Case Report - (2022) Volume 6, Issue 6
Rectal Gastrointestinal Stromal Tumors: Usefulness of Endoscopic Ultrasound
Maroua Saidi Idrissi1*,
M Salihoun1,
A Acharki2,
K Znati2 and
N Kabbaj2
1Department of Gastroenterology “EFD-HGE”, Ibn Sina University Hospital,, Morocco
2Department of Anatomical Pathology, Ibn Sina University Hospital, Morocco
*Correspondence:
Maroua Saidi Idrissi, Department of Gastroenterology “EFD-HGE”, Ibn Sina University Hospital,,
Morocco,
Received: 01-Jun-2022, Manuscript No. IPJCGH-22-13536;
Editor assigned: 03-Jun-2022, Pre QC No. IPJCGH-22-13536 (PQ);
Reviewed: 17-Jun-2022, QC No. IPJCGH-22-13536;
Revised: 22-Jun-2022, Manuscript No. IPJCGH-22-13536 (R);
Published:
29-Jun-2022, DOI: 10.36648/2575-7733.6.6.28
Abstract
Rectal gastrointestinal stromal tumors (GIST) account for 0.1% of all rectal tumors. They are generally diagnosed
in adults over 50 years of age with non-specific symptoms. Immunohistochemistry performed on biopsy tissue
sample confirms the diagnosis. In this study, we report two cases of histologically and immunohistochemically confirmed
rectal GISTs diagnosed using Endoscopic ultrasound (EUS) with fine needle aspiration (FNA), a procedure of
crucial value in the diagnosis of these tumors.
Both patients were male, presenting with non-specific symptoms. Standard endoscopy showed a rectal mass in
both cases, biopsies were negative. EUS showed a well-defined hypoechoic mass and evaluated adjacent organs
and of blood/lymphatic nodes involvement. EUS-FNA was performed in both cases. Cytology and immunocytochemistry
studies showed spindle cell pattern with positive immunohistochemical staining for CD117, typical of
GISTs.
Keywords
Rectal Gastrointestinal Stromal Tumor (GIST); Rectal tumor; Endoscopic ultrasound; Fine needle aspiration
Introduction
GISTs are neoplasms of mesenchymal origin rarely located in
the rectum, making up about 0.1% of all tumors arising in the
rectum and accounting for about 2% of all GISTs, with an estimated
incidence rate of 0.45 per million individuals [1,2].
EUS is an important tool for rectal GIST diagnosis, by determining
the wall layer of origin and assessing malignity features [3].
The addition of EUS-guided biopsy allows tissue acquisition for
histopathological confirmation.
We report two new cases of rectal GISTs, detailing the clinicopathological
and endoscopic features of this tumor.
Case Presentation
Case 1
A 60 year old male, with no relevant past medical or surgical history, complained of lower abdominal pain located in the
hypogastric region and constipation. Clinical examination was
unremarkable and digital rectal exam (DRE) was normal. Colonoscopy
identified an ulcerated mass measuring 20 mm in diameter,
located about 12 cm from the anal verge. EUS showed
a well-defined hypoechoic heterogenous mass with central calcifications
located in the middle rectum, measuring 36, 6 mm
× 25 mm, originating from the muscularis propria, compressing
the bladder and coming in contact with the distal ileum (Figures
1 and 2). EUS-FNA has been successfully performed using
a 19-gauge (G) needle, with 2 passes. The cytopathological
analysis showed spindle cells containing ovoid nucleus with
fine chromatin. Immunohistochemical staining showed positive
expression for CD117 and Dog1, consistent with a gastrointestinal
stromal tumor (Figure 3).
