Keywords
Ketoconazole; Jellies; Xanthan gum; Sodium carboxy methyl cellulose.
Introduction
Oral candidiasis is one of the
common fungal infections, affecting the oral
mucosa. These lesions are caused by the
yeast Candida albicans. Candidiasis is
defined as an infection caused by a fungi of
the genus Candida, and the term oral
candidiasis is only used when describing a
clinically visible lesion in the oral cavity.1
The conventional formulations for the local
delivery of drugs to the oral cavity are the
mouth paints, rinses, troches, creams and
suspensions.2,3 One way to improve the
efficacy in eradicating the infection is to
deliver the antifungal locally in the oral
cavity. Better stability and longer residence
time will allow more of the antifungal to
penetrate through the oral mucous layer to
act on Candida species for longer duration
of time. Therefore some researchers had
prepared and reported new formulation such
as gels, mucoadhesive tablets, pH sensitive
excipients composition mucoadhesive
microspheres, which were able to reside in
oral cavity for an extended period for more
effective candidiasis eradication.4,5The
present investigation is designed to improve
patient compliance. Advantages of the
Ketoconazole jellies as dosage forms
include increase in bioavailability, reduction
in dose size, and in gastric irritation, bypass
first pass metabolism. The present work is
aimed at preparing a formulation of
Ketoconazole jellies, for relief of oral
candidiasis.
Materials and Methods
Ketoconazole was received as a gift
sample from M/s Alkem Laboratories Pvt.
Ltd., Mumbai. Carboxy Methyl cellulose
sodium salt was obtained from Sol Fine
Chemicals Pvt. Ltd., Mumbai. Xanthan gum was obtained from Local market. Sucrose
was brought from SD Fine Chemicals,
Mumbai. Citric acid was received from
CDH Pvt. Ltd., Mumbai. All other
chemicals and solvents used are of analytical
grade and used as procured.
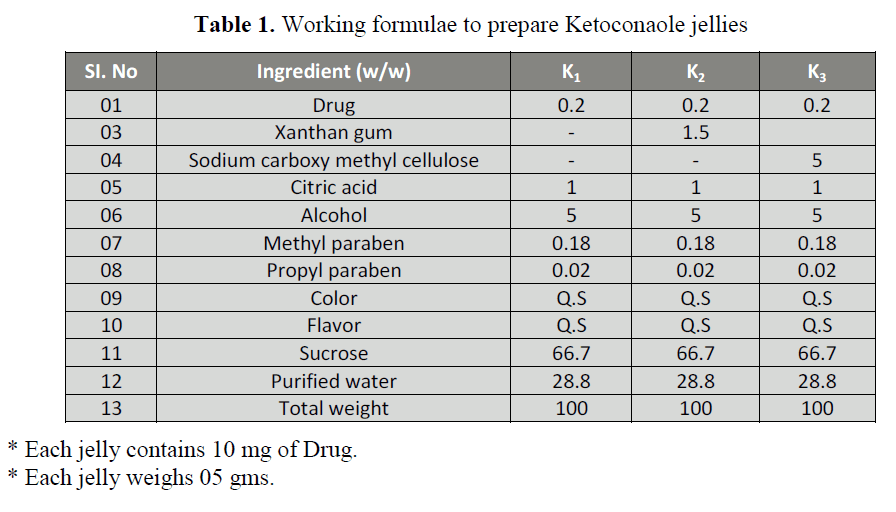
Preparation of Medicated Jellies [6]
All the formulations were prepared
using freshly boiled and cooled distilled
water as per the composition listed in Table
1. Ketoconazole jellies were prepared by
heating and congealing method. Syrupy base
was prepared in a copper vessel dissolving
the required amounts of sugar in water on
heating and stirring at 80°C for about 90
min. accurately weighed polymer powder
was dispersed in 10 mL of purified water
maintained at 90°C throughout preparation.
The dispersion was stirred using a magnetic
stirrer (2MLH, Remi Equipment Pvt. Ltd.,
Mumbai, India) for 20 min to facilitate
hydration of gelling agent. Ketoconazole
was taken in another beaker and solubilized
using alcohol. Then simple syrup was added
to it under continuous stirring. Then citric
acid and preservatives were added under
continuous stirring. Color and flavor was
added to this under continuous stirring at
60°C. The final weight was adjusted with
purified water, mixed, transferred to
polyethylene molds, sealed and allowed to
cool at room temperature (25°C ± 5°C) to
form a jelly like texture. After the jelly is set
it is wrapped in to the gelatin paper and
stored in dry place.
