Keywords
Fine particle fraction; Aerodynamic behavior;
Propranolol; Ethanol; Pulmonary absorption-propranololinhalable
microparticles-absolute bioavailability
Introduction
The advantages of lung as the respiratory organ such as high
level of the blood flow rate, the permeability of the capillary,
the wide surface for absorption of drugs and the low level of
peptidase enzyme in compare with the gastrointestinal system
make it the suitable organ for delivering of drugs to the
systemic circulation [1,2]. Respiratory drug delivery has many
advantages include eliminating the hepatic first pass
metabolism effect and delivering of the impermeable
macromolecules such as amino glycosides to the blood
circulation [3]. These advantages cause to apply from this rout
as the alternative for the oral and the parenteral rout of
administration for peptide and proteins [4-8]. For this reason
the drug delivery via the respiratory system is more
considerable process in the recent decades.
There are three types of inhalable dosage forms include
nebulizer solutions, pressurized metered dose inhalers (PMDI)
and dry powder inhalers (DPI) for respiratory drug delivery
[9-11]. Among these types of dosage forms DPIs as the solid
pharmaceutical dosage forms are more stable than the others.
The formulation of the DPIs is not the complicated process
[12]. As a matter of fact in the formulation of the DPIs the
micro particles of drug as the active ingredients is surrounded
by the solid particles of the inert materials that plays the role
of vehicle in the formulation. The different inert material can
play the role of the DPIs vehicle. Lactose is applied as the
vehicle in most of the DPI formulations [13,14].
The physicochemical characteristic of the active ingredient
and the DPI formulations is the effective factor on the lung
deposition of the drugs [15]. The stability of drugs and the absence of propellant in the formulation of DPIs proposed
them as a good alternative to MDIs [16]. DPI formulations
consist of drug with an aerodynamic particle size ideally
smaller than 5 m [17]. The flow and dispersion properties of
these small particles are influence by inter particle forces,
including electrostatic, Van der Waals, capillary and
mechanical forces. The intensity of these forces is affected by
several physicochemical properties of particles, including size
distribution, morphology, density and surface composition
[18]. A study has suggested a more important role for powder
formulation than the inhaler design [19]. There are several
methods for production of respirable microparticles such as
supercritical fluid technique [20].
Spray drying is another technique that has been widely used
to manipulate the physical properties of pharmaceutical
materials [21]. Also micronized but spherical particles can be
prepared by spray drying method. The amorphous particles
are characterized by a low area of contact and a smaller and
more homogenous particle size distribution resulting in a
higher respirable fraction than mechanically micronized drugs
[22]. Spray drying also allows a control over particle shape,
morphology and density dependent on the spray drying
conditions [23].
The oral rout of administration of propranolol suffers from
the hepatic first pass metabolism effect [24]. This effect cause
to decrease the oral bioavailability of the propranolol. The oral
absolute bioavailability of the propranolol as the unselective B
blocker was reported in the text 0.3 is the value of the oral
bioavailability of propranolol that has been reported in the
text [25]. In this study the deposition profiles of propranolol
micro particles using spray drying procedures as a dry powder
inhalation was investigated by the in vitro methods and the
pulmonary absorption of these formulations in rats was
measured by the in vivo method.
Materials and Methods
Materials
Propranolol powder was supplied by ABIDI Company (Iran,
Tehran). Lactose monohydrate was purchased from DMV
(Amsterdam, the Netherlands). All solvents which were used
supplied by Merck (Frankfurt, Germany) and were at least
analytical grade.
Methods
Spray drying: Solutions (1 g/50 ml) of propranolol in
different Water:Ethanol ratios (Table 1) were spray dried using
a lab scale spray drier (Buchi 191, Buchi, Switzerland).
| Solvents |
| Water% |
Ethanol% |
Code |
| 100 |
0 |
E:W(0:100) |
| 75 |
25 |
E:W(25:75) |
| 50 |
50 |
E:W(50:50) |
| 25 |
75 |
E:W(75:25) |
Table 1: Spray drying vehicle with different solvent ratios.
