Anila Mitre1, Blerta Laze2 and Merije Elezi1*
1Department of Mathematical Natural Sciences, University of Tirana, Tirana, Albania
2Department of Molecular Biology, Intermedica Clinic, Tirana, Albania
3Department of Food Technology and Nutrition, University of Tetovo, Tetovo, Northern Macedonia
- *Corresponding Author:
- Merije Elezi
Department of Mathematical Natural Sciences
University of Tirana
Tirana, Albania
E-mail: mas.shariq@gmail.com
Received Date: October 26, 2021; Accepted Date: November 09, 2021; Published Date: November 16, 2021
Citation: Mitre A, Laze B, Elezi M (2021) The Evaluation of Quantitative Hbsag Assay and HBV-DNA Assay in Chronic Hepatitis B Infection. J Clin
Gastroenterol Hepatol. Vol.5 No.3:3.
Keywords
Quantitative HbsAg; HBV DNA;
Chronic hepatitis B; Electrochemiluminescence
Introduction
Chronic Hepatitis B Virus (HBV) infection is a major global
health problem which infects approximately 350 million people
worldwide. The prolonged infection increases the risk of
developing liver failure, cirrhosis or Hepatocellular Carcinoma
(HCC) [1-3] leading to death.
Since 1960, the surface Hepatitis B Virus Antigen (HBsAg) has
been used as a hallmark of chronic HBV infection [4]. Recent
studies with quantitative assays have shown that serum HBsAg
level changes significantly during different phases of chronic
HBV infection by inversely correlating with HBV immune control:
The higher the control the lower the level of HbsAg [5]. These
results are in accordance with the hypothesis that the level of
HBsAg reflects the complex interaction between the virus and
the immune system and provides additional information on the
viral load, measured through HBV DNA level [6]. HBsAg
production changes quantitatively and qualitatively over time of
infection, and is dynamically regulated during different phases
of infection [7].
Several studies have shown a clear correlation between
serum levels of the surface hepatitis B antigen and circular DNA.
HBsAg is the main protein of the viral envelope, and serological
measurement of it has led to the diagnosis of hepatitis B
infection. If the infection occurs, HBsAg is the first detectable
immunological serum marker, which is usually present from
weeks to months before the onset of clinical symptoms. In case
of recovery of acute HBV infection, HBsAg is no longer
detectable in serum 6 months after its last recent onset [8,9].
If HBsAg persists for more than 6 months after acute hepatitis,
the presence of chronic hepatitis B infection should be assumed.
A Chronic hepatitis B virus patient with high levels of
aminotransferases, high viral load of HBV DNA and histological
abnormalities should be considered for therapy [10].
Various therapies are used to treat patients with HBV
infection. The current standard for controlling these therapies is the determination of HBV DNA. They should lower the viral load
as much as possible, theoretically below the lower limit of
detection of the PCR Real-Time method (10-15 IU/mL), to ensure
the virological suppression and prevention of complications.
However, the ideal end of therapy is the steady loss of HBsAg
with or without anti-HBs seroconversion.
However, recent studies suggest monitoring the interferon
alpha pegylated therapy by HBsAg and HBV DNA determination
[11-13]. Furthermore, the determination of HBsAg is likely to
predict stable virological response and loss of HBsAg. Further
studies are needed to confirm these findings and to determine
the importance of HbsAg determination in monitoring chronic
hepatitis B infection.
Chronic Hepatitis B Virus (HBV) infection is classified into four
phases (or types of immune response): immune-tolerant phase,
immune-clearance phase, chronic active phase, and chronic
inactive phase, which correlate with patients’ immune response
to HBV.
Also, the initial immune response and the subsequent initial
stage of chronic infection usually depend on the age at which
the patient has acquired HBV, the results of HBV therapy and the
genotype of HBV.
Phases of chronic HBV infection are not always considered as
static and patients who first develop the immunotolerant phase
usually progress to the immunoactive phase and then to the
chronic inactive carrier condition [14].
Methodology
A total of 200 samples (52 females and 148 males) from
different patients with chronic hepatitis B infection, without
starting the treatment, are involved in this study. These patients
were diagnosed with Chronic Hepatitis B at the Gastro
hepatology Clinic of Mother Teresa University Hospital Center in
Tirana, based on their disease history, ultrasound, biochemical
and immunological examinations. Serum samples were tested,
from March 2012 to April 2013, for quantitative HBsAg HbeAg,
anti-Hbe, anti-Hbc and anti-Hbc-IgM levels with Cobas 6000
Roche instrument and HBV DNA levels with Cobas Taq Man
instrument, at “Intermedica” Medical Clinic, in Tirana, Albania
determination.
