases as group 4 (Table 1).
| Patients | Controls | |
|---|---|---|
| Total number | 114 | 40 |
| New n (%) | 88 (77) | |
| Relapsed n (%) | 26 (23) | |
| Age range ( yrs) | 18-78 | 20-45 |
| Mean + SD | 45.7+18.7 | 32.3 + 8.4 |
| Sex n (%) | ||
| * Males | 94 (82) | 24 (60) |
| * Females | 20 (18) | 16 (40) |
| Newly diagnosed n (%) | 88 (77) | |
| * Stage I or stage II | 18 (16) | |
| * Stage III | 34 (30) | |
| * Stage IV | 36 (31) | |
| Relapsed cases n (%) | 26 (23) |
*NHL (Non-Hodgkin’s Lymphoma), Stage of malignancy (I-IV) (Ann Arbor staging), SD=Standard Deviation
Table 1: Data of NHL patients and controls.
Assay of serum level of VEGF
Only group 1 and 4 showed significant elevation in VEGF in comparison to control group (p<0.05) (Table 2 and Figure 1). Comparison between the different groups of patients showed no significant differences between them as regard VEGF level.
| Group1 | Group 2 | Group 3 | Group 4 | Controls | |
|---|---|---|---|---|---|
| (n=18) | (n=34) | (n=36) | (n=26) | (n=40) | |
| VEGF (ng/L) Mean ± SD | 420.5 ± 144.8* | 357.4 ± 187.2 | 379.6 ± 201.7 | 389.5 ± 204.6* | 294.2 ± 95 |
| Copper (μg/dl) Mean ± SD | 141.8 ± 29.5* | 145.2 ± 48.8** | 146.4 ± 41.1** | 179 ± 49** | 97.5 ± 17.2 |
| Zinc (μg/dl) Mean ± SD | 85.7 ± 32.4 | 67 ± 32.6** | 95.8 ± 59.5 | 96 ± 47.3 | 107.2 ± 39.2 |
| Copper/Zinc Mean ± SD | 1.8 ± 0.6* | 2.8 ± 1.9** | 1.8 ± 0.8** | 2.9 ± 2.6* | 1 ± 0.5 |
| NO (μM/L) Mean ± SD | 21.1 ± 5.1 | 22.6 ± 5.4 | 19.4 ± 6.8 | 20.6 ± 9.6 | 21.2 ± 6.5 |
NHL (Non Hodgkin's Lymphoma), VEGF (Vascular Endothelial Growth Factor) * Significant, (p value less than 0.05) ** highly Significant, (p value less than 0.001) SD=Standard Deviation
Table 2: Serum levels of VEGF, Copper, Zinc, Copper/Zinc and Nitric oxide in NHL patients and controls.
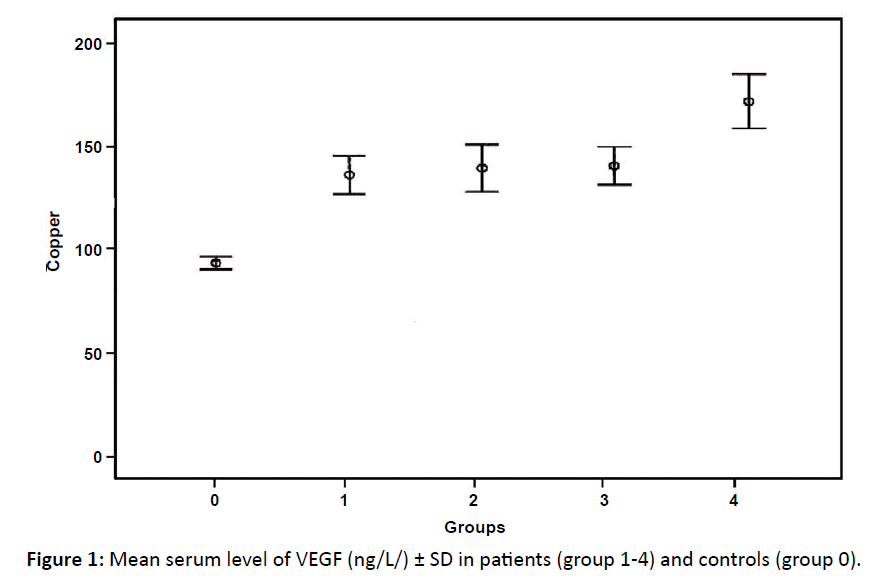
Figure 1: Mean serum level of VEGF (ng/L/) ± SD in patients (group 1-4) and controls (group 0).
