Research Article - (2018) Volume 8, Issue 5
Onuoha OU1, Osuocha KU2 and Chukwu EC3*
1Department of Biochemistry, Michael Okpara University of Agriculture Umudike, Nigeria
2Department of Biochemistry, Gregory University Uturu, Abia State, Nigeria
3Department of Biochemistry, Federal University, Lafia Nasarawa State, Nigeria
Corresponding Author:
Chukwu EC
Department of Biochemistry
Federal University, Lafia Nasarawa State, Nigeria
Tel: +21656400412
E-mail: callychuksu@gmail.com
Received Date: Aug 14, 2018; Accepted Date: Sep 05, 2018; Published Date: Sep 10, 2018
Citation: Onuoha OU, Osuocha KU, Chukwu EC (2018) Phytochemical Profiling, Hypolipidemic, Haematological and Body Weight Effects of Acanthus Montanus Leaf Extracts in Male and Female Albino Rats. Eur Exp Biol Vol. 8 No. 5:30. doi:10.21767/2248-9215.100071
Copyright: © 2018 Onuoha OU, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Acanthus montanus leaves in the eastern part of Nigeria has been used in treatment diseases. This work was designed to evaluate phytochemical constituents, hypolipidemic, hematological and body weight effects of Acanthus montanus extracts in male and female albino rats. GC-MS analysis showed eleven bioactive compounds. Ninety (90) albino rats of 45 male and 45 female rats were used for the study, divided into eighteen groups of five rats. Alphabets and figures were used to identify the male and female rats. Groups A and 1 received normal rat chow, Groups B and 2, C and 3 , D and 4, E and 5 received 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg of aqueous extract of A. montanus leaves while groups F and 6, G and 7, H and 8, I and 9 received 200 mg/kg, 400 mg/kg , 600 mg/kg and 800 mg/kg of methanol extract of A. montanus. Results showed significant (P<0.05) decrease in HDL, TG and cholesterol while HDL increased significantly (P<0.05). The hematological test showed no significant (P<0.05) difference in most test groups relative to control. The plant leaves may be helpful in managing heart related diseases.
Keywords
Phytochemical; Hypolipidemic; Haematological; Acanthus Montanus
Introduction
Plants use in traditional medicine has continued to serve as a source of medicines to mankind especially in rural areas worldwide. The medicinal potencies or therapeutic values of plants are attributed to their chemical constituents. These plantderived medicines are either used raw as in traditional herbal practices or as purified bioactive compounds [1]. Nigeria has a large number of plant species, some of which have been used for centuries in folkloric medicines to diagnose, prevent and treat various ailments [2,3]. Several medicinal plants have been screened for their therapeutic relevance and these plants constitute important sources of active natural products which differ widely in terms of structure, biological properties and mechanism of actions. Plant derived-substances have recently become of great interest due to their versatile applications and are the richest bio-resource of drugs in traditional medicine, modern medicines, food supplements, folk medicines, pharmaceutical intermediates and chemical entities for synthetic drugs [4]. In southern Nigeria, A. montanus is fondly called Agamsoso, Elele nyiju, Agamefu [5,6]. It is called Ogwu magburu nwa okuko by Southeast people while the Ijaw people call it Edule imemein. In Western Nigeria, it is called Ahon-ekun or Ikunmu-arugbo. This plant has and employed in traditional medicine. Based on its wide usage in traditional medicine, this work is aimed evaluating the actual bioactive constituents of A. montanus and its associated biochemical effects in order to ensure holistic utilization of this plant.
Methods
Fresh leaves of Acanthus montanus were obtained from Umuoda Amuzu in Aboh Mbaise L.G.A of Imo State. They were identified by Professor G. G. E. Osuagwu of the Department of Plant Science and Biotechnology, Michael Okpara University of Agriculture, Umudike. The leaves were washed with tap water and spread on the laboratory bench to dry. The dried leaves were ground to fine powder. 200 g was separately soaked in 500 ml of water and methanol respectively for 48 hours. Each extraction mixture was filtered using whatman No. 4 filter paper and concentrated by evaporation to dryness forming the residue.