Based on international protocols, patients were classified into
four groups:
Immune tolerant phase patients, who resulted positive for
HBeAg with normal ALT values.
Immune-clearance phase patients, who resulted positive and
negative for HBeAg with high values of ALT and HBV DNA.
Chronic active patients who resulted negative for HBeAg with
high values of quantitative HBsAg (4-5 log IU/ml) and moderate
growth ALT values.
Chronic inactive carrier who resulted negative for HBeAg with
normal values of ALT and HBV DNA.
Blood sampling and centrifugations were performed in two
different ways: gel tube and centrifugations for 10 min with 30,000 revolutions/min for quantitative HbsAg; EDTA tube and
centrifugations for 30 min with 1500 revolutions/min for HBV
DNA. The examinations for quantitative HbsAg were performed
within 2 hours of receiving the patient's blood. This
determination was performed with sandwich principle of
electrochemiluminescence technique, applied on Cobas 6000
instrument and the results are expressed in IU/mL. Samples
were stored at-20 degrees Celsius to BMM at the time of the
HBV DNA determination, which was performed with PCR-RT
technique applied on Cobas TAQ-MAN instrument and the
results, are expressed in IU/mL.
Furthermore, the results were statistically processed with
SPSS program (version 15.0; SPSS, Inc., Chicago, IL).
Results and Discussion
The evaluation of results showed high levels of HBV DNA [log 8
(IU/ml)] and quantitative HBsAg [approximately (log 5(IU/ml)]
at immune tolerant phase. Also, the first group (immune
tolerant phase patients) showed high levels of HBV DNA and
quantitative HBsAg, comparing it with the other three groups.
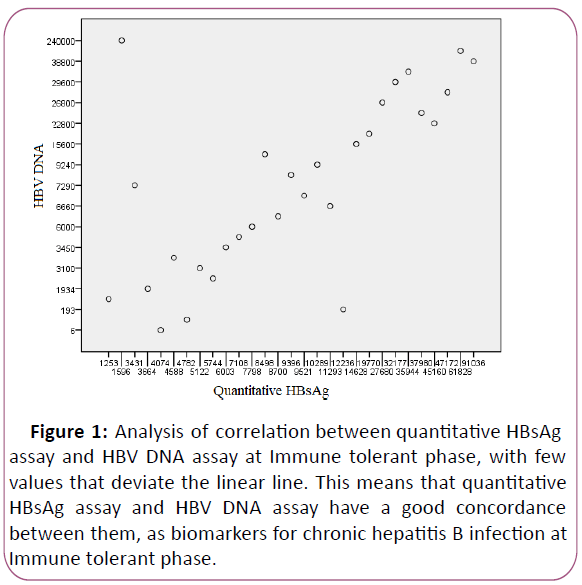
The correlation between quantitative HBsAg assay and HBV DNA
assay (Figure 1) was good at immune-tolerant phase [r=0.676
(p<0.01)].
Figure 1: Analysis of correlation between quantitative HBsAg
assay and HBV DNA assay at Immune tolerant phase, with few
values that deviate the linear line. This means that quantitative
HBsAg assay and HBV DNA assay have a good concordance
between them, as biomarkers for chronic hepatitis B infection at
Immune tolerant phase.
There was no correlation between HBV DNA levels and
HBeAg, ALT or AST levels. The quantitative HBsAg and HBV DNA
relationship was stable in patients that resulted positive for
HBeAg. This means a stable relationship between viral
replication and HBsAg levels (ccDNA levels) before the immune-clearance
phase.
The evaluation of results showed low levels of HBV DNA and
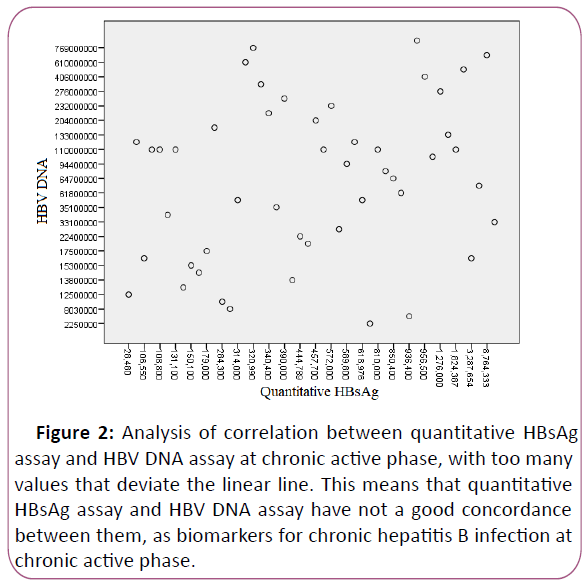
higher levels of quantitative HBsAg at chronic active phase.