Preparation of blends: Powder formulations containing
propranolol and lactose with ratio 1:1 were prepared. In each
mixing process 0.5 gram of spray dried propranolol sample was
blended with 0.5 gram of lactose in a turbula mixer (Dorsa
Iran) at 46 Rev/min for 30 min.
Analytical method: The concentration of propranolol in
plasma samples was measured by HPLC method [26].
Experimental
Particle size analysis
The particle size of samples were determined by laser light
scattering (Malvern mastersizer x, Malvern, UK).
Approximately 20 mg of sample was suspended in water and
sonicated at 25°C for 4 min. A few drop of each sample was
poured into the small volume cell of the instrument to obtain
an obscuration of sample between 18 and 20%. The analysis
was carried out in triplicate for each sample.
p
Scanning electron microscopy
Morphology of each sample was examined by scanning
electron microscopy (SEM) (Philips XL 30 scanning microscope,
Philips, The Netherlands) at 25 Kev. Samples were gold coated
prior to analysis (SCD005 Sputter coater, Bal-Tec, Germany).
Particle density
The bulk density of the samples was determined by
measurement of the volume of a known mass of the material
that had been poured in to a 25 ml graduated cylinder. The
true density was also determined using a helium pycnometer
(Multipycnometer, Quantachrome, USA). Each sample was
analyzed in triplicate.
Drug assay determination
Quantification of propranolol-lactose blend content
uniformity and in vitro lung deposition was by UV-VIS
spectrophotometer at 290 nm. Linearity was confirmed
between 2 and 500 µg/ml. Lactose did not interfere with the
propranolol response.
In vitro deposition
One capsule, containing 10 mg of propranolol was
introduced to an Andersen cascade impactor via a Spinhaler
(dahlia, India). After aerosolization of the powders for 4sec at a
flow rate of 60 l/min, the inhaler, capsule shell, throat,
preseparator, the seven stages and plates and filter were
washed with dichloromethane as the solvent. The
aerodynamic characteristics of propranolol in each sample were determined as follows: Fine particle dose (FPD) was
determined as the amount of drug deposited on stage 1 to the
filter. The effective cut-off diameter of stage 1 of Anderson
cascade impactor at 60 l/min was reported to be <6.18 µm
[27].
Fine Particle Fraction (FPF) was calculated as the percentage
of the ratio of the FPD to the total amount of the drug
recovered per capsule. The emitted dose (ED) was defined as
the total drug recovered from throat, preseparator, seven
stages and plates and filter. The percentage emitted was
calculated as the ratio of ED to the total drug which recovered
per capsule and expressed as percentage. Dispersibility was
defined as the ratio of FPF per ED percentage.
In vivo studies
Male Wister rats (The Pasteur institute, Iran), weighing
250-300 g, were anaesthetized with an intraperitoneal
injection of ketamin (50 mg/kg) and xylene (10 mg/kg). All the
animals were fasted for 16 h before the experiments; they
were allowed free access to water.
Drug administration
3 mg of drug as the powder was introduced into the lung
through the obtuse syringe which was connected through the
tracheal cannula to a depth of 2.5 cm below the tracheal
incision. The tip of the syringe was located 1-2 mm above the
bifurcation of the trachea. The powder was introduced over a
period of 1-2 sec, to the rat which was maintained at an angle
of 80°. Then, the tubing was withdrawn completely and 45 sec
after administration of the powder the animal was positioned
to an angle of 10°.
Propranolol solution in PBS (3 mg/0.2 ml) was intravenously
administered into the caudal vein by bolus injection.
Absorption studies
Absorption of propranolol from rat lung was investigated by
the reported method [28]. All animals were fasted for 16 h
before the experiments but had free access to water. After the
animal was secured on its back on animal board, the trachea
was exposed through a longitudinal incision along the ventral
aspect of the neck. The trachea was then cut transversely,
halfway through, between the fourth and fifth tracheal rings
caudal to the thyroid cartilage.
For determination of the drug concentrations in plasma, 250
µl blood samples were taken from jugular vein
0.25,0.5,0.75,1,1.5,2,4,6,8 after dosing, centrifuged at 1800 g
for 10 min, and the plasma was separated and stored at 30°C
until analysis.
