Keywords
Monastrol; Dihydropyrimidinones; Bignelli’s; IC50; Pharmacophore
Introduction
Dihydropyrimidinones (DHPMs) are the products of Bignelli’s reaction, which is also known as multicomponent reaction. A multicomponent reaction (MCR) is the process in which three or more reactants are combined in a single step to form a product that incorporates structural individuality of each reagent. MCRs affectionate ease and effectiveness to synthetic approaches [1]. Dihydropyrimidinones consist of pyrimidine core unit having resemblance with the structure of nucleic acid bases found in DNA and RNA. Their involvement in DNA and RNA has great importance in drug design [2]. Recent progress in the DHPM class of the anticancer agent monastrol 5, an allosteric inhibitor of human kinesin Eg5 has led to the interest for proficient pharmacophore variation of Biginelli DHPMs [3]. Human kinesin Eg5 plays a critical role in bipolar spindle generation during mitosis, inhibition of which leads to mitotic arrest and consequent apoptotic cell death. It is consequently considered as one of the potential targets in cancer chemotherapy [4].
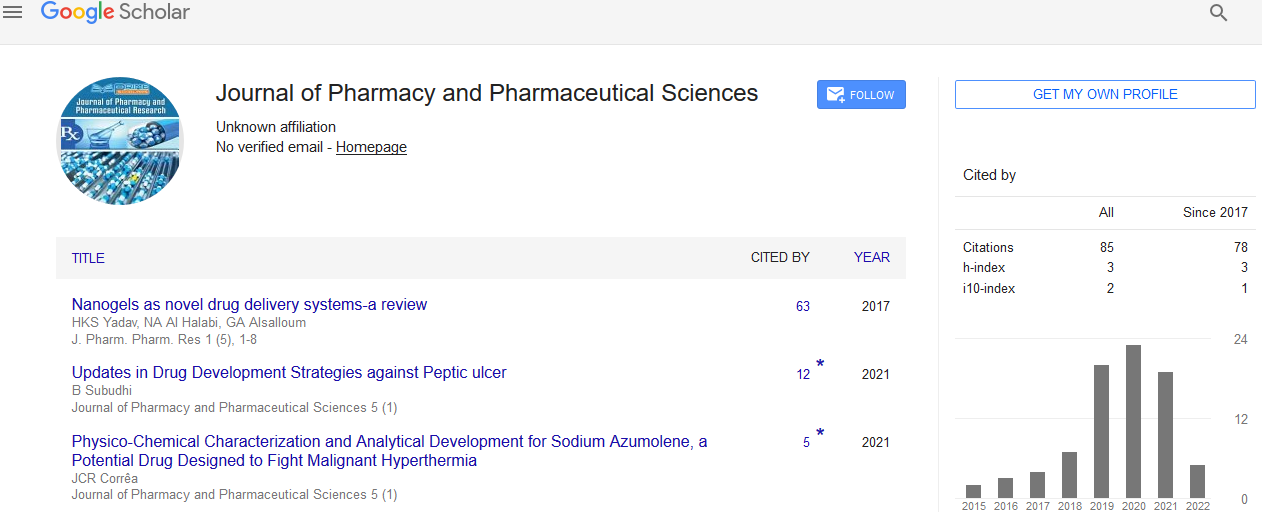
Pietro Bignelli in 1893 discovered Bignelli’s reaction [5] that was known as MCR that involves condensation of acetoacetic esters 1, urea or thiourea 2 and aromatic aldehydes 3 in ethanol with catalytic amount of HCl (Figure 1). The product of condensation of three components was known as DHPM 4.
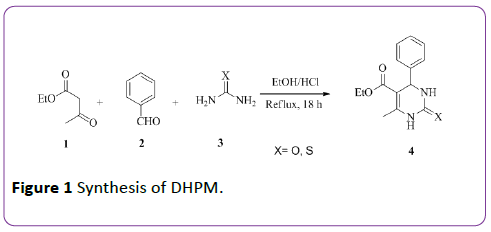
Figure 1: Synthesis of DHPM.