Experimental design
GC-MS analysis of Acanthus montanus was performed using Perkin-Elmer GC Clarius 500 system and Gas chromatography interface to a mass spectrometer (GC-MS). Biochemical analysis involves ninety (90) albino rats consisting of 45 male and 45 female rats, aged about nine weeks and weighing between 116 g-122 g, were used for the study. The rats were purchased from the Animal house, Department of Pharmacology, University of Nigeria Nsukka. In male and female rats group A and 1 received normal rat chow and groups B and 2, C and 3 , D and 4, E and 5 received 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg of aqueous extract of A. montanus leaves while groups F and 6, G and 7, H and 8, I and 9 received 200 mg/kg, 400 mg/kg, 600 mg/kg and 800mg/kg of methanol extract of A. montanus. Alphabets were used to represent the male rats while the female rats were represented numerically. Members of each group of animals were housed in a steel cage and allowed to acclimatize to laboratory condition for one week prior to commencement of feeding experiments. All rats were allowed free access to water and feed ad libitum. The extracts were administered orally for twenty eight days and the animals were fasted overnight, anaesthesized and sacrificed. The treatment of experimental animals was in accordance to the National Institute of Health (NIH) guidelines for the care and use of laboratory animals (NIH, 1985).
Quantitative phytochemical assessment of Acanthus montanus leaves extract using gas chromatography - mass spectroscopy
GC-MS analysis of the extract was performed using Perkin- Elmer GC Clarius 500 system and Gas chromatography interface to a mass spectrometer (GC-MS). To analyze the sample the column oven temperature and Injector temperature was set at 800°C and 200°C respectively. The flow control mode was maintained in linear velocity with a split injection mode split ratio of 20. The column flow was 1.46 ml/min with a helium carrier gas of 99.9995% purity. The column oven temperature program was set as follows: The temperature was set at 80°C with 2 minutes hold time. By the rate of 10, the temperature was 300°C with 10 minutes hold time. The column at 5 mins was used with a length of 30 millimeters and diameter of 0.25 mm and its film thickness will be 0.25 μm. The ion source temperature for MS condition was 200°C and interface temperature was 240°C. Starting m/z (Mass to charge) ratio was 40 and ending with m/z ratio of 700 (40-700 m/z).
Identification of components
Interpretation of mass spectrum GC-MS was conducted using the database of National Institute of Standard and Technology (NIST). The spectra of the unknown components were compared with those of known components stored in the NIST library. The names, molecular weights and structures of the components of the test materials were ascertained.
Lipid profile assessment
Serum total cholesterol, serum triglyceride, high density lipoprotein and low density lipoprotein cholesterol was determined by the method of Fassati and Prencipe using randox reagent kit [7].
Haematological studies
The method of cynomethaernoglobn was used for determination of haemoglobin level [8]. Centrifuge method described by Cheestrough was used for the determination of Packed Cell Volume (PCV) [9]. The method was used for the determination of platelets count. The method of formol citrate solution counting was used for counting the red blood cells while Turk's solution method was used in white blood cell determination. Mean cell haemoglobin concentration was carried out automatically using the Sysmex Automated Haematology Analyser KX-21N [10]. Mean Corpuscular Haemoglobin Concentration (MCHC) was calculated from values of PCV and HbC as follows: HbC (g/dl) × 100 / PCV (%)=MCHC (g/dl) while mean cell volume was done by the method described by Oladimeji et al.
Statistical analysis
The statistical analysis of result was done using Students Package For Social Sciences (SPSS) version 20 computer software and data collected was analyzed using Analysis of Variance (ANOVA). Means were separated using One Way Analysis of Variance.
Results
At week 1 body weights of Groups B, G, H and C were significantly (P<0.05) higher than control group (Group A) as other groups showed no significant difference, while at week 2 body weights of groups B, C and G showed no significant (P<0.05) difference when compared to control group. However, there was significant (P<0.05) decrease in the body weights of other groups compared to the control group. The body weight of rats at week 3 showed significant (P<0.05) decrease in groups E, F and H as group B, C, D and G showed no significant (P<0.05) difference when compared to the control group. Week 4 showed a significant (P<0.05) increase in group B as groups C, D, E and G showed no significant difference while F, H and I showed a significant (P<0.05) decrease when compared to control group (Figures 1-3).
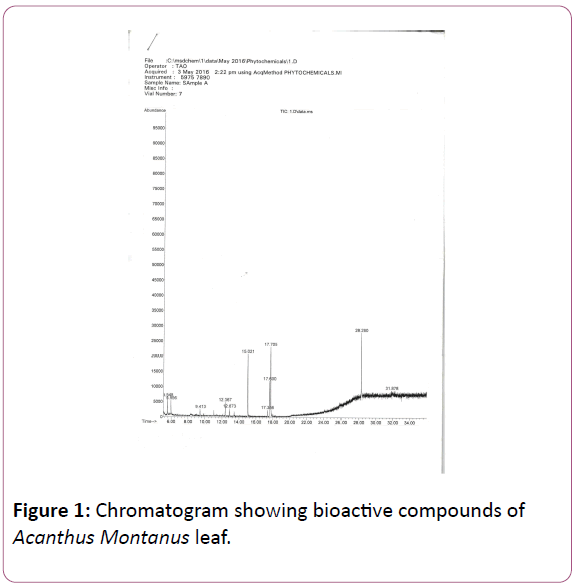
Figure 1: Chromatogram showing bioactive compounds of Acanthus Montanus leaf.