Pharmacokinetic parameters
The pharmacokinetic parameters, Cmax (maximum plasma
concentration) and Tmax (the peak plasma concentration time)
were obtained from the plasma concentration-time curve [18].
AUC0-t and AUCt-8 were calculated by numerical integration using a linear trapezoidal formula. The mean residence time
(MRT) was determined using AUMC/AUC. The Ka (absorption
rate constant) was determined using the residual method, Ke was calculated from terminal section of the plasma
concentration-time curve and T1/2 was calculated from
0.693/ke.
Statistical analysis
The T test as the Statistical Analysis test was apply in this
study. The P<0.05 was consider as the significance.
Results and Discussion
Physical characterization
Table 2 shows the particle size distribution data for all of the
samples. The commercial propranolol was shown to have a
volume median diameter (d 50%) of 37.5 with a mode at
50.99. The SEM photograph is shown in Figure 1 suggests a
columnar shape for the commercial propranolol crystal with a
particle size predominantly smaller than 400 (Figure 1). This
sample had not a suitable particle distribution for inhalation.
| Particle size (μm) |
Density g/ml |
| Sample |
d 50% |
Mode(s) |
Bulk |
True |
| Propranolol before Spray drying |
37.5 ± 1.96 |
50.99 ± 2.46 |
0.673 ± 0.03 |
1.125 ± 0.007 |
| E:W(0:100) |
2.24 ± 0.075 |
4.35 ± 0.22 |
0.202 ± 0.002 |
1.065 ± 0.007 |
| E:W(25:75) |
2.23 ± 0.072 |
3.47 ± 0.02 |
0.383 ± 0.04 |
1.055 ± 0.007 |
| E:W(50:50) |
2.07 ± 0.085 |
3.52 ± 0.02 |
0.281 ± 0.041 |
1.075 ± 0.007 |
| E:W(75:25) |
2.16 ± 0.125 |
3.51 ± 0.02 |
0.254 ± 0.002 |
1.02 ± 0.014 |
Table 2: Particle size distribution and densities of the samples
(Mean, n=3).
Figure 1: SEM photograph of the sample: Propranolol before
spry drying-), E:W(25:75), E:W(75:25).
The spray drying process produced micro particles with
different particle size distribution pattern and densities
depending on the nature of the vehicle, which had been used
in the preparation of the feed. All of the spray dried
propranolol micro particle were shown to have a monomodal
particle size distribution with a particle size smaller than 5.
According to Figure 1 increasing the percentage of ethanol
in the spray drying vehicle caused to formation of the
amorphous and spherical shape propranolol micro particles.
The number of the crystal shaped propranolol micro particles
will be increased by increasing in the percentage of water in
the spray drying vehicle.
According to Table 2 both the true density value and the
bulk density value of the commercial powder were different to
that of the spray dried samples.
Table 2 Particle size distribution and densities of the samples
(Mean, n=3).
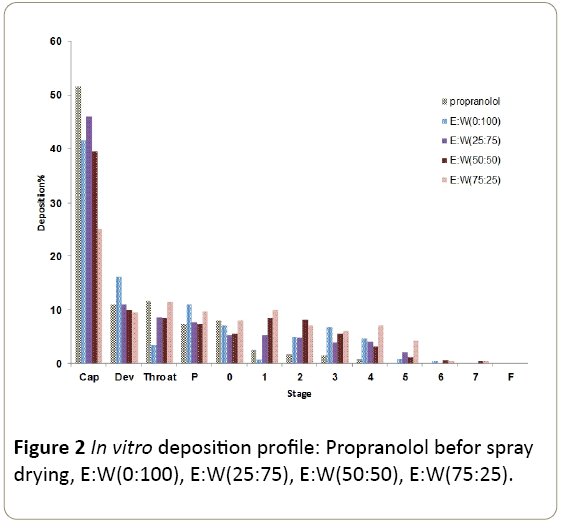
In vitro deposition
Deposition data for each micronized propranolol powder
after aerosolization of the samples at 60 L/min through a
spinhaler ®, using an Andersen cascade impactor are
presented in Figure 2 and Table 3. The amount of propranolol
deposited on various stages of the Andersen cascade impactor
varied for different samples.