Thus, special attention was given to this MCR in the late 20th century to synthesize many compounds comprising DHPM core unit with different biological activities. Batzeladine 6 is a natural compound isolated from Batzella species having DHPM core unit and reported to exhibit potent HIV-gp-120-CD4 inhibitor. Some of DHPMs compounds like SQ329267 and SQ325478 were reported to exhibit anti-hypertensive activity [6] as shown in Figure 2.
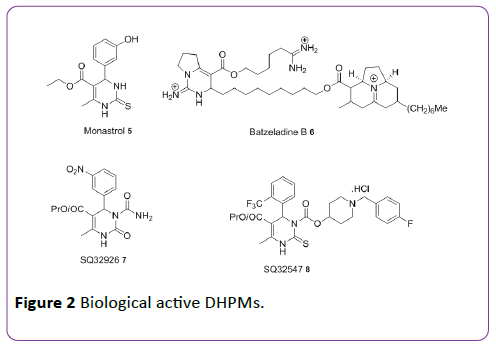
Figure 2: Biological active DHPMs.
Various pharmacological activities of fused analogues of DHPMs have been reported such as anti-inflammatory activity, anti-bacterial, anticancer activity [7-8] etc. Fused pyrimidines continue to attract significant attention because of their great practical utility, primarily due to very wide spectrum of biological activities. Scheele et al. and co-workers have begun the chemistry of fused pyrimidines in 1766 when he isolated uric acid [9-10] and reported the pharmacological study of various fused pyrimidine derivatives.
Aly et al. [11] and Nagarajaiah et al. [12] have reported the synthesis and biological screening of fused pyrimidine analogues against bacterial species. Various scaffolds derived from DHPMs are shown in Figure 3 [13].
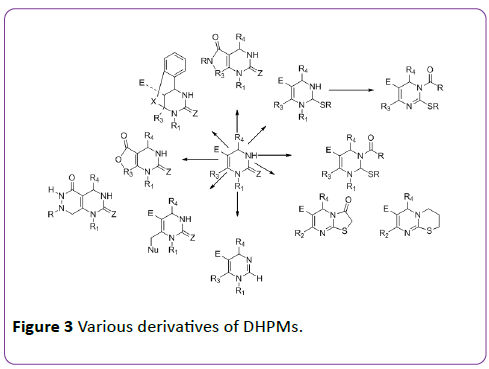
Figure 3: Various derivatives of DHPMs.
By taking into consideration of importance of DHPMs core unit, we have synthesized some bicyclic analogues of dihydropyrimidine. It has reported that N3 and C5 substitutions in DHPMs are very important for enhancement of the biological activity [14] of such compounds.
Thiazolo-pyrimidine and pyrimido-thiazine has great importance in medicinal chemistry. Therefore, by taking into consideration of importance of these bicyclic systems, we have synthesized some bicyclic analogues of dihydropyrimidine with substitution at C5 with benzoyl moiety.
Materials and Methods
Melting points were taken in capillary tubes and were determined on VEEGO melting point apparatus and were uncorrected. Infrared spectra were obtained on THERMO FT-IR spectrophotometer. Nuclear magnetic resonance (NMR) spectra were recorded on Bruker AC-400 F, 400MHz spectrometer in CDCl3/DMSO.
Chemical shifts were reported in parts per million (ppm) with tetramethylsilane as the internal standard. Data reported were as follows: chemical shift, multiplicity as singlet (s), doublet (d), triplet (t), quartet (q), broad singlet (br s), multiplet (m), and coupling constants (Hz).
The reactions were monitored by thin layer chromatography (TLC) using F254 silica gel pre-coated TLC plates (Merck) and column chromatography using silica gel (60 to 120 mesh). Solvents used for synthesis, extraction and chromatography were dried and freshly distilled. All evaporations were carried out under reduced pressure on Buchii-Rotary evaporator.