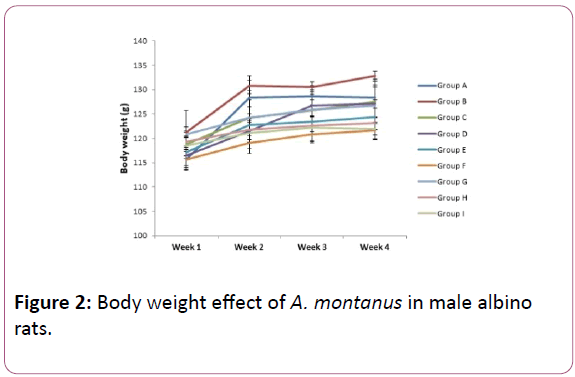
Figure 2: Body weight effect of A. montanus in male albino rats.
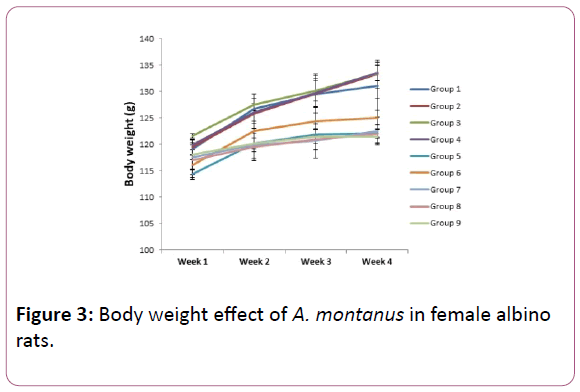
Figure 3: Body weight effect of A. montanus in female albino rats.
Week 1 showed no significant (P<0.05) difference in groups 2,3,4,7,8 and 9 as significant (P<0.05) decrease was seen in groups 5 and 6 compared to control group. In week 2 no significant difference was seen in groups 2,3 and 4 as the other groups showed significant decrease compared to the control group. Week 3 showed a significant (P<0.05) decrease in group 5,6,7,8 and 9 as groups 2, 3 and 4 showed no significant (P<0.05) difference. Groups 2,3 and 4 also showed no significant difference as groups 5,6,7,8 and 9 all shwed significant (P<0.05) decreases when compared to the control group (Table 1).
| S/No | Rt | Compound Name | Formular | Mw | Area% |
|---|---|---|---|---|---|
| 1 | 5.548 | 1,2,3-Propanetriol, monoacetate | C5H10O4 | 134 | 4.228 |
| 2 | 5.956 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl | C6H8O4 | 144 | 3.434 |
| 3 | 9.413 | 2(3H)-Benzoxazolone | C7H5NO2 | 135 | 1.93 |
| 4 | 12.37 | Bicyclo(3.3.1)nonan-9-one, 2-(1-pyrrolidinyl)- | C13H21NO | 207 | 4.177 |
| 5 | 12.87 | Heptanoic acid, 4-methoxyphenyl ester | C14H20O3 | 236 | 2.235 |
| 6 | 15.02 | n-Hexadecanoic acid | C16H32O2 | 256 | 16.448 |
| 7 | 17.36 | 1-Methylheptyl trans-2,2-dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylate | C18H32O2 | 280 | 2.234 |
| 8 | 17.6 | Tetradece-11-yn-1-ol | C14H24O | 208 | 10.675 |
| 9 | 17.71 | 9,12-Octadecadienoic acid | C18H32O2 | 280 | 27.375 |
| 10 | 28.28 | 9,12,15-Octadecatrien-1-ol, (Z,Z,Z)- | C18H32O | 264 | 25.186 |
| 11 | 31.88 | Tetracosahexane, 2,6,10,15,19,23-hexamethyl- | C30H50 | 410 | 2.077 |
Table 1: GC-MS phytochemical constituents of Acanthus Montanus leaves.