Figure 2: In vitro deposition profile: Propranolol befor spray
drying, E:W(0:100), E:W(25:75), E:W(50:50), E:W(75:25).
| Remained in capsule % |
ED% |
FPF% |
Sample |
| 2.32 ± 51.6 |
5.46 ± 45.7 |
1.11 ± 7.61 |
Propranolol |
| 1.53 ± 41.67 |
0.4 ±5 7.33 |
1 ± 19.47 |
E:W(0:100) |
| 1 ± 46 |
0.58 ± 53.57 |
4.57 ± 20.66 |
E:W(25:75) |
| 2.38 ± 39.57 |
1.91 ± 59.8 |
6.54 ± 28.37 |
E:W(50:50) |
| 0.92 ± 25.2 |
0.73 ± 74.5 |
1.15 ± 35.77 |
E:W(75:25) |
Table 3: In vitro deposition data.
These results suggested different aerodynamic properties of
the drug particles aerosolized from commercial and spray
dried samples (p<0.05). The comparison of the effect of the
type of spray drying vehicle on the physicochemical properties
of propranolol indicated many changes in characteristics of
samples such as particle size distribution and in vitro deposition profile.
According to Table 3 Spray dried samples processed from
75% ethanol and 25% water solution produced significantly
(p<0.05) higher percentage emission and higher FPF than the
other spray dried samples and commercial propranolol
powder.
In order to improve the emitted dose (ED) there are two
options, namely, reducing the amount adhering to the surface
of the inside wall of a capsule and improving the flow ability of
the powder [29,30]. As far as the first option is concerned, the
material of the capsule shell could influence drug adhesion
[31]. In the DPI formulation with the Spinhaler®, a
conventional gelatin capsule was adopted as the container for
the unit dose. There are some reports describing the
relationship between the adhesive force and the moisture
content of a polymer film, whereby a higher moisture content
results in a higher amount of adhering particles [32,33]. The
particles captured in stages 1-7 and the filter, were expected
to deposited in the lung lobe or trachea after inhalation. This
was defined as the fine particle dose (FPD). A hard gelatin
capsules usually contains 13%-15% water as a weight ratio and
it is hard to reduce the water below 10% because this makes
the capsules very brittle. The amount of drug remained in the
capsules for each samples was shown in Table 3. Increasing in
the percentage of ethanol in the spray drying vehicle resulted
in the production of micro particles with lower affinity to
remain in the capsule shell. The affinity of micro particles spray
dried from 50% water and 50% ethanol for remaining in the
capsule is more than the others.
The remained drug in the capsule shell is dependent to the
material of the capsule shell and the water content of the
conventional gelatin capsule, there are several reports that
confirm this phenomenon [31-33].
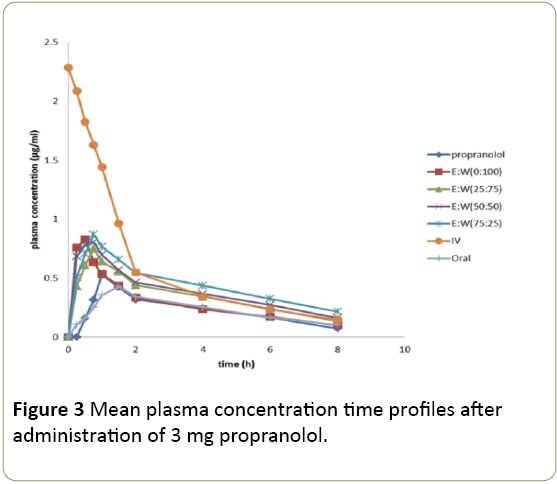
In vivo pulmonary absorption
The pulmonary absorption of drugs was generally influenced
by various physicochemical and biological factors. The
physicochemical factors include molecular size of drugs [34],
lipophilicity of drugs [35], pH in drug solution [36], various
additives [37,38] etc. We examined the effect of various spray
drying propranolol single dose 3 mg was administered via intra
tracheal, intravenous and oral rout to healthy rats and the
plasma concentration of propranolol was measured. The peak concentration achieved via intra tracheal rout was between
30-45 min depending on the type of the spray drying vehicle.