There was no correlation between quantitative HBsAg assay and
HBV DNA assay (Figure 2), r=0.247.
Figure 2: Analysis of correlation between quantitative HBsAg
assay and HBV DNA assay at chronic active phase, with too many
values that deviate the linear line. This means that quantitative
HBsAg assay and HBV DNA assay have not a good concordance
between them, as biomarkers for chronic hepatitis B infection at
chronic active phase.
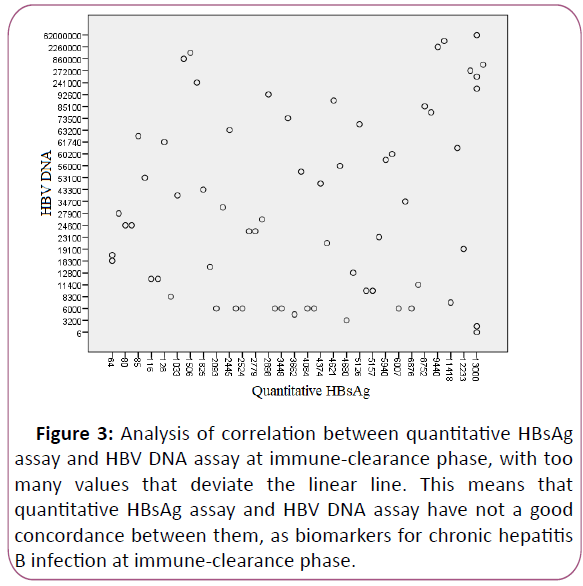
An immune control of viral replication at immune-clearance
phase, with increasing levels of HBsAg/HBV DNA report, is
caused by decreasing levels of HBV DNA and increasing levels of
quantitative HBsAg. The evaluation of results showed no
correlation between quantitative HBsAg assay and HBV DNA
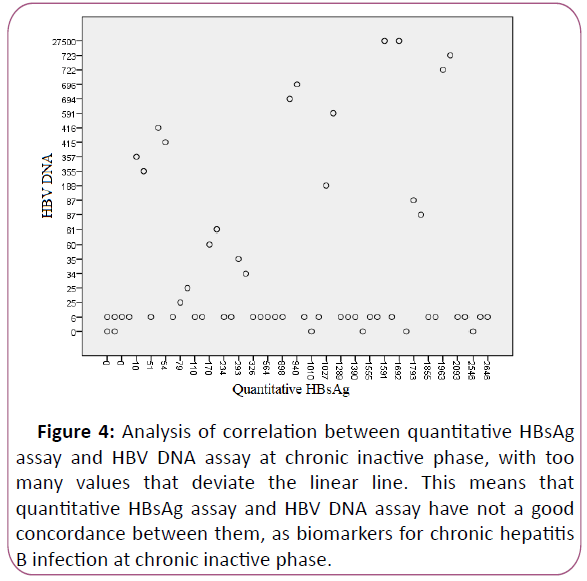
assay, r=0.120 (Figures 3 and 4).
Figure 3: Analysis of correlation between quantitative HBsAg
assay and HBV DNA assay at immune-clearance phase, with too
many values that deviate the linear line. This means that
quantitative HBsAg assay and HBV DNA assay have not a good
concordance between them, as biomarkers for chronic hepatitis
B infection at immune-clearance phase.
Figure 4: Analysis of correlation between quantitative HBsAg
assay and HBV DNA assay at chronic inactive phase, with too
many values that deviate the linear line. This means that
quantitative HBsAg assay and HBV DNA assay have not a good
concordance between them, as biomarkers for chronic hepatitis
B infection at chronic inactive phase.
The evaluation of results showed low levels of HBsAg and
normal levels of transaminases (ALT, AST) at chronic inactive
phase (negative values of HBeAg), and no correlation between
quantitative HBsAg assay and HBV DNA assay, r=0.22 (Figure 4).
These reflect the immune clearance tendency.
Furthermore, the evaluation of results showed that active
phase patients were younger than inactive phase patients. Also,
the number of infected males (145) resulted higher than
infected females (51). 33% of patients resulted positive for
HBeAg, and 67% of patients resulted negative for HBeAg. Also,
patients who resulted positive for HBsAg had higher levels of
HBV DNA than patients who resulted negative for HBeAg.
The results of this study are in concordance with some studies
conducted in Iran. This is thought to be related with the same
genotype through these populations [15]. On the other hand,
the results of this study are not in concordance with some other
studies conducted in Korea [16-21].