Table 1: Working formulae to prepare Ketoconaole jellies
Characterisation of prepared
Ketoconazole jellies
Physical observation[7]
The prepared jellies were observed
visually for clarity, odor, texture and
presence of any particles. The texture was
evaluated in terms of stickiness and
grittiness by mild rubbing the jelly between
two fingers.
Weight variation [8]
The average weight of ten jellies was
taken to determine weight variation. The
jellies were taken out of the molds in a
beaker and weighed individually, pooled and
mixed.
Determination of pH [9,10]
The pH of the formulation influences
the taste and stability of oral jellies. The pH
of prepared jellies was measured using a
digital pH meter (LI 120, Elico Ltd.,
Hyderabad, India) at room temperature
(25°C ± 5°C). For this purpose, 0.5 g of jelly
was dispersed in 50 mL of distilled water to
make a 1% solution, and the pH was noted.
Syneresis [11]
Gels experience syneresis or deswelling
due to the release of liquid,
resulting in shrinkage of gels and reduce
quality. Syneresis is the contraction of the
gel upon storage and separation of water
from the gel. It is more pronounced in the
gels, where lower concentration of gelling
agent is employed. All the jellies were
observed for signs of syneresis at room temp
(25°C ± 5°C). The formulations showing
signs of syneresis were rejected and not
considered for further studies.
Drug-excipient compatibility Studies [12]
The drug and excipients were mixed
together in 1:1 ratio and placed in
borosilicate colored glass vials. These vials were sealed and placed in an oven
maintained at 40°C and 75% RH. The
samples were observed after 15, 30 and 45
days for any color change or lump
formation. Fourier transforms infrared
(FTIR) spectra of the pure drug and its
mixtures of gelling agents were measured by
preparing dispersion in dry KBr using
attenuated total reflectance FTIR
spectrophotometer (Bruker, UK). The
absorption maxima in the spectra obtained
were compared, and the presence of
additional peaks corresponding to the
functional groups was noted.
Stability Studies [13,14]
A physically stable medicated oral
jelly should retain its viscosity, color,
clarity, taste, and odor throughout its shelflife.
The stability studies were performed at
two temperatures i.e., 37°C and 45°C over a
period of six months. Sufficient number of
samples (10) were packed in amber colored
screw capped bottles and kept in incubator
maintained at 37°C. Samples were taken at
intervals of 15 days for the drug content
estimation.
In vitrodrug dissolutionStudies [15,16]
USP XXIII Dissolution test
apparatus was used by taking 100 ml of pH
6.8 buffer in 1000 ml dissolution flask and
jelly was placed in it, rotating paddle at a
speed of 150 rpm and temperature 37±1ºC
was maintained. 5 ml aliquots were
withdrawn at 01, 02, 03, 04, 05 and 06
minutes intervals, after each withdrawal of a
sample an equal volume of dissolution
medium was added to the dissolution flask.
The filtered samples were diluted and
analyzed spectrophotometrically at 274.0
nm.
Antifungal Activity [17]
Microbiological studies were carried
out to ascertain the antifungal activity of the
prepared formulation as against the pure
drug. Ketoconazole is known to possess
superior antifungal activity against fungal
infections. In present work, antifungal
activity of Ketoconazole was tested by using
the yeast Candida albicans, which is the
most frequently encountered human fungal
pathogen being responsible for a wide range
of superficial infections. The prepared jellies
were evaluated for in-vitro antifungal
activity using standard Agar cup-plate
method.