Assay of serum level of copper
Serum copper showed significant elevation in group 1 and highly significant elevation in groups (2, 3 and 4) of patients in comparison with control group with maximal elevations observed in group 4 (p<0.001) (Table 2). Figure 2 displays a comparison between the different groups of patients regarding serum copper where there was only a significant difference between group1 and 4 in the serum copper level.
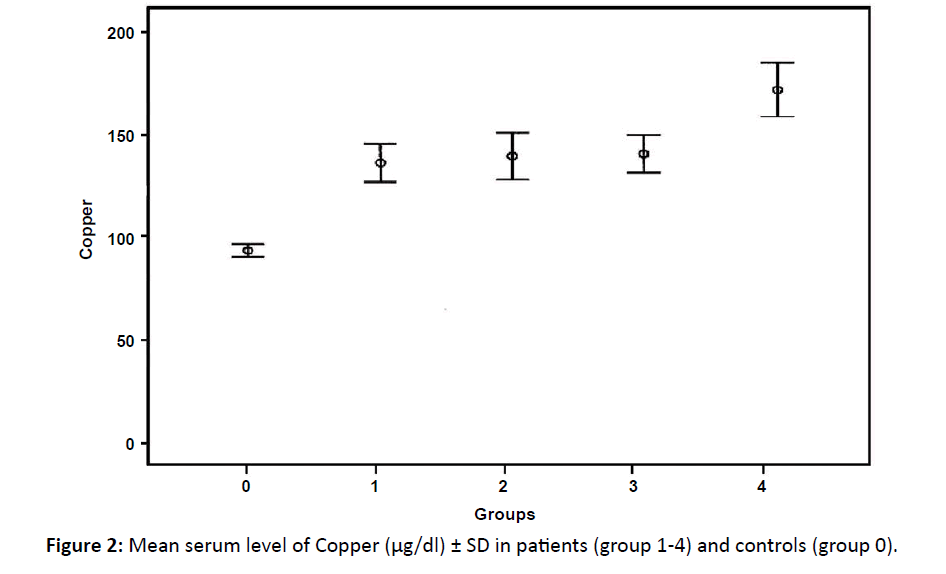
Figure 2: Mean serum level of Copper (μg/dl) ± SD in patients (group 1-4) and controls (group 0).
Assay of the serum level of zinc
Serum zinc showed highly significant decrease only in group 2 compared to control group (p<0.001) while no significant difference between each of 1st, 3rd and 4th groups with the control group (Table 2 and Figure 3). Comparison between the different groups of patients showed only a significant difference between group1 and 2 in serum zinc level.
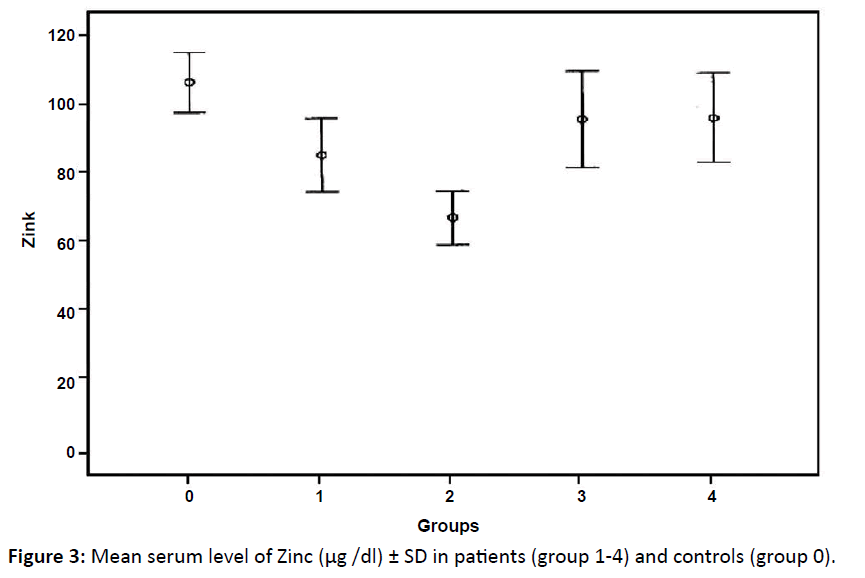
Figure 3: Mean serum level of Zinc (μg /dl) ± SD in patients (group 1-4) and controls (group 0).