General method for synthesis of 4- (substituted-phenyl)-6-methyl-2- thioxo-1,2,3,4-tetrahydropyrimidine-5-ylphenylmethanone (RS-1 to RS-15)
Benzoylacetone 9 (0.01 mol), substituted arylaldehyde 10 (0.01 mol), thiourea 11 (0.015 mol) was dissolved in minimum quantity of methanol. Few drops of conc. HCl were added to the reaction mixture as a catalyst. The reaction mixture was then refluxed in water bath for 6 hours. The progress of reaction was monitored by TLC. The reaction mixture was allowed to cool at room temperature. The solid separated upon cooling was filtered, washed with cold methanol and characterized as 4- (substituted-phenyl)-6-methyl-2-thioxo-1,2,3,4- tetrahydropyrimidine-5-yl-phenylmethanone (RS-1 to RS-5).
The 4-(substituted-phenyl)-6-methyl-2-thioxo-1,2,3,4- tetrahydropyrimidine-5-yl-phenylmethanone (RS-1 to RS-5) (0.005 mol) was dissolved in dimethylformamide (DMF) and then added 1,2-dibromoethane (0.01 mol) or 1,3- dibromopropane (0.01 mol) into it and refluxed it at 140°C for 3 h. The reaction mixture was poured into crushed ice. The resultant product was filtered off to collect the final 5- (substituted-phenyl)-7-methyl-3,5-dihydro[3,2-a] [1,3]pyrimidin-6-yl)(phenyl)methanone (RS-6 to RS-10) or 6- (substituted-phenyl)-8-methyl-2,3,4,6-tetrahydsssropyrimido [2,1-b][1,3]thiazin-7-yl)(phenyl) methanone (RS-11 to RS-15).
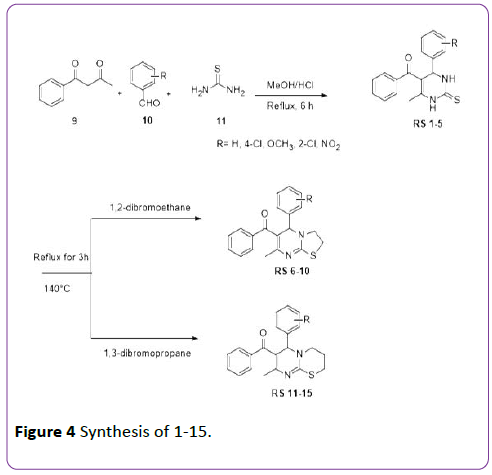
Figure 4: Synthesis of 1-15.
Characterization of 4-phenyl-6-methyl-2- thioxo-1,2,3,4-tetrahydropyrimidin-5yl)- (phenyl) methanone (RS-1)
Mol. Formula: C18H16N2OS; Mol. Wt. 308; Physical state: solid; Color: yellow; Isolated yield: 81%; MP: 223-225°C; TLC System: Hexane: Ethyl acetate (7:3); Rf value: 0.46, FTIR (KBr) cm-1: 3299 (N-H str.), 2968-3191 (C-H str.), 1735 (C=O), 1618 (C=C), 1210 (C=S str.), 1330 (C-N str.).
Characterization of 4-(4-chlorophenyl)-6-methyl-2- thioxo-1,2,3,4-tetrahydropyrimidin- 5yl) (phenyl)methanone (RS-2)
Mol. Formula: C18H16N2OClS; Mol. Wt. 343; Physical state: solid; Color: yellow; Isolated yield: 74%; MP: 243-245°C; TLC System: Hexane: Ethyl acetate (7:3); Rf value: 0.52, FTIR (KBr) cm-1: 3280 (N-H str.), 3282 (C-H str.), 1676 (C=O str), 1599 (C=C str), 1264 (C=S), 1350 (C-N str), 766 (C-Cl str.).