Results represent mean ± standard deviation (n=5). Different superscripts represent significant difference at 90% confidence level. Values in the same column having the same superscript are not significantly (P<0.05) different (Table 2).
| Triacylglycerol | Cholesterol | HDL | LDL | |
|---|---|---|---|---|
| Group A | 314.46 ± 1.23f | 73.90 ± 0.17gh | 25.03 ± 0.04a | 9.81 ± 0.03g |
| Group B | 295.40 ± 0.91de | 72.18 ± 2.14f | 29.60 ± 0.04b | 4.87 ± 0.11f |
| Group C | 310.38 ± 0.74f | 74.30 ± 2.14h | 31.24 ± 2.68c | 4.55 ± 0.08e |
| Group D | 293.68 ± 10.76de | 70.20 ± 0.76ef | 32.80 ± 0.17d | 4.70 ± 0.20ef |
| Group E | 278.90 ± 15.30c | 66.28 ± 0.62c | 39.11 ± 1.25f | 2.56 ± 0.19a |
| Group F | 289.67 ± 0.12d | 67.80 ± 2.26cd | 29.98 ± 0.15bc | 3.85 ± 0.15d |
| Group G | 300.09 ± 2.26e | 69.64 ± 2.56de | 34.38 ± 1.64e | 3.36 ± 0.2c |
| Group H | 210.40 ± 0.03a | 52.41 ± 0.12a | 38.34 ± 0.24f | 3.76 ± 0.08d |
| Group I | 246.14 ± 2.06b | 61.98 ± 1.32b | 39.42 ± 0.08f | 2.75 ± 0.06b |
Table 2: Lipid profile assessment of male albino rats (mg/dl).
Results represent mean ± standard deviation (n=5). Different superscripts represent significant difference at 90% confidence level. Values in the same column having the same superscript are not significantly (P<0.05) different (Table 3).
| Triacylglycerol | Cholesterol | HDL | LDL | |
|---|---|---|---|---|
| Group 1 | 300.42 ± 1.20f | 70.34 ± 0.84f | 26.67 ± 2.46ab | 8.43 ± 0.64e |
| Group 2 | 296.26 ± 2.79f | 68.49 ± 3.01ef | 28.70 ± 0.80a | 3.49 ± 1.63cd |
| Group 3 | 282.84 ± 1.96de | 69.23 ± 1.69f | 24.68 ± 0.89a | 4.21 ± 0.54d |
| Group 4 | 287.60 ± 7.64e | 66.71 ± 1.92e | 29.08 ± 1.97b | 3.39 ± 0.06bcd |
| Group 5 | 253.66 ± 1.23b | 60.18 ± 1.61bc | 32.20 ± 2.87c | 2.97 ± 0.04bc |
| Group 6 | 267.23 ± 8.53b | 62.44 ± 0.50cd | 27.23 ± 1.26ab | 3.52 ± 0.12cd |
| Group 7 | 279.42 ± 7.12d | 64.18 ± 1.44d | 31.82 ± 2.08c | 1.93 ± 0.12a |
| Group 8 | 238.92 ± 1.49a | 59.54 ± 2.79b | 39.67 ± 2.01d | 3.53 ± 0.30cd |
| Group 9 | 234.27 ± 1.51a | 57.01 ± 0.06a | 37.66 ± 1.79d | 2.60 ± 0.28ab |
Table 3: Lipid profile assessment of female albino rats (mg/dl).
Results represent mean ± standard deviation (n=5). Different superscripts represent significant difference at 90% confidence level. Values in the same column having the same superscript are not significantly (P<0.05) different (Tables 4 and 5).
| HB (g/dl) | PCV (%) | WBC (X1012/L) | Platelet (X10/L) | |
|---|---|---|---|---|
| Group A | 13.20 ± 0.78b | 39.00 ± 6.32d | 4.30 ± 0.52b | 180.00 ± 12.04e |
| Group B | 11.90 ± 0.29ab | 35.00 ± 2.55bcd | 3.90 ± 0.32ab | 160.00 ± 3.81bc |
| Group C | 10.00 ± 1.58a | 33.00 ± 3.16a | 3.80 ± 0.25a | 150.00 ± 5.39a |
| Group D | 11.30 ± 0.79ab | 34.70 ± 2.10b | 3.90 ± 0.32ab | 172.00 ± 4.00cd |
| Group E | 11.00 ± 1.58ab | 34.00 ± 1.58bc | 3.50 ± 0.25a | 164.80 ± 1.92c |
| Group F | 9.70 ± 4.53a | 37.00 ± 2.24bcd | 3.90 ± 0.16ab | 170.00 ± 3.81d |
| Group G | 10.32 ± 1.57a | 36.00 ± 4.47bcd | 3.90 ± 0.25ab | 167.80 ± 1.79d |
| Group H | 10.84 ± 0.32ab | 38.00 ± 2.55cd | 3.50 ± 0.16a | 155.00 ± 3.54ab |
| Group I | 11.00 ± 2.24ab | 33.00 ± 2.24a | 3.60 ± 0.32a | 159.00 ± 3.81bc |
Table 4: (A) Haematological assessment of male albino rats.