Assay of the copper/zinc ratio
There was highly significant elevation of copper/zinc ratio in all groups compared to controls with (p<0.001) in group 2 and 3 and (p<0.05) in group 1 and 4 compared to control group (Table 2 and Figure 4).
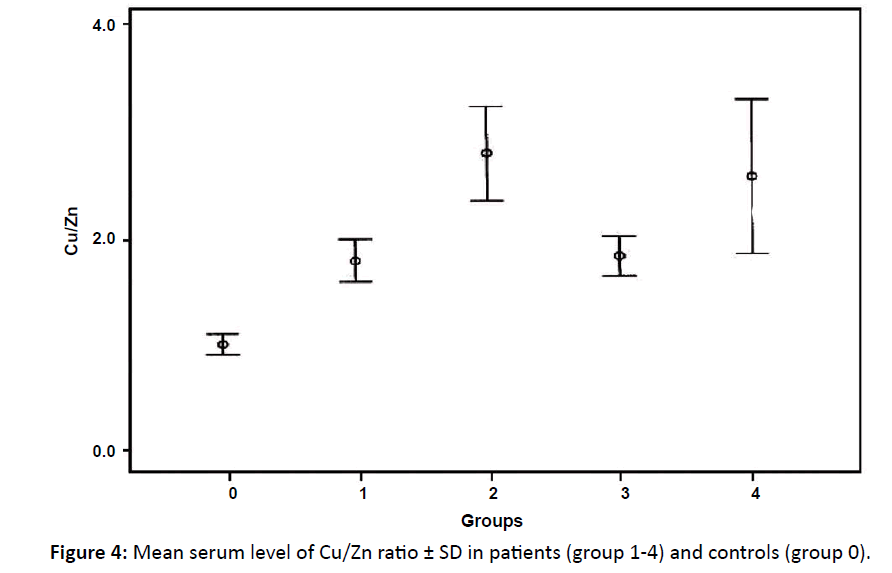
Figure 4: Mean serum level of Cu/Zn ratio ± SD in patients (group 1-4) and controls (group 0).
Comparison between the different groups of patients showed a significant difference between group1 and group 3 with group 4 in the copper zinc ratio.
Serum level of nitric oxide
NO showed no significant difference between control group and patients' groups (Table 2 and Figure 5) and between the different groups of patients regarding No level.
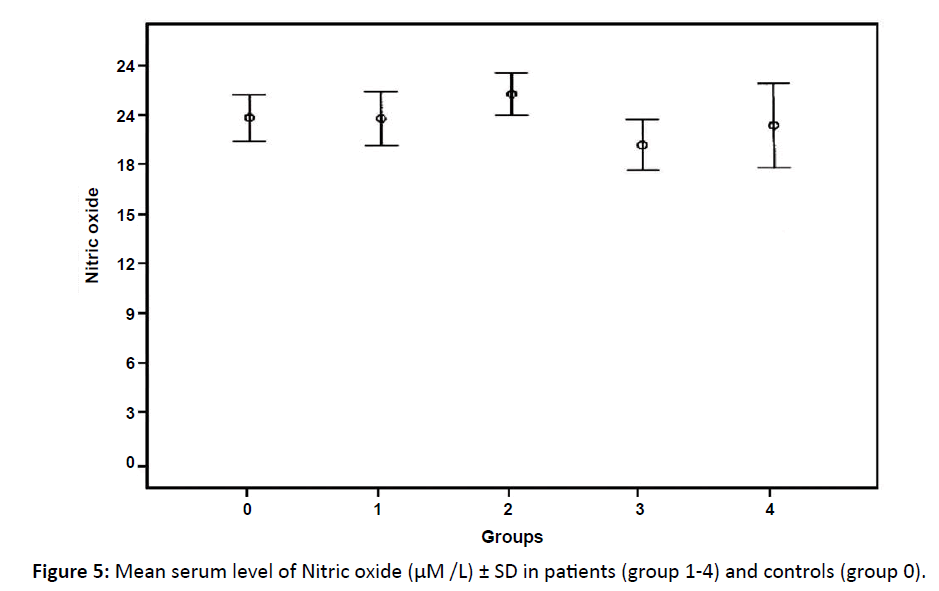
Figure 5: Mean serum level of Nitric oxide (μM /L) ± SD in patients (group 1-4) and controls (group 0).