Characterization of 4-(4-methoxyphenyl)-6- methyl-2-thioxo-1,2,3,4-tetrahydro pyrimidin-5yl) (phenyl)methanone (RS-3)
Mol. Formula: C19H19N2O2S; Mol. Wt. 339; Physical state: solid; Color: yellow; Isolated yield: 63%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (7:3); Rf value: 0.61, FTIR (KBr) cm-1: 3285 (N-H str.), 3173 (C-H str.), 1588 (C=O str), 1572 (C=C str), 1197 (C=S), 1377 (C-N str), 1107 (C-O).
Characterization of 4-(2-chlorophenyl)-6-methyl-2- thioxo-1,2,3,4-tetrahydropyrimidin-5yl) (phenyl)methanone (RS-4)
Mol. Formula: C18H16N2OClS; Mol. Wt. 343; Physical state: solid; Color: brown; Isolated yield: 71%; MP: 178-180°C; TLC System: Hexane: Ethyl acetate (7:3); Rf value: 0.54, FTIR (KBr) cm-1: 3448 (N-H str.), 2931 (C-H str.), 1685 (C=O str), 1602 (C=C str), 1172 (C=S), 1380 (C-N str), 766 (C-Cl).
Characterization of 4-(4-nitrophenyl)-6-methyl-2- thioxo-1,2,3,4-tetrahydropyrimidin-5yl) (phenyl)methanone RS-5
Mol. Formula: C18H16N3O3S; Mol. Wt. 354; Physical state: solid; Color: yellow; Isolated yield: 72%; MP: 181-183°C; TLC System: Hexane: Ethyl acetate (7:3); Rf value: 0.53, FTIR (KBr) cm-1: 3317(N-H str.), 2844-2983 (C-H str.), 1626 (C=O str), 1598 (C=C str), 1171 (C=S), 1375 (C-N str), 1375-1563 (N-O).
Characterization of 5-phenyl)-7-methyl-3,5- dihydro[3,2-a][1,3]pyrimidin-6-yl) (phenyl)methanone (RS-6)
Mol. Formula: C20H18N2OS; Mol. Wt. 334; Physical state: semisolid; Isolated yield: 71%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.51, FTIR (KBr) cm-1: 2848-2917 (C-H str.), 1724 (C=O str), 1596 (C=C str), 1375 (C-N str), 717 (C-S str.), 1HNMR 400 MHz, CDCl3 TMS=0, δ: 2.65 (2H, s), 2.89 (3H, s), 3.2-3.35 (2H, m), 5.9 (1H, s), 7.15 (1H, d,J=8.89 Hz,Ar-H), 7.22-7.29 (2H, m, Ar-H), 7.34-7.39 (2H, m, Ar-H), 7.76-7.79 (3H, m, Ar-H), 7.89-7.92 (2H, m, Ar-H).
Characterization of 5-(4-chlorophenyl)-7-methyl-3,5- dihydro[3,2-a][1,3]pyrimidin-6-yl) (phenyl)methanone (RS-7)
Mol. Formula: C20H18N2OSCl; Mol. Wt. 369; Physical state: semisolid; Isolated yield: 68%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.46, FTIR (KBr) cm-1: 2935 (C-H str.), 1715 (C=O str), 1667 (C=C str), 1390 (C-N str), 663 (C-S) 1HNMR: 400 MHz, CDCl3 TMS=0, δ: 2.75 (2H, s), 2.91 (3H, s), 3.0-3.2 (2H, m), 5.96 (1H, s), 7.35 (2H, d,J=8.89 Hz,Ar-H), 7.52-7.59 (4H, m, Ar-H), 7.74-7.79 (3H, m, Ar-H).