| MCHC (g/dl) | MCH (pg) | MCV (fl) | RBC (X1012/l) | |
|---|---|---|---|---|
| Group A | 33.85 ± 1.11abcd | 29.33 ± 0.78efg | 86.67 ± 0.51e | 4.5 ± 0.19a |
| Group B | 34.00 ± 0.41abcd | 28.33 ± 0.68fgh | 83.33 ± 0.12f | 4.2 ± 0.11ab |
| Group C | 30.30 ± 0.68ef | 28.57 ± 0.34fgh | 94.29 ± 0.42d | 3.5 ± 0.38b |
| Group D | 32.56 ± 0.57d | 28.97 ± 0.34fgh | 88.97 ± 0.42d | 3.9 ± 0.38b |
| Group E | 32.35 ± 0.64cd | 33.33 ± 0.77a | 103.03 ± 0.74a | 3.4 ± 0.36b |
| Group F | 26.22 ± 0.78h | 24.25 ± 0.80i | 92.5 ± 0.63d | 4.0 ± 0.32ab |
| Group G | 28.67 ± 0.67fg | 28.67 ± 0.38fgh | 100.00 ± 3.26b | 3.6 ± 0.38ab |
| Group H | 28.53 ± 1.00g | 27.79 ± 1.11h | 97.44 ± 0.90c | 3.9 ± 0.38ab |
| Group I | 33.33 ± 0.94bcd | 31.43 ± 0.75ed | 94.29 ± 1.61d | 3.5 ± 0.22b |
Table 5: (B) Haematological assessment of male albino rats.
Results represent mean ± standard deviation (n=5). Different superscripts represent significant difference at 90% confidence level. Values in the same column having the same superscript are not significantly (P<0.05) different (Tables 6 and 7).
| HB (g/dl) | PCV (%) | WBC (X1012/L) | Platelet (X10/L) | |
|---|---|---|---|---|
| Group 1 | 11.50 ± 0.05bc | 38.00 ± 2.23d | 3.80 ± 0.16d | 151.00 ± 1.58d |
| Group 2 | 11.20 ± 0.09abc | 31.80 ± 1.79ab | 3.50 ± 0.16abc | 145.00 ± 1.58b |
| Group 3 | 10.60 ± 0.06a | 37.00 ± 4.30d | 3.70 ± 0.16cd | 150.00 ± 1.58cd |
| Group 4 | 11.84 ± 0.25c | 35.00 ± 1.58bcd | 3.60 ± 0.16bcd | 165.80 ± 0.84e |
| Group 5 | 11.00 ± 1.58ab | 32.20 ± 3.49abc | 3.40 ± 0.16ab | 138.00 ± 1.58a |
| Group 6 | 10.90 ± 0.35ab | 35.80 ± 3.83cd | 3.70 ± 0.16cd | 148.00 ± 1.58c |
| Group 7 | 10.50 ± 0.50a | 35.00 ± 2.55bcd | 3.30 ± 0.16a | 190.00 ± 3.16h |
| Group 8 | 11.10 ± 0.02abc | 36.00 ± 2.24cd | 3.50 ± 0.16abc | 170.00 ± 1.58f |
| Group 9 | 10.68 ± 0.08ab | 31.00 ± 0.25a | 3.36 ± 0.17a | 180.00 ± 2.24g |
Table 6: (A) Haematological assessment of female albino rats.
| MCHC (g/dl) | MCH (pg) | MCV (fl) | RBC (X1012/l) | |
|---|---|---|---|---|
| Group 1 | 30.26 ± 0.84ef | 30.26 ± 1.17e | 99.99 ± 1.89b | 3.8 ± 0.22ab |
| Group 2 | 35.22 ± 0.52a | 28.72 ± 0.80fgh | 81.54 ± 0.99f | 3.8 ± 0.32ab |
| Group 3 | 28.65 ± 1.00fg | 27.89 ± 0.99gh | 97.37 ± 1.35c | 3.9 ± 0.57ab |
| Group 4 | 33.83 ± 0.72abcd | 32.89 ± 1.18ab | 97.22 ± 1.13c | 3.6 ± 3.16ab |
| Group 5 | 34.16 ± 0.69abc | 32.35 ± 1.31abc | 94.71 ± 1.49d | 3.8 ± 1.22b |
| Group 6 | 30.45 ± 0.65e | 28.68 ± 0.84fgh | 94.21 ± 2.29d | 3.8 ± 0.86ab |
| Group 7 | 30.00 ± 2.84efg | 29.17 ± 0.94ef | 97.22 ± 1.2c | 3.6 ± 0.95ab |
| Group 8 | 30.83 ± 1.63e | 31.71 ± 0.93bc | 102.86 ± 1.79a | 3.5 ± 0.94b |
| Group 9 | 34.45 ± 2.48ab | 32.36 ± 1.96abc | 93.94 ± 3.18d | 3.3 ± 0.86b |
Table 7: (B) Hematological assessment of female albino rats.