Conclusion
The evaluation of the results showed a different correlation
between quantitative HBsAg assay and HBV DNA assay at
different phases of chronic hepatitis B infection; at Immune-
Tolerant phase (IT), the correlation was good [r=0.676 (p<0.01)].
There was no correlation between these assays during the other
phases of infection.
In conclusion, there is a weak correlation between
quantitative HBsAg assay and HBV DNA assay. So, quantitative
HBsAg assay cannot substitute the HBV DNA assay (except the
immune-tolerant phase) and it cannot be used as the only
biomarker for chronic hepatitis B infection.
References
- Lee WM (1997) Hepatitis B virus infection. N Engl J Med 337:1733-1745.
- Wright TL, Lau JYN (1993) Clinical aspects of hepatitis B virus infection. Lancet 342:1340-1344.
- Hoofnagle JH, di Bisceglie AM (1997) The treatment of chronic viral hepatitis. N Engl J Med 336:347-356.
- Hoofnagle JH (1981) Type B hepatitis: Virology, serology and clinical course. Semin Liver Dis 1:7-14.
- Ji Lee H, Young Kim S, Min Lee S, Heo J, Hoi Kim H, et al. (2012) Hepatitis B Surface Antigen Quantitative Assay: Performance Evaluation and Correlation with Hepatitis B Virus DNA during 96 Weeks of Follow-up in Chronic Hepatitis B Patients. 32: 420-425.
- Scaglioni PP, Melegari M, Wands JR (1996) Recent advances in the molecular biology of hepatitis B virus. Baillieres Clin Gastroenterol 10:207-225.
- Lin LY, Wong VW, Zhou HJ, Chan HY, Gui HL, et al. (2010) Relationship between serum hepatitis B virus DNA and surface antigen with covalently closed circular DNA in HBeAg-negative patients. J Med Virol 82:1494-1500.
- Coursaget P, Yvonnet B, Bourdil C (1987) HBsAg positive reactivity in man not due to hepatitis B virus. Lancet 2:1354-1358.
- Shiels MT, Taswell HF, Czaja AJ (1987) frequency and significance of concurrent hepatitis B surface antigen and antibody in acute and chronic hepatitis B. Gastroenterol 93:675-680.
- European Association for the Study of the Liver (2019) EASL Clinical Practice Guidelines: Management of chronic hepatitis B. J Hepatol 50:227-242.
- Marcellin P, Bonino F, Lau GK (2009) Sustained response of hepatitis B e antigen-negative patients 3 years after treatment with peginterferon alpha-2a. Gastroenterol 136:2169-2179.
- Perillo R (2009) Hepatitis B surface antigen quantification as a current-day paradox: Obtaining the gold in the face of dimishing returns. Hepatol 49:4.
- Brunetto MR, Moriconi F, Bonino F (2009) Hepatitis B Virus Surface Antigen Levels: A Guide to Sustained Response to peginterferon alpha-2a in HBeAg-Negative Chronich Hepatitis B. Hepatol 49:1141-1150.
- Lok AS, McMahon BJ (2007) Chronic hepatitis B. Hepatol 45:507-539.
- Ganji A, Esmaeilzadeh A, Mokhtarifar A (2011) Correlation between HBsAg quantitative assay results and HBV DNA levels in chronic HBV Hepat Mon 11:342-345.
- Sonneveld MJ, Rijckborst V, Boucher CA, Zwang L, Beersma MF, et al. (2011) A comparison of two assays for quantification of Hepatitis B surface Antigen in patients with chronic hepatitis B. J Clin Virol 51:175-178.
- Ganem D, Prince AM (2004) Hepatitis B virus infection-natural history and clinical consequences. N Engl J Med 350:1118-1129.
- Marquardt O, Heermann KH, Seifer M, Gerlich WH (1987) Cell type specific expression of pre S 1 antigen and secretion of hepatitis B virus surface antigen. Brief Report. Arch Virol 96:249-256.
- Mühlbacher A, Weber B, Bürgisser P, Eiras A, Cabrera J, et al. (2008) Multicenter study of a new fully automated HBsAg screening assay with enhanced sensitivity for the detection of HBV mutants. Med Microbiol Immunol 197:55-64.
- Thomas HC, Lemon S, Zuckerman AJ (2005) Viral Hepatitis. In: Kann M, Gerlich WH, editors. Structure and molecular virology. 3rd ed. Oxford: Blackwell Publishing pp:149-180.
- Tong S, Kim KH, Chante C, Wands J, Li J (2005) Hepatitis B virus e antigen variants. Int J Med Sci 2:2-7.