Characterization of 5-(4-methoxyphenyl)-7- methyl-3,5-dihydro[3,2-a][1,3]pyrimidin-6-yl) (phenyl)methanone (RS-8)
Mol. Formula: C21H21N2O2S; Mol. Wt. 365; Physical state: semisolid; Isolated yield: 58%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4), Rf value: 0.61, FTIR (KBr) cm-1: 2848-2917 (C-H str.), 1724 (C=O str), 1596 (C=C str), 1375 (C-N str), 717 (C-S str.), 1172 (C-O str.), 1HNMR: 400 MHz, CDCl3 TMS=0, δ: 2.55 (2H, s), 2.93 (3H, s), 3.34-3.36 (2H, m), 4.12 (3H, s, OCH3), 5.81 (1H, s), 7.25 (2H, d,J=8.28 Hz,Ar-H), 7.32-7.37 (4H, m, Ar-H), 7.54-7.59 (3H, m, Ar-H).
Characterization of 5-(2-chlorophenyl)-7-methyl-3,5- dihydro[3,2-a][1,3]pyrimidin-6-yl) (phenyl)methanone (RS-9)
Mol. Formula: C20H18N2OSCl; Mol. Wt. 369; Physical state: semisolid; Isolated yield: 62%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.54, FTIR (KBr) cm-1: 2845-2931 (C-H str.), 1699 (C=O str), 1586 (C=C str), 1373 (C-N str), 715 (C-S), 635 (C-Cl).
Characterization of 5-(4-nitrophenyl)-7-methyl-3,5- dihydro[3,2-a][1,3]pyrimidin-6-yl) (phenyl)methanone (RS-10)
Mol. Formula: C20H18N3O3S; Mol. Wt. 380; Physical state: semisolid; Isolated yield: 65%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.51, FTIR (KBr) cm-1: 2934-3073 (C-H str.), 1691 (C=O str), 1596 (C=C str), 1335 (C-N str), 710 (C-S), 664 (C-Cl), 1HNMR: 400 MHz, CDCl3 TMS=0, δ: 2.71 (2H, s), 3.2 (3H, s), 3.9-4.2 (2H, m), 5.85 (1H, s), 7.35 (2H, d,J=8.89 Hz,Ar-H), 7.52-7.59 (2H, m, Ar-H), 7.74-7.79 (3H, m, Ar- H), 7.87-7.89 (2H, m,Ar-H).
Characterization of 6-phenyl-8-methyl-2,3,4,6- tetrahydropyrimido[2,1-b][1,3]thiazin-7-yl) (phenyl)methanone (RS-11)
Mol. Formula: C21H20N2OS; Mol. Wt. 348; Physical state: semisolid; Isolated yield: 73%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.49, FTIR (KBr) cm-1: 2926-3068 (C-H str.), 1722 (C=O str), 1592 (C=C str), 1345 (C-N str), 702 (C-S str.), 1HNMR: 400 MHz, CDCl3 TMS=0, δ:2.19-2.25 (1H, m), 2.36-2.41 (1H, m), 2.95 (2H, s), 3.2 (3H, s), 3.39-3.40 (2H, m), 5.6 (1H, s), 6.85 (1H, s,Ar-H), 7.24-7.28 (2H, d, J= 6.85Hz, Ar-H)7.32-7.35 (2H, d, J=6.85Hz Ar-H), 7.44-7.49 (3H, m, Ar-H), 7.65-7.69 (2H, m, Ar-H).
Characterization of 6-(4-chlorophenyl)-8- methyl-2,3,4,6-tetrahydropyrimido[2,1-b] [1,3]thiazin-7-yl)(phenyl)methanone (RS-12)
Mol. Formula: C21H20N2OSCl; Mol. Wt. 383; Physical state: semisolid; Isolated yield: 64%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.42, FTIR (KBr) cm-1: 2880-3060 (C-H str.), 1721 (C=O str), 1612 (C=C str), 1386 (C-N str), 716 (C-S), 660 (C-Cl), 1HNMR: 400 MHz, CDCl3 TMS=0, δ: 2.14-2.18 (1H, m), 2.22-2.26 (1H, m), 2.85 (2H, s), 3.0 (3H, s), 3.42-3.49 (2H, m), 5.93 (1H, s), 6.91 (2H, d,J=7.36 Hz, Ar-H), 7.45-7.48 (2H, m, Ar-H), 7.54-7.59 (3H, m, Ar-H), 7.65-7.78 (2H, m, Ar-H).
