Research Article - (2018) Volume 5, Issue 1
Panpan Huang, Jie Tan, Wenlin Gong, Zhenghua Zhang, Rong Zheng, Chaofan Zhou, Guoqing Zhang, Wenxiu Dai, Siyu Wu, Ying Zhang, Yong Huang, Sufang Zhou* and Yongxiang Zhao*
National Center for International Research of Biological Targeting Diagnosis and Therapy, Guangxi Key Laboratory of Biological Targeting Diagnosis and Therapy Research, Collaborative Innovation Center for Targeting Tumor Diagnosis and Therapy, Guangxi Medical University, Nanning, Guangxi 530021, China
Corresponding Authors:
Yongxiang Zhao
National Center for International Research of Biological Targeting Diagnosis and Therapy
Guangxi Key Laboratory of Biological Targeting Diagnosis and Therapy Research
Collaborative Innovation Center for Targeting Tumor Diagnosis and Therapy
Guangxi Medical University, Nanning, Guangxi 530021, China
Tel: +86-77-1531-7061
E-mail: yongxiang_zhao@126.com
Sufang Zhou
National Center for International Research of Biological Targeting Diagnosis and Therapy
Guangxi Key Laboratory of Biological Targeting Diagnosis and Therapy Research
Collaborative Innovation Center for Targeting Tumor Diagnosis and Therapy
Guangxi Medical University, Nanning, Guangxi 530021, China
Tel: +86-77-1531-7061
E-mail: zsf200000@163.com
Received Date: February 26, 2018; Accepted Date: March 26, 2018; Published Date: March 31, 2018
Citation: Huang P, Tan J, Gong W, Zhang Z, Zheng R, et al. (2018) Towards an Effective Management on the Teaching and Learning of Accounting in Secondary Schools: Case Report. Br J Res Vol. 5 No. 1: 40. doi: 10.21767/2394-3718.100040
Copyright: © 2018 Huang P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
A simple and rapid colorimetric method for determination of alpha-fetoprotein (AFP) has been developed here using specific aptamers and free gold nanoparticles (AuNPs). In the absence of AFP, the free aptamers prevent aggregation of AuNPs in a high salt environment, which cause the mixed solution remain red. Conversely, when there are existing AFP in colorimetric system, the aptamers is consumed to decrease, forming aptamer-AFP complex. AuNPs were aggregated due to losting the protection from aptamers in the high salt solution. The color of the solution turns to blue at this time. Subsequently, a color change of red-to-blue can be observed by the naked eye with the increasing of AFP. The linear response range for AFP was 10 to 2000 ng/ml and the detection limit was 5 ng/ml. This colorimetric biosenser does not require any modification, and its composition is cheap and easy to obtain. In addition, this proposed colorimetric method is also used in simulative hepatocellular carcinoma (HCC) serum samples and showed good feasibility, holding great potential for preliminary screening of serum of HCC in the future.
Keywords
AFP detection; Colorimetric assay; Aptamer; Gold nanoparticle; Hepatocellular carcinoma
Introduction
Alpha-fetoprotein (AFP), a member of the serum albumins family, is used as one of the significant protein tumour markers of hepatocellular carcinoma (HCC) for the screening and diagnosis [1-3]. At present, the Enzyme Linked Immunosorbent Assay (ELISA) based on combination of antigen and antibody is used to detect the level of AFP in serum. Furthermore, there are a lot of sensitive methods based on ELISA have been developed to detect the AFP [4-10]. These methods based on ELISA are designed to amplify their detection signals to improve their sensitivity. However, these systems generally include substrate, capture antibodies, label antibodies and so on, which can lead to high cost, long detection time and complex operation and result in these ELISA-based systemes difficult to apply in clinical laboratory analysis.
At present, the agents used to capture cancer markers consists not only of antibodies, but also of aptamers which have been increasingly used. Aptamers is a single-stranded DNA or RNA that is screened out using pre-selected target with SELEX (systematic evolution of ligands by exponential enrichment) techniques [11,12]. Meanwhile, aptamer is also called chemical antibody and act like antibody. In general, antibody is susceptible to temperature and pH and always undergo irreversible denaturation, while aptamer hardly possess these disadvantages and gradually replace antibody for tumor markers detection [13]. Aptamer exhibits some of the distinct advantages such as easier synthesis, better stability, lower cost compared to the antibodies [14,15]. Come to this point, aptamer is expected to be new capture tool for tumor marker. However, it is not all know exactly about the conformational changes and binding sites of aptamer when aptamer bind to its targeting molecule. So, it is difficult to design a labled aptamer which does not affect binding of target and aptamer [16]. In this case, unmodified aptamer is the most ideal capturing tool for targeting.