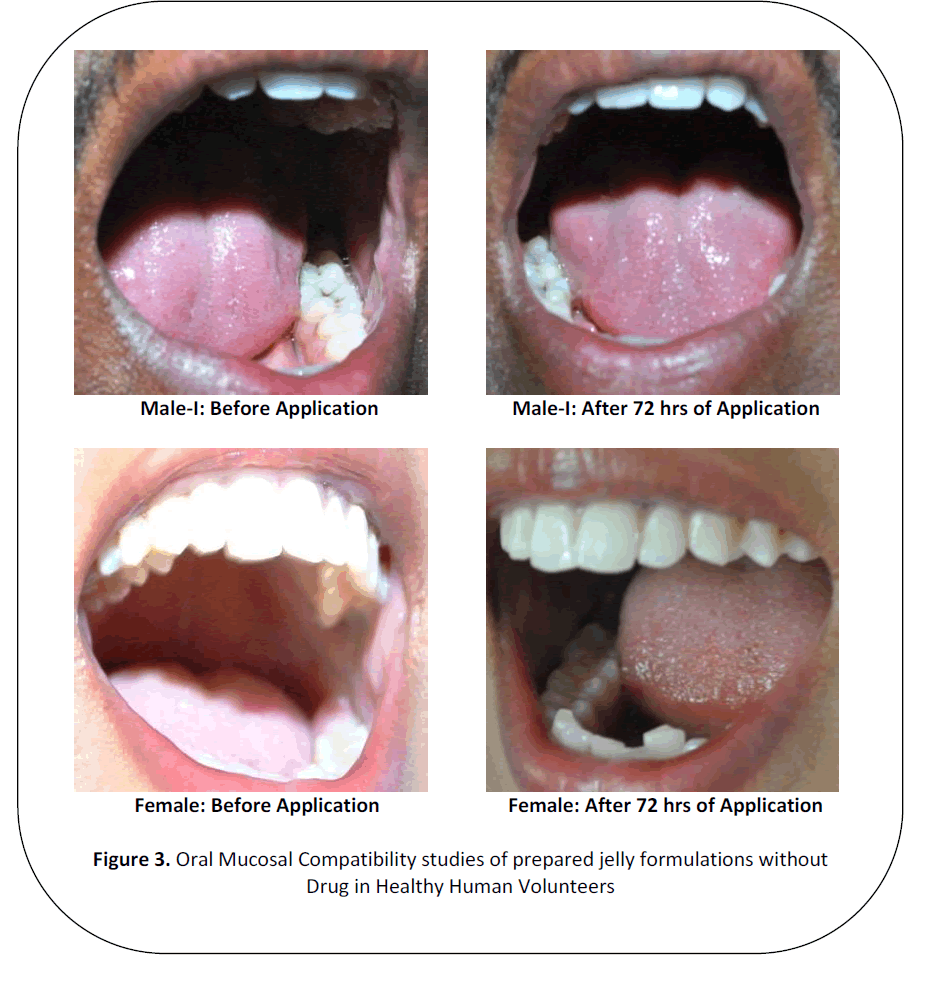
Oral Mucosal Irritancy Assessment [18,19]
Oral mucosal compatibility studies
of prepared formulations prepared without
drug were carried out in healthy human
volunteers under the supervision of qualified
physicians
By examining each volunteer's oral
cavity using focus lens to notice any
changes in tissues after the usage of
formulations. Then photographic imaging of
oral cavity of human volunteers were taken
after subsequent application for 72 hrs i.e.,
at completion of study period and these
images were compared to determine the
difference with the images taken at 0 hour of
study i.e., prior to first usage of formulation.
Mucosal irritation was also evaluated by
questioning the human volunteers at regular
intervals of time about the feeling of itching
or irritancy which appears to be highly
subjective for the study. Finally the oral
mucosal skin irritancy was evaluated for any
changes like oral erythematic, inflammation,
redness, hemorrhagic lesions or acute
painful ulcers (canker sores).