Characterization of 6-(4-methoxyphenyl)-8- methyl-2,3,4,6-tetrahydropyrimido[2,1-b] [1,3]thiazin-7-yl)(phenyl)methanone (RS-13)
Mol. Formula: C22H23N2O2S; Mol. Wt. 379; Physical state: semisolid; Isolated yield: 61%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.59, FTIR (KBr) cm-1: 2872-2931 (C-H str.), 1659 (C=O str), 1509 (C=C str), 1387 (C-N str), 664 (C-S), 1062 (C-O), 1HNMR: 400 MHz, CDCl3 TMS=0, δ: 2.11-2.15 (1H, m), 2.26-2.29 (1H, m), 2.85 (2H, s), 3.0 (3H, s), 3.32-3.35 (2H, m), 3.8 (3H, s, OCH3), 5.6 (1H, s), 6.85 (2H, d, J=8.68 Hz, Ar-H), 7.32-7.35 (4H, m, Ar-H), 7.44-7.49 (3H, m, Ar- H).
Characterization of 6-(2-chlorophenyl)-8- methyl-2,3,4,6-tetrahydropyrimido[2,1-b] [1,3]thiazin-7-yl)(phenyl)methanone (RS-14)
Mol. Formula: C21H20N2OSCl; Mol. Wt. 383; Physical state: semisolid; Isolated yield: 57%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.53, FTIR (KBr) cm-1: 2923-3071 (C-H str.), 1695 (C=O str), 1597 (C=C str), 1333 (C-N str), 712 (C-S), 686 (C-Cl).
Characterization of 6-(4-nitrophenyl)-8- methyl-2,3,4,6-tetrahydropyrimido[2,1-b] [1,3]thiazin-7-yl)(phenyl)methanone (RS-15)
Mol. Formula: C21H20N3O3S; Mol. Wt. 394; Physical state: semisolid; Isolated yield: 63%; MP: 185-187°C; TLC System: Hexane: Ethyl acetate (6:4); Rf value: 0.48, FTIR (KBr) cm-1: 2844-3054 (C-H str.), 1710 (C=O str), 1549 (C=C str), 1374 (C-N str), 738 (C-S), 1221-1447 (N-O), 1HNMR: 400 MHz, CDCl3 TMS=0, δ:2.16-2.25 (1H, m), 2.32-2.39 (1H, m), 3.09 (2H, s), 3.4 (3H, s), 3.52-3.58 (2H, m), 5.68 (1H, s), 7.25 (2H, d, J=8.68 Hz, Ar- H), 7.39-7.41 (2H, m, Ar-H), 7.54-7.59 (3H, m, Ar-H), 8.49-8.53 (2H, m, Ar-H).
Experiment Procedure
HepG2 cells were plated in 96multiwell plate (104 cells/well) for 24 h before treatment with the compounds to allow attachment of cell to the wall of the plate, in which cell number was kept at 4000-5000 using neubauer cell counting chamber. Test compounds were dissolved in DMSO and diluted with HBBS media to the appropriate volume. After 24 h of incubation, different concentrations of the compounds under test (6.25, 12.5, 25.0, 50.0 and 100 μM) were added to the cell monolayer. Triplicate wells were prepared for each individual dose. Then cultures were incubated were incubated with the compounds for 72 h at 37°C and in atmosphere of 5% CO2. After 72 h, cells were fixed, 5 μL of MTT (5 mg/ml in DMSO) was added to each well, and the plate was incubated for a further four hours at 37°C in dark. The resulting formazon was dissolved in 50 μL dimethyl sulphoxide and absorbance of the solution was read at 595 nm using an ELISA plate reader (Biorad, Model 680, Japan). The relation between surviving fraction and drug concentration was plotted to get the survival curve of each tumor cell line after the specified time. The concentration required for 50% inhibition of cell viability (IC50) was calculated.