Discussion
Angiogenesis is essential for lymphoma growth, progression and metastasis which is stimulated by many proangiogenic factors as basic Fibroblast Growth Factor (FGF), Vascular Endothelial Growth Factor (VEGF) and Platelet Derived Growth Factor (PDGF). There is emerging evidence suggesting that tumour progression of haematological malignancies depends on the induction of new blood vessel formation.18 Using different markers to assess blood vessel maturity showed a higher number of immature blood vessels in diffuse large B-cell lymphoma than in reactive nodes or follicular lymphoma.19
Growing evidence has demonstrated that control and modulation of angiogenic activity is important for the development, repair and growth of normal and abnormal tissues including cancer tissues. Antiangiogenic therapy could therefore play an important role in the clinical treatment of number of diseases including cancer, arthritis, psoriasis and diabetic retinopathy.20
VEGF plays a vital role in the progression of Non-Hodgkin’s lymphoma (NHL) and there is a significant correlation between VEGF overexpression and overall survival in NHL.21
In this study, the mean level of VEGF in total patients' group was higher than normal controls this matched with who concluded that in non-Hodgkin lymphoma the main mechanism of angiogenesis seems to be dependent on the VEGF pathway and its expression, particularly by stromal cell.22 Also, Wróbel et al. found that VEGF serum level was significantly higher in NHL group than in the control. This observation may have prognostic and clinical significance and provides rationale for use antiangiogenic agents in NHL therapy. 23
Elbaz et al. revealed that the levels of the basic FGF and VEGF in NHL patients were significantly increased in pre-treatment patients compared to controls. Also, they found that the levels of growth factors in whole NHL patients before treatment were significantly higher than controls and significantly decreased in total cases under treatment.24
Minoia et al. showed increased angiogenic activity and complexity of the vascular network, with larger, flattened and branched vessels, in indolent NHL at recurrence compared to diagnosis, supporting the role of angiogenesis in disease progression.25 Moreover, Wrobel et al. documented the association between the polymorphic features located within the VEGF and bFGF genes in NHL patients with prognosis and progression of the disease.1
In this study, significant elevation in VEGF was found in group 1 (stage I and II) and group 4 (relapsed cases) but insignificant changes occurred in group 2 and 3 in comparison with the control group.