Results: characterisation ofPrepared clotrimazole jellies
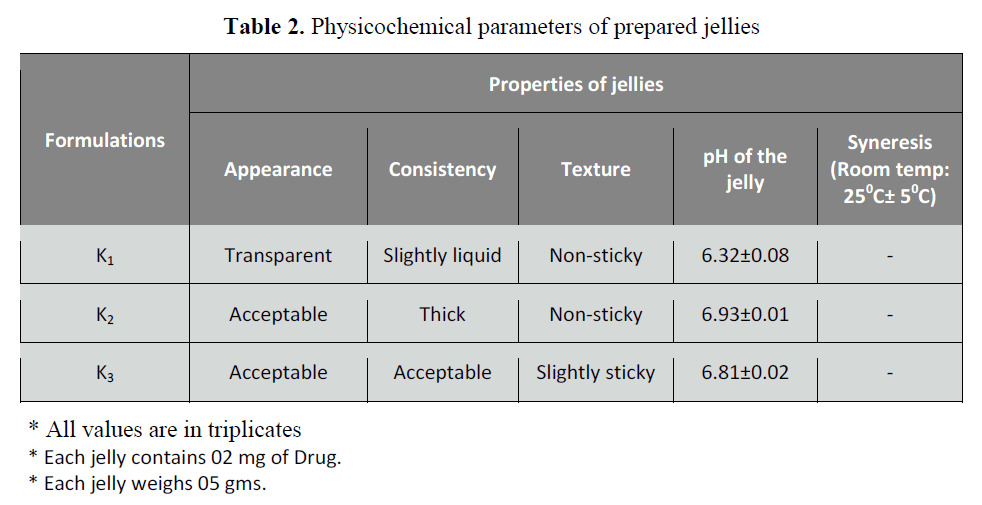
Physical observation
Physical observation of jellies is
important to justify the patient acceptance
and compliance of the products. The
observed parameters are summarized in Table 2. of all the formulations K3 showed
best results being transparent and slightly
sticky with an acceptable consistency.
Table 2: Physicochemical parameters of prepared jellies
Weight variation
The weight variation was found
between 4.95%±0.58% and 5.79%±0.83% in
all prepared jelly formulations.
Determination of pH
The results of pH of prepared jelly
formulations are summarized in Table 3.
The pH of the formulation influences the
taste and stability of oral jellies. The pH of
the prepared formulations was found in the
range of 6.32 ± 0.03-6.93 ± 0.04 which was
slightly acidic. Sucrose may precipitate in
the presence of citric acid on standing.
Therefore, a minimum quantity of citric acid
was added just to maintain the pH.
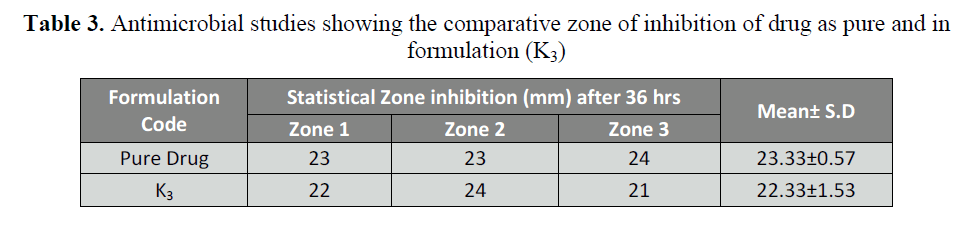
Table 3: Antimicrobial studies showing the comparative zone of inhibition of drug as pure and in
formulation (K3)
Syneresis
Syneresis was more pronounced in
the formulations, where lower concentration
of gelling agent was employed. It was
observed after 24 h of jelly preparation. The
formulations K1 showed syneresis at room
temperature (25°C ± 5°C) (Table 3).
Stability studies
A physically stable medicated oral
jelly should retain its viscosity, color,
clarity, taste, and odor throughout its shelflife.
The samples were characterized for
change in various parameters such as
appearance, pH, viscosity, sugar
crystallization, stiffness, syneresis and drug
content at the end of 90 days. A freshly made sample was used as a reference
standard for subjective evaluations.
Formulation K3 showed best results.