Discussion
GC-MS is a powerful technique used for many applications which has very high sensitivity and specificity. The phytocomponents with their respective Retention Time (RT), molecular formula, Molecular Weight (MW) and relative percentages peak area (%) are presented in (Table 2). It showed the presence of 11 bioactive compounds, namely; [1,2,3- Propanetriol, monoacetate], [4H-Pyran-4-one,2,3-dihydro-3,5- dihydroxy-6-methyl], [2(3H)-Benzoxazolone], [Bicyclo(3.3.1)nonan-9-one,2-(1-pyrrolidinyl)-], [Heptanoic acid, 4-methoxyphenyl ester], [n-Hexadecanoicacid], [1-Methylheptyl trans-2, 2-dimethyl-3-(2-methyl-1-propenyl) cyclopropanecarboxylate], [Tetradece-11-yn-1-ol], [9,12- Octadecadienioc acid], [9,12,15-Octadecatrien-1-ol,] and [Tetracosahexane,2,6,10,15,19,23-hexamethyl-]. This result is in contrast to report by Okenwa and Jude, who reported the presence of 9 bio-compounds on the same plant [11]. This difference may be as a result of the solvent used for extraction as they used ethanol while this study used methanol. It may also be as a result of harvesting season of the plant or other environmental factors. Karikalan and Rajangam [12], noted that 1,2,3-Propanetriol monoacetate is of glycerol origin with no known bioactive component. The compound 4H-pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl, has also been observed as one of the bioactive compounds in the plant Asparagus racemosus and shown to have anti inflammatory and antimicrobial activities [13]. Jananie et al. [14], reported that the compound is of flavonoid origin and possesses antimicrobial and anti-inflammatory effects. Benzoxazolone compounds have been shown to possess pharmacological properties which include antibacterial, anti-inflammatory, detoxification, tranquilizers and analgesic activities [15]. The tranquilizing property of this component might account for the slight doping effect observed in the rats during the study.
The compound bicyclo(3.3.1)nonan-9-one,2-(1-pyrrolidinyl) is of the alkaloid family and possesses important medicinal properties which include treatment of stomach pain [12].The bio-compound heptanoic acid, 4-methoxyphenyl ester is a saturated fatty acid with antimicrobial activity [16]. The compound n-hexadecanoic acid, was reported by Janani and Singaravadivel [13], after their study on Asparagus rasemosus to possess antioxidant, hypercholesterolemic, antiandrogenic, hemolytic 5 alpha reductase inhibitor and also used as flavor, lubricant nematicide and pesticide. Parthipan et al. [16], also reported the same activities on Pleiospermium alatum plant. Karikalan and Rajangam [14], also noted the presence of this compound in Marsilea quadrifolia plant to posses antitumor, anti-inflammatory and antioxidant activities. The compound Tetradece-11-yn-1-ol, 9, is an alcoholic compound with antimicrobial activity. Janani and Singaravadivel [13], also reported the presence of the compound 9,12-octadecadienioc acid after their studies on Asparagus rasemosus to have antiarthritic. anti-coronary, anti-inflammatory, hyper cholesterolemic, nematicide and insectifugal properties. It was also observed to be hepatoprotective and cancer preventive effects. This may account for the hypolipidemic property observed in this work.
The compound 9,12,15-octadecatrien-1-ol, have also been reported by Marimuthu et al. [17], in the study of gossypium seeds and shown to have antifungal and antibacterial properties The compound was detected in Pleiospermium alatum plant and observed to posses anti-inflammatory, hypocholesterolemic, cancer preventive, hepatoprotective, nematicide, insectifuge, antihistaminic, antieczemic, antiacne, 5-Alpha reductase inhibitor, antiandrogenic, antiarthritic, anticoronary and insectifugal activities [16]. It was also found in Coscinium fenestratum (Gaertn.) Colebr to possess similar properties [18]. The presence of this compound may account for the role of this plant in hypolipidemic activities observed in this study. Parthipan et al., reported the presence of the bio-compound tetracosahexane, 2,6,10,15,19,23-hexamethyl on Pleiospermium alatum plant to have cancer preventive properties, antimicrobial, sunscreen chemo preventive, antitumor immunostimulant, pesticide and antioxidant properties.