Result and Discussion
In this study the new fused dihydropyrimidine derivatives (RS 6-15) were synthesized by cyclization of 4-(substitutedphenyl)- 6-methyl-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-ylphenylmethanone (RS 1-5) with 1,2-dibromoethane or 1,3- dibromopropane under reaction conditions as shown in scheme 2. In this reaction, thio and nitrogen atoms of DHPM acts as nucleophile which attacks on methylene atoms of 1,2- dibromoethane or 1,3-dibromopropane and leads to final products 5-(R-phenyl)-7-methyl-3,5-dihydro[3,2-a] [1,3]pyrimidin-6-yl)(phenyl)methanone (RS 6-10) or 6-(Rphenyl)- 8-methyl-2,3,4,6-tetrahydropyrimido[2,1-b] [1,3]thiazin-7-yl)(phenyl)methanone (RS 11-15). The synthesized compounds (RS 6-15) were characterized by TLC, IR and NMR. In IR spectra C=O strechings were found around 1659-1715 cm-1 and C-H strechings were found in 2800-3000 cm-1. In NMR spectra methyl and methine signals were found between 2.0-3.2 ppm and C-H signals (Ar-H) were found between 6.98-7.98 ppm. The physicochemical data and IC50 values of synthesized compounds are given in Table 1.
| Sr. No. |
Code |
Final Product |
M.P. (ºC) |
Yield (%) |
Rf Value* |
IC50 (µg/mL) |
| 1 |
RS-6 |
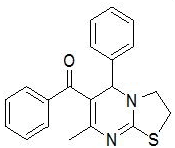 |
151-153 |
71 |
0.51 |
198±7.8 |
| 2 |
RS-7 |
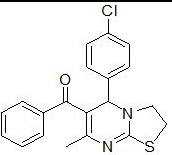 |
156-161 |
73 |
0.49 |
124±3.2 |
| 3 |
RS-8 |
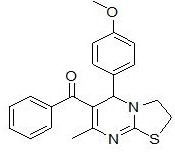 |
158-162 |
68 |
0.46 |
143±5.2 |
| 4 |
RS-9 |
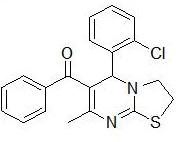 |
175-177 |
64 |
0.42 |
246±8.6 |
| 5 |
RS-10 |
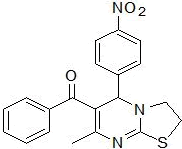 |
187-189 |
58 |
0.61 |
87±0.2 |
| 6 |
RS-11 |
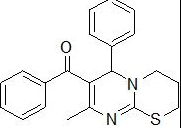 |
159-160 |
61 |
0.59 |
214±6.2 |
| 7 |
RS-12 |
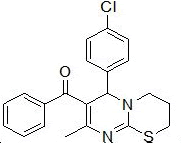 |
175-177 |
62 |
0.54 |
128±1.9 |
| 8 |
RS-13 |
 |
181-183 |
57 |
0.53 |
134±2.4 |
| 9 |
RS-14 |
 |
193-193 |
65 |
0.51 |
258±2.3 |
| 10 |
RS-15 |
 |
186-188 |
63 |
0.48 |
78±3.7 |
*Solvent system: Hexane: Ethyl acetate (6:4)
Table 1: Structural details and IC50 of RS6-15.
Cytotoxic Studies
All the newly synthesized compounds were evaluated against hepatic cancer cell line i.e., HepG2 in the animal tissue culture laboratory at ISFCP, Moga using MTT assay. Almost all the synthesized compounds RS (6-15) demonstrated moderate to biological activity against HepG2 cancer cell lines except RS-6 and RS-11.