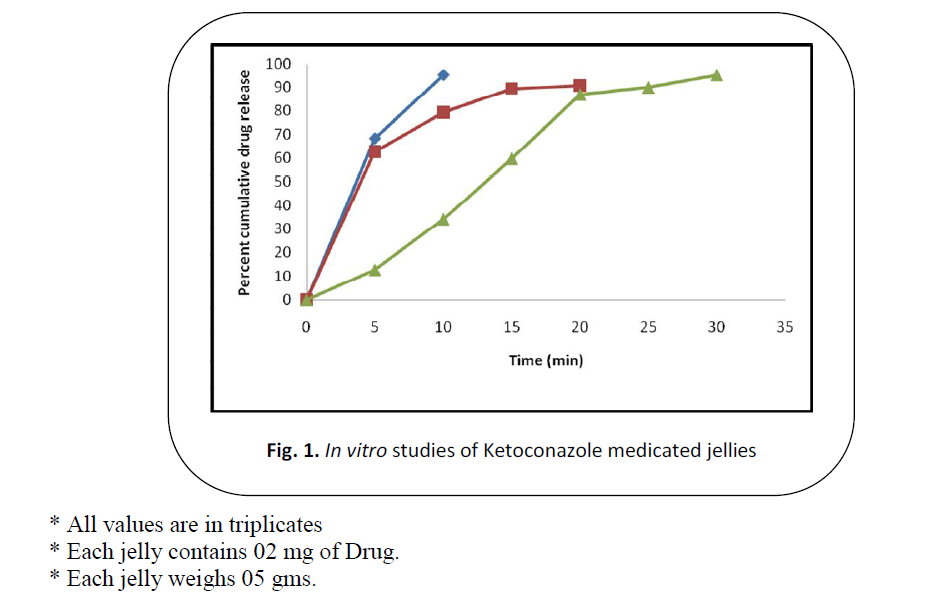
In vitro dissolution testing
The in vitro dissolution study was
carried out to compare Ketoconazole release
kinetics from the prepared jellies. The
results are summarized in Figure 1. K3 showed optimal results.
Figure 1: In vitro studies of Ketoconazole medicated jellies
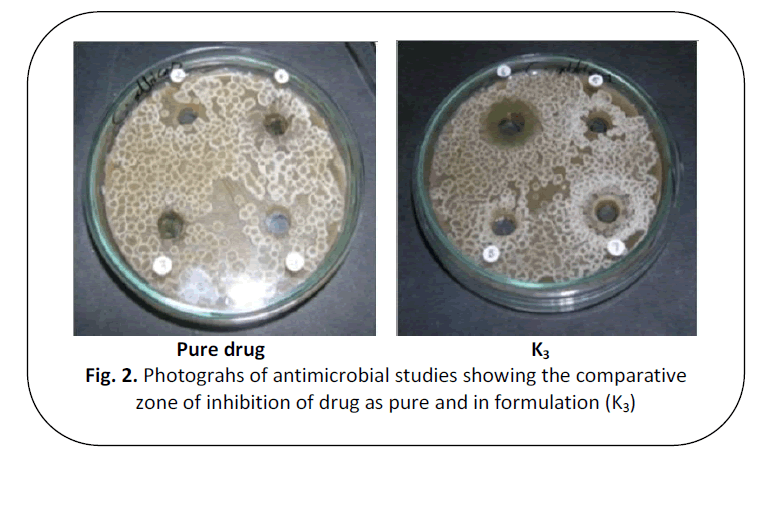
Figure 2: Photograhs of antimicrobial studies showing the comparative
zone of inhibition of drug as pure and in formulation (K3)
Figure 3: Oral Mucosal Compatibility studies of prepared jelly formulations without
Drug in Healthy Human Volunteers
Antifungal studies
The anti-microbial study reveals that
zone inhibition of various prepared
formulations was found to be equal on
comparison with the activity of pure drug.
This indicates that there is no change in the
molecular activity of the drug present in the
formulations. The results are summarized in table 4.
Table 4. Oral Mucosal Compatibility studies of prepared jellies formulations without Drug in
Healthy Human Volunteers
Oromucosal compatability tests
Results of in vivo studies in healthy
human volunteers under the supervision of
qualified team of physicians revealed that no
redness or ulcer formation or any irritation
on oral mucosa was observed. Hence, the
formulations prepared were compatible to
use as drug delivery.
Conclusion
It is found that sucrose based
Medicated jellies will be ideal dosage forms
for patients. These will have additional
advantages of patient compliance,
convenience and comfortness for efficient
treatment including low dose, immediate
onset of action, reduced dosage regimen and
economic. The Physico-chemical
characterization revealed that all the
formulations were found to be shown
acceptable weight variation, pH, viscosity,
spreadability and syneresis. The drug
content estimation showed uniform drug
content in all the formulations. IR
spectroscopic studies indicated that there were no drug-excipients interactions.
Addition of hydrophilic mucoadhesive
polymers like xanthan gum yield good
results to prolong dissolution time and the
drug release in salivary pH conditions for a
period of 30 minutes. The stability studies
proved that the prepared Medicated jellies
were found to be stable when stored at air
tight containers or twist strips. Hence the
present piece of investigation will be used
for industry, research and development
division. The anti-microbial study reveals
that zone inhibition of various prepared
formulations was found to be equal on
comparison with the activity of pure drug.