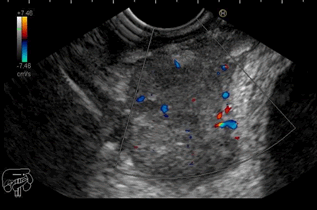
Figure 1: EUS showed a well-defined heterogeneous slightly echogenic solid mass with central calcifications and no significant flow on color Doppler
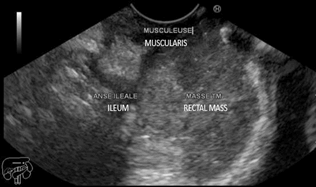
Figure 2: EUS revealed contact between the rectal mass and the distal ileum
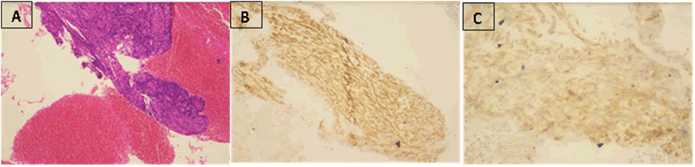
Figure 3: Cytological and immunohistochemical findings. Cell blocks showing spindle cells containing ovoid nucleus with fine chromatin x (A, hematoxylin and eosin [H and E], x 100), week nuclear staining for dog1(B x 400) and positive staining for CD117 in tumor cells (C x 400)
Case 2
A 49 year old male, presented with abdominal pain and cramping, tenesmus, and incomplete stool evacuation stool. DRE
found a palpable indurated mass rectal mass 5 cm from the
anal verge. Flexible sigmoidoscopy showed an ulcerative budding
mass extending from 5 cm from the anal verge to 25 cm,
repeated biopsies were negative.
EUS revealed a noncircumferential submucosal solid hypoechoic
mass invading the mucosa, located in the lower rectum
extending to 25 cm from the anal margin, where it became
stenosing. The rectal sphincters were not invaded; multiples
perirectal lymph nodes were detected.
EUS-FNA performed using a 19-G needle with 3 passes. Cytology
and immunocytochemistry studies showed spindle cell
pattern with positive Immunohistochemical staining for CD117,
typical of GISTs.
Discussion
Rectal GISTs generally occur in middle aged individuals between
the fifth and sixth decades, predominantly in males [4]. In our
study both patients were male with a mean age of 54 years, in
line with the pathological literature. GISTs probably originate
from the interstitial cells of Cajal, are spindly shaped (70% of
cases) and typically defined as CD 117/KIT and/or DOG1 positive,
making immunohistochemical staining for these markers
an important diagnostic tool [5]. Rectal GISTs have a worse
prognosis than other gastro-intestinal locations and high local
recurrence rate [2]. After rectal GIST resection, the most common
cause of death GIST is distant metastasis, with a frequent
involvement of the liver [1].
Clinically, the reported symptoms of rectal GISTs are rectal
bleeding, tenesmus, abdominal pain with painful contractions, abnormal evacuation (mucus, pus, etc.), transit disorders,
urinary complaints or altered general condition [6-8]. In this
study, the most frequent symptoms were abdominal pain and
constipation.
Rectal GISTs are frequently found incidentally, either on
cross-sectional imaging, screening colonoscopy, or clinical examination.
Low lying GISTs may be felt as a smooth, firm mass
on physical examination [2]. In our study, both patients were
symptomatic and physical examination identified a rectal mass
in one case.
Computed tomography and magnetic resonance imaging are
useful imaging methods for detection and staging of these
neoplasm [2,9]. Rectal GISTs appear as large, bulky, exophytic
rectal masses with heterogenous enhancement. The presence
of features such as well demarcated margins, prominent extraluminal
location and no surrounding adenopathy, and lack of
bowel lumen constriction despite the large size of the tumor,
allow GISTs to be differentiated from malignant epithelial neoplasms
[8].
Endoscopic examinations are key in rectal GIST’s diagnosis.
GISTs can have an exophytic and/or intramural pattern of
growth. The main endoscopic finding is a round smooth mass
covered with normal mucosa in endoluminal tumors, and a
stiffening of the rectal wall in exophytic forms [10]. Other findings
on endoscopy such as Irregular borders, ulceration, and growth during endoscopic follow up are considered clinically
malignant features [11].
Conventional biopsy by endoscopy is frequently negative since
biopsy sample provides only mucosal tissue [10,12]. The Reported
diagnostic yield of this approach is poor, ranging from
17 to 42% [12]. In this context, EUS is an invaluable tool for
the diagnosis, by determining the wall layer of the origin and
assessing its echogenicity [3]. Since rectal GISTs typically arise
within the muscularis propria [8].