The concentration time profiles of propranolol after intra
tracheal, intravenous and oral administration of propranolol
dry powder inhaler were presented in Figure 3. The
pharmacokinetic parameters of propranolol were summarized
in Tables 4 and Table 5.
Figure 3: Mean plasma concentration time profiles after
administration of 3 mg propranolol.
| Sample |
Tmax (h) |
Cmax (µg/ml) |
Ka (1/h) |
Ke (1/h) |
T1/2 (h) |
| Propranolol |
1 |
0.53 ± 0.081 |
2.39 ± 0.791 |
0.42 ± 0.219 |
1.96 ± 1.021 |
| E:W(0:100) |
0.5 |
0.83 ± 0.111 |
5.76 ± 0.367 |
0.31 ± 0.096 |
2.38 ± 0.7962 |
| E:W(25:75) |
0.75 |
0.75 ± 0.108 |
3.60 ± 0.204 |
0.34 ± 6.798E-17 |
1.99 ± 0.1221 |
| E:W(50:50) |
0.75 |
0.80 ± 0.077 |
2.48 ± 0.200 |
0.24 ± 0.101 |
3.23 ± 1.442 |
| E:W(75:25) |
0.75 |
0.87 ± 0.08 |
3.05 ± 0.507 |
0.34 ± 6.798E-17 |
1.99 ± 0.1231 |
| Oral |
1.5 |
0.53 ± 0.086 |
1.20 ± 0.546 |
0.28 ± 0.111 |
2.83 ± 1.45 |
| IV |
0 |
2.28 ± 0.145 |
------ |
0.25 ± 0.070 |
2.83 ± 0.885 |
Table 4: Pharmacokinetic parameters for the samples after
administration of 3 mg of propranolol.
| Sample |
MRT (h) |
AUC0-8 h
(µgh/ml) |
Cl (ml/h) |
Vd (ml) |
F Bioavailability |
| Propranolol |
3.20 ± 0.189 |
1.84 ± 0.538 |
439.5 ± 8.987 |
4274.87 ± 425.7 |
0.30 ± 0.0931 |
| E:W(0:100) |
2.70 ± 0.326 |
2.31 ± 0.738 |
439.52 ± 4.019 |
3128.85 ± 311 |
0.396 ± 0.161 |
| E:W(25:75) |
3.07 ± 0.126 |
2.81 ± 0.637 |
439.52 ± 9.8 |
3301.76 ± 391 |
0.46 ± 0.114 |
| E:W(50:50 |
3.19 ± 0.224 |
3.59 ± 0.850 |
439.52 ± 2.22 |
2924.57 ± 178.5 |
0.69 ± 0.272 |
| E:W(75:25) |
3.21 ± 0.201 |
3.56 ± 0.768 |
439.52 ± 5.6 |
2954.71 ± 131.8 |
0.61 ± 0.154 |
| Oral |
3.37 ± 0.254 |
1.85 ± 0.456 |
439.52 ± 6.8E |
5374.11 ± 2213 |
0.33 ± 0.068 |
| IV |
2.18 ± 0.229 |
4.69 ± 0.779 |
581.23 ± 130. |
1317.54 ± 86.53 |
1 |
Table 5: Pharmacokinetic parameters for the samples after administration of 3 mg of propranolol.
AUC increased when the percentage of ethanol was
increased in the spray drying vehicle. E: W (50:50) sample
showed the highest value of the AUC and Propranolol showed
the lowest value of the AUC when the powder was administered via the intra tracheal rout. According to Table 4 the values of Tmax and Ka showed that the rate of drug
absorption from the respiratory system is more than the oral
rout of administration (p<0.05).
According to Tables 4 and 5 when the powder administered
by oral and intra tracheal rout there was not any difference
between the pharmacokinetics parameters such as Ke, t1/2,
MRT and Cl related to the elimination phase of propranolol
from the animal body.