This indicates that there is no change in the
molecular activity of the drug present in the
formulations. Results of in vivo studies in
healthy human volunteers under the
supervision of qualified team of physicians
revealed that no redness or ulcer formation
or any irritation on oral mucosa was
observed. Hence, the formulations prepared
were compatible to use as drug delivery. The
present work Medicated Jellies offer patient
convenience, compliance and comfortness in
application and transportation with effective
treatment.
References
- Prasanna KR, Oral Candidiasis-A review, Scholarly J Med, 2012,2, 6-30.
- Dangi YS , Soni MS, Namdeo KP, Oral Candidiasis – A Review, Int,J,Pharm. Sci. 2010,2: 36-41
- Gallardo JM. Xerostomia: Etiology, diagnosis and treatment. Rev Med Inst Mex Seguro Soc. 2008;46:109–16.
- Chang JY, Oh YK, Kong HS, Kim EJ, Jang DD, Nam KT, et al. Prolonged antifungal effects of Clotrimazolecontaining mucoadhesive thermosensitive gels on vaginitis. J Control Release. 2002;82:39–50.
- Ning MY, Guo YZ, Pan HZ, Yu HM, Gu ZW. Preparation and evaluation of proliposomes containing clotrimazole.
- Chem Pharm Bull (Tokyo) 2005;53:620–4.
- Deborah Evangeline. D, Bhavani Shankar.R, Bharath Kumar.A, Ramesh Kumar Reddy. Formulation and Evaluation of Antimicrobial Activity of Medicated Jelly with Ajowan Extract. International Journal of Research in Pharmaceutical and Biomedical Sciences 2011; 2:691-694.
- Prakash K, Satyanarayana VM, Nagiat HT, Fathi AH, Shanta AK, Prameela AR. Formulation development and evaluation of novel oral jellies of carbamazepine using pectin, guar gum, and gellan gum. Asian J Pharm 2014;8:241-9
- Covington, A. K.; Bates, R. G.; Durst, R. (1985). Definitions of pH scales, standard reference values, measurement of pH, and related terminology. Pure Appl. Chem.1985; 57(3):531–542.
- Vishnu Vardhan Reddy Beeram (2010). Formulation, development and evaluation of cefixime oral medicated jelly. Indian Journal of Pharmaceutical Sciences. 78(2): 68-73.
- Suda N, Shinzato R, Kiyokawa M, Kaneuchi M, Sugawara M, Kohri N, et al. Development of acetylcysteine jelly for the prevention of radio contrastinduced reductions in renal function and its evaluation. Jpn J Pharm Health Care Sci 2005;31:355-9.
- Lucey JA. ADSA Foundation Scholar Award. Formation and physical properties of milk protein gels. J Dairy Sci 2002;85:281-94.
- Kasture VS and Belsare DP. Spectroscopy, 1st edition Career Publication. 2010;35-72.
- ICH Guidelines, Stability testing of active pharmaceutical ingredients and finished pharmaceutical products, WHO Technical Report Series, No. 953, Annex 2, 2009, 87-130.ICH Guidelines, Q1A-Q1F,
- www.ich.org/products/guidelines.html
- Phaechamud T, Tuntarawongsa S. Clotrimazole soft lozenges fabricated with melting and mold technique. Res J Pharm Bio Chem Sci 2010; 1(4):579-86.
- Satish gupte. Text book of medical microbiology.2002;8: 68.
- Brahmankar DM and Jaiswal Sunil B. Biopharmaceutics and Pharmacokinetics-A Treatise. In-vitro drug dissolution testing models, Vallabh Prakashan, Delhi.1995:290-293.
- Jain H.K. “Controlled and Novel drug Delivery”. Chapter 3-Oral Transmucosal Drug delivery, Devarajan P.V. and Adani M.H. CBS Publishers and distributors, New Delhi. 1st edition 1997; p59-60.
- Jain H.K. “Controlled and Novel drug Delivery”. Chapter5-Oral Transdermal Drug delivery, Mishra A.N. CBS Publishers and distributors, New Delhi. 1st edition 1997; p125Ap.
- T. Salunke, R. Mayee. Formulation and evaluation of medicated jelly of bitter drugs. IJPI 2013; 3(5): 1-14.