The typical EUS features of a GIST are a hypoechoic, round to
oval, homogeneous mass, with well-defined contours. GISTs
are seen as fourth layer tumors in EUS. Large or clinically malignant
GISTs can display irregular margins [12]. EUS finding such
as irregular shape, heterogeneous echotexture, central necrosis,
echogenic foci or calcification are associated with high risk
GISTs [13]. Contrast enhanced harmonic (CH) imaging of EUS
is a useful tool in differentiating GISTs from benign SELs and
evaluating the malignancy potential of GISTs.
Hyper enhancement pattern on CH-EUS has an accuracy ranging
from 82.2 to 100% in the diagnosis of GISTs [3]. CH-EUS allows
the visualization of the intratumoral micro vascularity to
estimate the malignancy potential of GISTs [14]. CH-EUS findings
predicting high grade malignancy are irregular and abundant
intra tumor blood vessels, heterogeneous enhancement
patterns and the existence of non-enhancing spots [3].
The principal differential diagnoses of GISTs by imaging methods
and EUS are schwannoma, leiomyoma, leiomyosarcoma
[14]. Thus, tissue sampling is required for a conclusive diagnosis.
EUS-FNA is an effective tissue sampling method for the diagnosis
of submucosal lesions [12], with a diagnostic accuracy
rate ranging from 70 to 90% [11].
For the diagnosis of GISTs, the sensitivity of EUS-FNA cytology
was 78.4%, influenced by size, location, shape, and layer of origin
in a study by Sepe PS and al [15]. However, despite EUS-FNA
good accuracy in identifying spindle cells and mitotic activity, it
has a limited ability to procure tissue for immunohistochemistry,
which is a critical element in establishing the diagnosis and
prognosis of GISTs [16].
EUS-FNA has an inconsistent diagnostic yield in different
studies, ranging from 46 % to 93 % [17]. The non-diagnostic
FNA-specimens in GISTs, can leads to a lack of prognostic information
based on the tumor mutation profile and proliferation
rate, making it an obstacle for the early personalized management
of GISTs [18].
Core needles for EUS-guided fine-needle biopsy (EUS-FNB)
emerged to overcome the limitations of EUS-FNA and optimize
the diagnostic yield of lesions such as GISTs [17].
In a retrospective large multicenter study, FNB for suspected
GISTs was superior to EUS-FNA in establishing the diagnosis of
GISTs, with a diagnostic yield for FNB more than double that of
FNA, making FBN an effective method in preventing repeated
EUS procedures or unnecessary surgery on benign lesions discovered
[17]. In our study, EUS-FNA was the mainstay of obtaining
tissue and allowed an accurate pathological diagnosis
of the tissue specimen in both cases of rectal GISTs.
Conclusion
EUS is a crucial technique in the diagnosis of rectal GISTs. It
identifies the affected rectal layer, assesses the tumor size, and
evaluates the involvement of adjacent organs and blood/lymphatic
vessels for tumor staging. The addition of FNA or FNB to
EUS enables tissue analysis for diagnosis confirmation and high
risk features identification, thus helping in the guidance for the
best therapeutic management. In our case report, EUS helped
the dagnosis of two case of rectal Gists by providing tissue sapling
for cytological and immunohistochemical analysis.
REFERENCES
- Hamada M, Ozaki K, Horimi T, Tsuji A, Nasu Y, et al. (2008) Recurrent rectal GIST resected successfully after preoperative chemotherapy with imatinib mesylate. Int J Clin Oncol. 13(4):355- 360.
[Crossref] [Google Scholar] [PubMed]
- Kane WJ, Friel MC (2019) Diagnosis and treatment of rectal gastrointestinal stromal tumors. Dis Colon Rectum. 62(5):537-540.
[Crossref] [Google Scholar] [PubMed]
- Tamura T, Kitano M (2019) contrast enhanced endoscopic ultrasound imaging for gastrointestinal subepithelial tumors. Clin Endosc. 52(4): 306-313.
[Crossref] [Google Scholar] [PubMed]
- Miettinen M, Furlong M, Sarlomo-Rikala M, Burke A, Sobin LH, et al. (2001) Gastrointestinal stromal tumors, intramural leiomyomas and leiomyosarcomas in the rectum and anus: A clinicopathologic, immunohistochemical, and molecular genetic study of 144 cases. Am J Surg Pathol. 25:1121-1133.