Table 4 Pharmacokinetic parameters for the samples after
administration of 3 mg of propranolol.
It is clear that intra tracheal administration of spray dried
propranolol micro particles caused to 2 fold increases in
absolute bioavailability in compare with the oral rout of
administration (E:W(50:50) sample). This phenomenon
confirms the suitability of administration of propranolol micro
particle via the intra tracheal rout which can easily absorbed
from the pulmonary system.
Conclusion
The first advantage of pulmonary rout of administration of
drugs is by passing the first pass metabolism effect when the drug is administered via the pulmonary rout in comparison
with the oral rout. The second advantage of pulmonary rout of
administration of drugs is that the food cannot interact with
the absorption of drug when the drug is administered via the
pulmonary rout in comparison with the oral rout in addition
the different pH related to the different site of gastrointestinal
tract cannot have effect on the absorption of drug when the
patient use the pulmonary rout instead of oral rout. The
pharmacological effect of drug appears faster than the oral
rout.
The results of the study showed that it is possible to prepare
the inhalable propranolol micro particle by spray drying method. The type of the spray drying vehicle is the effective
factor on the physicochemical properties and aerodynamic
behavior of the micro particles. It is important effective factor
on the rate and extent of the pulmonary absorption of drug.
References
- Kumaresan C, Subramanian N, Gover Antoniraj M, Ruckmani K (2012) Dry Powder Inhaler - Formulation aspects. Pharma Times 44: 14-18.
- Mark MB, CoryJB (2009) Nanoparticle formulations in pulmonary drug delivery. Medicinal Research Reviews 29: 196-212.
- Felix R (2001) Effect of inhaled tobramycin on early Pseudomonas aerojinosa colonization in patients with cystic fibrosis, Research letters 358: 983-984.
- Farr SJ, Gonda I, Licko V (1998) Respiratory drug delivery. Interpharm Press, IL, USA: 25-33.
- Surendrakumar K, Martin GP, Hodgers ECM, Jansen M, Blair JA (2003) Sustain release of insulin from sodium hya1uronate based dry powder formulations after pulmonary delivery to beagle dogs. J Controlled release 91: 385-394.
- Wall DA, Smith PL (1997) Inhalation delivery of therapeutic peptides and proteins. New York, USA 453-469.
- Pitt C, Platz RM (1994) US Patent No. 5354934.
- van Zandwijk N (1997) Inhalation delivery of therapeutic peptides and proteins. Marcel Dekker, New York, USA 301-313.
- Malcolmson RJ, Embleton JK (1998) dry powder formulations for pulmonary delivery. Pharm Sci Technol: 394-398.
- Brocklebank DRF, Wright J, Barry P, Cates C, Davies L, et al. (2001) Comparison of the effectiveness of inhaler devices in asthma and chronic obstructive airways disease. a systematic review of the literature. Health Technol Asses 5: 1-149.
- Dolovich MB, Ahrens RC, Hess DR, Anderson P, Dhand R, et al. (2005) Device selection and outcomes of aerosol therapy. Evidence-based guidelines. Chest 127: 335-371.
- David GE (2005) Comparing Clinical Features of the Nebulizer, Metered-Dose Inhaler, and Dry Powder Inhaler. Respiratory Care 50: 1313-1321.
- Louey MD, Stewart PJ (2002) Particle interactions involved in aerosol dispersion of ternary interactive mixtures. Pharm Res 19: 1524-1531.
- Ganderton D, Kassem NM (1992) Dry powder inhalers, Advances in Pharmaceutical Sciences. Academic Press, London: 165-191.
- Heyder J (2004) Deposition of inhaled particles in the human respiratory tract and consequences for regional targeting in respiratory drug delivery. Proc Am Thorac Soc 1: 315-320.
- Morice AH, Adler M, Ellis S, Hewitt A (2000) Do patients prefer dry powder inhalers or metered dose inhaler? A retrospective combined analysis. Curr Ther Res 63: 496-506.