Results of the body weights of the experimental rats are presented in Figures 2 and 3, for male and female albino rats respectively showed that as the control group was increasing, the groups that were exposed to both extracts were also increasing for both male and female albino rats in each week as compared. Ogbuleka et al. [19], noted that increase in body weight of experimental rats is indicative that the plant has nutritional composition. Although significant (P<0.05) decrease were observed on the group that was exposed to 800 mg/kg of the aqueous and methanol extract during the four weeks of experiment, the changes may not have any biochemical implication as each group increased during the four weeks administration. This shows that the plant may not have a toxic effect on animal body weight. This is in consonance to earlier report of the plant on animal body weight [20,21].
Lipid profile assessments are used to ascertain the level of lipid in the blood. Lipids are important in cardiovascular diseases and are also useful in modifying the composition, structure, and stability of cellular membranes [22]. A decrease in some cases may lead to hyperthyroidism and certain forms of anemia [23]. The lipid profile assessment shows that triglyceride had no significant difference in the groups that received 400 mg/kg aqueous extract for male and 200 mg/kg aqueous extract for female albino rats as compared to their respective controls. 200 mg/kg aqueous extract for male, 400 mg/kg aqueous for female, 600 mg/kg, 800 mg/kg aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg methanol extracts for both male and female rats showed a significant (P<0.05) decrease when compared to their various control groups. Cholesterol showed no significant (P<0.05) difference in the group that received 400 mg/kg of the aqueous extract while the groups exposed to 200 mg/kg, 600 mg/kg, 800mg/kg aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800mg/kg methanol extract for male albino rats showed significant (P<0.05) decreases compared to the control group that was not exposed to the extracts. The female albino rats showed no significant (P<0.05) difference in the groups that were exposed to 200 mg/kg and 400mg/kg of aqueous extract while 600 mg/kg, 800mg/kg aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800mg/kg methanol extracts showed significant (P<0.05) decreases when compared to the control group. This result is in correlation with previous report in the plant leaves to possess hypolipidemic properties [24]. High density lipoprotein showed a significant (P<0.05) increase on the groups exposed to 200 mg/kg, 400 mg/kg, 600 mg/kg, 800mg/kg of aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800mg/kg methanol extracts for male albino rats compared to the control group. The female rats did not show significant (P<0.05) difference in the groups that were exposed to 200 mg/kg, 400 mg/kg, 600 mg/kg of aqueous and 200 mg/kg of methanol extracts while 800 mg/kg aqueous, 400 mg/kg, 600 mg/kg and 800mg/kg methanol extracts showed a significant (P<0.05) increase compared to control group. Chinyere et al. [22], reported that decrease in cholesterol increases the activity of HDL which is responsible for transporting cholesterol out of the blood. The Low density lipoprotein showed significant (P<0.05) decrease in the groups exposed to 200 mg/kg, 400 mg/kg, 600 mg/kg, 800mg/kg aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg methanol extracts for the male and female rats compared to their various control groups. Decrease in LDL is associated with reduced risk of coronary heart disorder. This is indicative that the plant may possess cardiovascular and hypolipidemic properties. Haematology is simply the study of blood and bloodforming tissues which help diagnose disorders associated with the blood.
The haematology result showed a significant (P<0.05) decrease in the groups exposed to 400 mg/kg aqueous, 200 mg/kg and 400 mg/kg methanol extracts while 200 mg/kg, 600 mg/kg, 800 mg/kg aqueous, 600 mg/kg and 800 mg/kg methanol extracts showed no significant (P<0.05) difference in the HB of the male rats compared to the control group. A decrease in haemoglobin concentration may be associated with anemic condition [25]. The female albino rats showed no significant (P<0.05) difference in the groups exposed to 200 mg/kg, 600 mg/kg, 800 mg/kg aqueous, 200 mg/kg, 600 mg/kg and 800 mg/kg methanol extacts while significant (P<0.05) decreases were observed in the groups exposed to 400 mg/kg aqueous and 400 mg/kg methanol extract when compared to the control group that was not exposed to the extracts. It can be deduced that both the aqueous and methanol extracts on moderate doses of 200 mg/kg may not have adverse effect on HB concentration. PCV showed no significant (P<0.05) difference in the groups exposed to 200 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 600mg/kg methanol extracts while significant (P<0.05) decreases were seen in the groups exposed to 400 mg/kg, 600 mg/kg, 800 mg/kg aqueous and 800 mg/kg methanol extracts compared to the control group for the male rats. The female rats showed no significant (P<0.05) difference in the groups exposed to 400 mg/kg, 600 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 600 mg/kg methanol extract while the groups exposed to 200 mg/kg, 800 mg/kg aqueous and 800 mg/kg methanol extracts showed significant (P<0.05) decreases when compared to the control group. These observed significant decreases may be attributed to anemic conditions.