[Crossref] [Google Scholar] [PubMed]
- Lee SJ, Hwang CS, Kim A, Kim K, Choi KU (2016) Gastrointestinal tract spindle cell tumors with interstitial cells of Cajal: Prevalence excluding gastrointestinal stromal tumors. Oncol Lett,12:1287-1292.
[Crossref] [Google Scholar] [PubMed]
- Rejab H, Kridis WB, Ameur HB, Feki J, Frikha M, et al. (2014) Tumeur stromale rectale: A propos d’une observation. Pan Afr Med J. 17:119. French.
[Crossref] [Google Scholar] [PubMed]
- Jallouli A, Jarti M, Haida MZ, El Bouatmani M, Errami AA, et al. (2021) rectal gastrointestinal stromal tumor (gist): about a rare and unusual case. International Journal of Innovative Research in Medical Science. 6(11): 814-817.
[Crossref] [Research Gate]
- Jiang ZX, Zhang SJ, Peng WJ, Yu BH (2013) Rectal gastrointestinal stromal tumors: Imaging features with clinical and pathological correlation. World J Gastroenterol. 19(20):3108-3116.
[Crossref] [Google Scholar] [PubMed]
- Puckett Y, Aryaie A (2017) Case report of diffusely metastatic rectal GIST. Int J Surg Case Rep.37:4-9.
[Crossref] [Google Scholar] [PubMed]
- Samlani-Sebbane Z, Diffaa A, Charaf K, Rabbani K, Narjis Y, et al. (2011) Le GIST rectal: A propos de deux observations et une revue de la littérature. J Afr Hepato Gastroenterol. 5(1):60-2.
[Crossref] [Google Scholar] [Research Gate]
- Nishida T, Kawai N, Yamaguchi S, Nishida Y (2013) Submucosal tumors: Comprehensive guide for the diagnosis and therapy of gastrointestinal submucosal tumors. Dig Endosc. 25:479-489.
[Crossref] [Google Scholar] [PubMed]
- Sepe PS, Brugge WR (2009) A guide for the diagnosis and management of gastrointestinal stromal cell tumors. Nat Rev Gastroenterol Hepatol.6:363-371.
[Crossref] [Google Scholar] [PubMed]
- Kang JH, Lim JS, Kim JH, Hyung WJ, Chung EY, et al. (2009) Role of EUS and MDCT in the diagnosis of gastric submucosal tumors according to the revised pathologic concept of gastrointestinal stromal tumors. Eur Radiol. 19:924–934.
[Crossref] [Google Scholar] [PubMed]
- Sakamoto H, Kitano M, Kudo M (2010) Diagnosis of subepithelial tumors in the upper gastrointestinal tract by endoscopic ultrasonography. World J Radiol. 2(8):289-297.
[Crossref] [Google Scholar] [PubMed]
- Sepe PS, Moparty B, Pitman MB, Saltzman JR, Brugge WR (2009) EUS-guided FNA for the diagnosis of GI stromal cell tumors: Sensitivity and cytologic yield. Gastrointest Endosc. 70:254-261
[Crossref] [Google Scholar] [PubMed]
- Obuch J, Wani S (2017) EUS-guided tissue acquisition in GI stromal tumors. Gastrointest Endosc. 86:516- 518.
[Crossref] [Google Scholar] [PubMed]
- Trindade AJ, Benias PC, Alshelleh M, Bazarbashi AN, Tharian B, et al. (2019) Fine- needle biopsy is superior to fine-needle aspiration of suspected gastrointestinal stromal tumors: A large multicenter study. Endosc Int Open. 7(7):E931- E936.
[Crossref] [Google Scholar] [PubMed]
- Hedenström P, Nilsson B, Demir A, Andersson C, Enlund F, et al. (2017) Characterizing gastrointestinal stromal tumors and evaluating neoadjuvant imatinib by sequencing of endoscopic ultrasound-biopsies. World J Gastroenterol. 23(32):5925-5935.
[Crossref] [Google Scholar] [PubMed]
Citation: Idrissi MS, Salihoun M, Acharki A, Znati K, Kabbaj N (2022) Rectal Gastrointestinal Stromal Tumors: Usefulness of Endoscopic Ultrasound. J Clin Gastroenterol Hepatol.6 No.6.28
Copyright: © Idrissi MS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.