- Zeng, XM, Martin GP, Marriott C (2001) Particle Interaction in Dry Powder Formulations for Inhalations (1stedn). Taylor and francis, London.
- Prime D, Atkins PJ, Slater A, Sumby B (1997) Review of dry powder inhalers. Adv Drug Del Rev 26: 51-58.
- Steckel H, Rasenack N, Muller BW (2003) In-situ-micrinization of disodium cromoglycate for pulmonary delivery. Eur J Pharm Biopharm 55: 173-180.
- Najafabadi AR, Vatanara A, Gilani K, Rafiee-Tehrani M (2005) Formation of salbutamol sulphate microparticles using solution enhanced dispersion by supercritical carbon dioxide. Daru13: 1-5.
- Najafabadi AR, Gilani K, Barghi M, Rafiee-Tehrani M (2004) The effect of vehicle on physical properties and aerosolisation behavior of disodium cromoglycate microparticles spray dried alone or with L-Leucine. Int J Pharm 296: 26-33.
- Dellamary LA, Tarara TE, Smith DJ, Woelk CH, Adractas A, et al. (2000) Hollow porous particles in metered dose inhalers. Pharm Res 17: 168-174.
- Hickey AJ, Martonen TB, Yang Y (1996) Theoretical relationship of lung deposition to the fine particle fraction of inhalation aerosols. Pharm Acta Helv 71: 185-190.
- Routledge PA, Shand D (1979) Clinical pharmacoklnetics of propranolol. Chn Pharmacokinet 4: 73-90.
- Addington WW (1979) Patient compliance: the most serious remaining problem in the control of tuberculosis in the United States. Chest 76: 741-743.
- Xiao S, Wei G, Guo H, Liu H, Liu C (2008) Determination of propranolol in dog plasma by HPLC method. Asian Journal of Pharmacodynamics and Pharmacokinetics 8: 153-158.
- Weda M, Zanen P, de Boer AH, Barends DM, Frijlink HW (2004) An investigation into the predictive value of cascade impactor results for side effects of inhaled salbutamol. Int J Pharm 287: 79-87.
- Enna SJ, Schanker LS (1972) Absorption of saccharides and urea from the rat lung. Am J Physiol 222: 409-414.
- Kawashima Y, Serigano T, Hino T, Yamamoto H, Takeuchi H (1998) Effect of surface morphology of carrier lactose on dry powder inhalation property of pranlucast hydrate. Int J Pharm 172: 178-188.
- Giry K, Pean JM, Girand L, Marsas S, Rolland H, et al. (2006) Drug/lactose co-micronization by jet milling to improve aerosolization properties of a powder for inhalation. Int J Pharm 321: 162-166.
- Podczeck F (1998) Evaluation of the adhesion properties of salbutamol sulphate to inhaler materials. Parm Res 15: 806-808.
- Iida K, Otsuka A, Danjo K, Sunada H (1992) Measurement of adhesive force between particles and polymer-filme. Chem Pharm bull 40: 189-192.
- Shimada Y, Yonezawa Y, Sunada H (2003) Measurement and evaluation of the adhesive force between particles by the direct separation method. J Pharm Sci 92: 560-568.
- Enna SJ, Schanker LS (1972a) Absorption of drugs from the rat lung. Am J Physiol 223: 1227-1231.
- Enna SJ, Schanker LS (1972b) Absorption of saccharides and urea from the rat lung. Am J Physiol 222: 409-414.
- Aracawa E, Kitazawa S (1987) Studies on the factors affecting pulmonary absorption of xanthine derivatives in the rat. Chem Pharm Bull 35: 2038-2044.
- Ohtani T, Murakami M, Yamamoto A, Takada K, Muranishi S (1991) Effect of absorption enhancers on pulmonary absorption of fluorescein isothiocyanate dextrans with various molecular weights. Int J Pharm 77: 141-150.
- Morita T, Yamamoto A, Takakura Y, Hashida M, Sezaki H (1994) Improvement of the pulmonary absorption of (ASu1,7)- elcalcitonin by various protease inhibitors in rats. Pharm Res 11: 909-913.