In the first series of compounds RS-6 and RS-11, in which phenyl group is unsubstituted, showed less cytotoxic activity having (IC50=198 ± 7.8 μg/mL) and (IC50=214 ± 6.2 μg/mL), respectively. The compound RS-9 depicts very low cytotoxic activity (IC50=246 ± 8.6 μg/mL) followed by compounds RS-14 (IC50=258 ± 2.3 μg/mL) with 2-chlorophenyl substitution. The compounds RS-8 and RS-13comprising electron donating methoxy group at para position of phenyl ring showed moderate cytotoxic activity having (IC50=143 ± 5.2 μg/mL) and (IC50=134 ± 2.4 μg/mL), respectively. Substitution with chloro group on phenyl group improve cytotoxicity activity as in case of compounds RS-7 (IC50=124 ± 3.2 μg/mL) and RS-12 (IC50=128 ± 1.9 μg/mL). Compounds RS-10 and RS-15 found to be most potent compounds in above series with IC50 values (IC50=87 ± 0.2 μg/mL) and (IC50=78 ± 3.7 μg/mL), respectively because of presence of electron withdrawing nitro group on phenyl ring.
The SAR studies revealed that the electronic nature of substituents on phenyl group have pronounced effect on the biological activity of the compounds. The electron withdrawing groups like chloro (RS-7 and RS-12), nitro (RS-10 and RS-15) favour the cytotoxic potential of compounds. The electron donating group like methoxy (RS-8 and RS-13) have moderate effect on the biological activity. Also compound (RS-6 and RS-11) with no substitution substituents on phenyl group showed less cytotoxicity and compounds (RS-9 and RS-14) having 2- chlorophenyl substitution showed very less cytotoxicity. Therefore, compounds bearing electron withdrawing groups showed potent cytotoxic activity and the compounds with electron donating group exhibited minimum cytotoxicity.
On the other hand, cyclizations on the dihydropyrimidine ring have better effect on the cytotoxic activity. In the presence of strong electron donating group (methoxy) (RS-8 and RS-13) at para-position of phenyl ring having thiazolo and thiazine ring showed moderate cytotoxic activity. Furthermore, the compounds with electron withdrawing group (nitro) (RS-10 and RS-15), milder electron donating group (chlorine) (RS-7 and RS-12) at para-postion of phenyl ring having thiazolo and thiazine ring showed potent cytotoxic activity. The unsubstituted phenyl group showed less cytotoxic activity (RS-6 and RS-11) and compounds with 2-chlorophenyl substitution showed very less cytotoxic activity (RS-9 and RS-14). The graphical representation of the IC50 values of the newly synthesized compounds (RS6-15) is given in Figure 4.
Figure 4: Synthesis of 1-15.
Conclusion
All the compounds exhibit good cytotoxic activity against HepG2 cancer cell lines. Compound RS-10 and RS-15 were the most potent compounds having (IC50=87 ± 0.2 μg/mL) and (IC50=78 ± 3.7 μg/mL), respectively followed by compound RS-7 and RS-12 having (IC50=124 ± 3.2 μg/mL) and (IC50=128 ± 1.9 μg/mL), respectively. The SAR studies revealed that electron withdrawing groups like nitro and chloro enhance the biological activity of the compounds. On the other hand, electron donating group like methoxy decrease the biological activity. The unsubstituted compounds like RS-6 (IC50=198 ± 7.8 μg/mL) and RS-11 (IC50=214 ± 6.2 μg/mL) decrease the activity. It may be concluded that the electrons withdrawing and donating groups on the phenyl ringof the fused dihydropyrimidinone play a decisive role in the inhibition of Eg5 enzyme to reveal anticancer activity. Although the new compounds were found to have moderate to weak activity against cancer cell lines, the reported results are expected to put in toward deeper imminent into structure-activity relationship.
Acknowledgements
Authors express their thanks to Mr. Parveen Garg, Honourable Chairman, ISF College of Pharmacy for providing us necessary facilities for this research work.