Serum VEGF levels in this study do not grade the disease stage as they were higher in stage I and II than both III and IV. This agreed with Xia et al. who concluded that high serum level of VEGF was not found associated with stage of lymphoma.26
VEGF is less expressed in stage III and IV in NHL in contrast to non hematological malignancies, the explanation of that is due to the differences in angiogenesis pathways and in the characteristics of blood vessels between lymphomas of various histologic subtypes and non hematological malignancies. Several authors reported that blood vessels in lymph nodes in follicular lymphoma are predominantly in the inter follicular areas with relative low vascularity in the neoplastic follicles.27,28 This pattern of vascular distribution is similar to that found in reactive lymph nodes and is lost in aggressive lymphoma. The second reason is the phenotype of blood vessels in different subtypes of lymphoma seems to vary.29
In this study, we found significant elevation in all groups of patients regarding serum copper but maximal elevation was found in 4th group (relapsed cases), also the increase in serum copper is correlated with the disease stage, higher level was observed among patients of the 4th group. These results agreed with the results of Gozdazoglu et al. who performed their study on patients of untreated pediatric NHL where the mean copper level was significantly higher than controls.30 Increased angiogenic activity shows worst prognosis due to invasion of tumor cells to lymph nodes.31 Many researchers studied the copper level in patients with untreated malignant lymphoma; they found that serum copper level was significantly higher before treatment and patients in stage III and IV had higher serum copper level.32-34
Copper ions stimulate proliferation and migration of endothelial cells. It has been shown that serum copper concentration increases as the cancer progresses and correlates with tumor incidence and burden. Copper ions also activate several proangiogenic factors as VGEF, tumor necrosis factor (TNF)-α and interleukin (IL)-1. It also enhances binding of angiogenin to endothelial cells.35
In this study, there was a decrease in the serum zinc levels in patients groups compared to controls but this was highly significant only in stage III (group2) and this agreed with previous works;33 Rosas et al. and Han et al. who concluded that serum zinc level was lower in the HD patients when compared to the healthy individuals15,34 while Wu, found no significant difference between patients and controls regarding serum zinc level in NHL.32 Zinc helps block the absorption of copper and acts to remove accumulated copper from the body as well as prevent its accumulation.34
In this study, the mean copper /zinc ratio showed highly significant elevations in group 2 and 3 and significant elevation in group 1 and group 4 (compared to controls). This result agreed with Gupta et al. and Rosas et al. found significant higher levels of copper/zinc ratio in patients than controls.33,34 On the other hand; Wu, noticed that copper/zinc ratio in patients in stage III and IV was higher than that measured in patients in stage I and II.32 Moreover, Han et al. concluded that both serum copper level and the copper/zinc ratio were strongly correlated to histopathological changes, clinical stage, and prognosis of Hodgkin’s disease.15
Numerous studies since the 1970s established excess copper and high copper/zinc ratio as prognostic indicators for lymphoma where higher levels correspond to more aggressive disease as the most aggressive tumors are the highly vascular but now it is known that the elevation of serum copper in patients is due to tumor growth and associated angiogenesis which needs copper as an important element in stimulation of endothelial growth, proliferation and activation of other angiogenic factor.36 The increase of serum copper in late stages is due to increased tumor burden.
Nitric oxide (NO) mediates a diverse array of biological activities, including vasodilatation, neurotransmission and immune defense. Insignificant differences between controls and patients in serum NO levels were found in renal cell carcinoma and even lower than normal but still insignificant in stage 3 and 4.37 This support the theory that lower concentration of NO promotes angiogenesis and hence tumor growth.38 Conversely, higher levels of NO were observed to be correlated with tumor burden in gastric cancer.39 In metastatic cancer breast, mean plasma NO level was 20 folds increased in comparison to mean control level.40
In the current study, the mean NO level was higher in patients in stage III but not significant. Also in the other groups there were no significant differences between controls and patients regarding NO. This may be due to the lower expression of NO in cases of NHL as stated by Mendes et al. where they stated that NO was positive in only 1/16 cases of NHL. So, NO levels are not suspected to be high in cases of NHL.41
Conclusion
Serum VEGF and copper levels have a diagnostic role in detection of conditions with increased angiogenesis as malignancy. Also serum copper can be used to detect progression of NHL as marked increase occurred with advanced stages. Moreover, they have a value in follow up cases of NHL. Monitoring copper/zinc ratio have prognostic role than copper only as increased copper/zinc ratio occurred in advanced stages. Conversely, decrease in serum zinc level has bad prognosis.
Nitric oxide level seems to have limited role in diagnosis and follow up of cases of NHL however, larger sample size together with the estimation of tissue levels in lymph nodes or bone marrow are still required.
So, this angiogenic profile might be used to monitor cancer therapy or it might be a predictor of outcome after cancer has been diagnosed and even aid in the selection of the proper anti-angiogenic treatment.
Ethical Committee Approval
Assiut University-Faculty of Medicine May 2013.