Isaac et al. [26], noted that increase in PCV is indicative of good transport of oxygen and absorbed nutrients. WBC showed significant (P<0.05) decrease in the groups exposed to 400, 800 mg/kg aqueous, 600 mg/kg and 800 mg/kg methanol extacts while 200 mg/kg, 600mg/kg aqueous, 200 mg/kg and 400mg/kg methanol extracts showed no significant (P<0.05) difference when compared to the control for the male albino rats. The female rats showed significant (P<0.05) decrease in the groups exposed to 200 mg/kg, 800 mg/kg aqueous, 400 mg/kg, 600 mg/kg and 800 mg/kg methanol extracts while no significant difference was seen on 400 mg/kg, 600 mg/kg aqueous and 200 mg/kg methanol extracts when compared to the control group. The plant leaves having reduced the WBC of the animals exposed to it shows that its consumption exposes the animals to risk of infection [27].
Platelets showed a significant (P<0.05) decrease in all the groups exposed to both extracts for the male albino rats. This decrease may be associated to clot formation as platelate helps in blood cloting. The female rats showed a significant (P<0.05) decrease in the groups exposed to 200 mg/kg, 800 mg/kg aqueous and 200 mg/kg methanol extracts as the other groups exposed to the extracts showed significant increase but for the group exposed to 400 mg/kg aqueous extract when compared to the control group.
MCHC showed no significant (P<0.05) difference in all the groups exposed to 200 mg/kg, 800 mg/kg aqueous and 800 mg/kg methanol extracts while 400 mg/kg, 600 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 600 mg/kg methanol extracts showed significant (P<0.05) decrease compared to the control group for the male rats. The female rats showed no significant (P<0.05) difference in the groups exposed to 400 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 600 mg/kg methanol extracts while the groups that were exposed to 200 mg/kg, 600 mg/kg, 800mg/kg aqueous and 800 mg/kg methanol extracts all showed significant (P<0.05) increase compared to the control group. A decrease in MCHC may be indication of microlytic anaemia while an increase is attributed to macrolytic anemia [28].
MCH showed no significant (P<0.05) difference in the groups exposed to 200 mg/kg, 400 mg/kg, 600 mg/kg aqueous, 400 mg/kg and 800 mg/kg methanol extracts while 800mg/kg aqueous showed an increase as 200 mg/kg, and 600 mg/kg methanol extracts showed significant (P<0.05) decrease compared to the control group that was not exposed to the extracts for the male rats. The female rats showed significant (P<0.05) increase in the groups exposed to 600 mg/kg, 800 mg/kg aqueous, 600 mg/kg and 800 mg/kg methanol extracts as significant (P<0.05) decrease were seen in groups exposed to 200 mg/kg, 400 mg/kg aqueous and 200 mg/kg methanol extracts while the group exposed to 400 mg/kg methanol extract did not show significant (P<0.05) difference when compared to the control group that was not exposed to the extracts in a non dosage dependent manner. The decrease is also associated to anemia [28].
MCV showed a significant (P<0.05) decrease only in the group exposed to 200 mg/kg of aqueous extract while those exposed to 400 mg/kg, 600 mg/kg, 800 mg/kg aqueous, 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg methanol extracts showed significant (P<0.05) increase compared to the control group in a non dosage dependent manner for the male rats. The female rats showed significant (P<0.05) increase in the group exposed to 600 mg/kg methanol extract and significant (P<0.05) decrease in the groups exposed to 200 mg/kg, 400 mg/kg, 600 mg/kg and 800 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 800 mg/kg methanol extracts when compared to control group. The decrease noted here shows that the plant leaves may have microlytic anemic treats.
RBC showed significant (P<0.05) decrease in the groups exposed to 400 mg/kg, 600 mg/kg, 800 mg/kg aqueous, and 800 mg/kg methanol extracts while 200 mg/kg aqueous, 200 mg/kg, 400 mg/kg and 600 mg/kg methanol extracts did not show significant (P<0.05) difference when compared to the control group for male albino rats. A decrease in RBC is indication of a defect in the erythropoietic system [26]. This is indicative that the plant leaves may have reduced the level of oxygen that would have been carried to the tissues and carbon dioxide that was returned to the lungs [26]. The female rats showed no significant (P<0.05) difference in all the groups exposed to the extracts compared to the control group. This plant leaves may be less toxic in animal model.