Colorimetric assay is especially attractive for its properties such as visible colour changes, simple operation and rapid reading [17,18]. These characteristics determine it particularly well suited for the rapid detection of the tumor markers. In colorimetric detection systems, to translate invisible signals into color changes is of great key. Nowadays, gold nanoparticles (AuNPs) have emerged as common colorimetric reporting probes for the detection of various molecules [19-23], which relies on its unique surface plasmon resonance (SPR) [24]. The citrate reduction method, one of the most popular methods, was used to synthesize AuNPs [25]. The raw materials for synthesizing AuNPs are not expensive and easy to obtain. In general, the newly synthesized AuNP solution is homogeneously dispersed and is red in color, while the change to blue in color comes from aggregated AuNPs due to the influence of external factors. AuNPs could be stabilized by ssDNA even in the salt condition, while the aggregation of AuNPs was induced by desorption of ssDNA with a few minutes [26-28]. Hence, AuNP is a cheap, simple and rapid colorimetric probe.
In this study, we developed a simple and rapid aptamer-based colorimetric biosensing detection method for AFP using AFP aptamer and unmodified gold nanoparticles. The AP273, a AFPbound ssDNA aptamer, was screened out using capillary electrophoresis (CE) SELEX technology [29] and used in this study. When the target molecule AFP is absent, free aptamers were absorbed onto the surface of AuNPs to prevent the aggregation, which made the color of reaction solution remain red. Then, free aptamers were consumed and decreased in amount when samples were detected. The AuNPs lose the protection from aptamers and tend to aggregation, which induced the color change from red to blue. This color change can be observed by the naked eye, and the accurate results were measured by UV-Vis spectrometer. In order to verify the feasibility and clinical applicability of this method, several simulative HCC serum samples were successfully analyzed and detected in a relatively short time and satisfactory results were obtained. This suggests that this method could be used in clinical large-scale rapid screening.
Materials and Methods
Reagents and chemicals
HAuCl4.4H2O was prochased from West Asia Chemical Industry Co., Ltd (Shandong, China). Sodium citrate and other chemical reagents were supplied by Tianjin bodi chemical Co., Ltd (Tianjin, China). Protein of AFP, GP73 and DKK1 all were bought in Sino Biological Inc (Beijing, China). Bovine serum albumin (BSA) was prochased from Solarbio (Beijing, China). The AP273 aptamer used in this study was synthesized and purified with PHLC by Sangon Biotech Co., Ltd (Shanghai, China). The sequence of the AFP aptamer called AP273 was Healthy serum samples were collected from department of clinical laboratory, the First Affiliated Hospital of Guangxi Medical University.
Instruments
The size of AuNPs was measured by transmission electron microscope (TEM, Hatichi, Japan). UV-Vis spectra data are collected with a Nanodrop 2000c (Thermo Scientific, USA).
Preparation of AuNPs
AuNPs were synthesized according to a reported reducing sodium citrate method [30]. In brief, a 20 ml solution of 1 mM HAuCl4 was heated to boiling. Then 2 ml solution of 38.8 mM sodium citrate was added to the above solution rapidly with steady stirring. The solution continued to be heated and the AuNPs was collected at the room temperature. Figure 1 showed the characterization of as-prepared AuNPs. The average size measured by TEM is about 13 nm (Figure 1a). The absorption spctra of synthesized AuNPs was measured and concentration of AuNPs was quantified in accordance with a molar extinction coefficient of 2.7 × 108 L mol-1 cm-1 at 520 nm (Figure 1b).
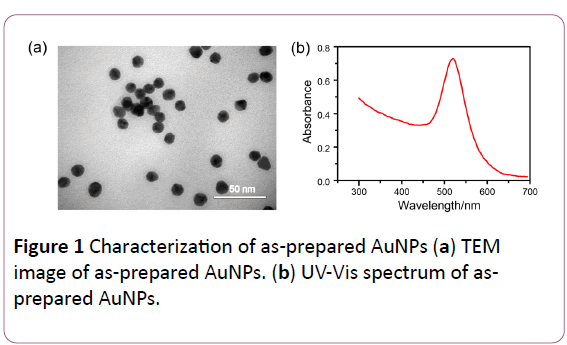
Figure 1: Characterization of as-prepared AuNPs (a) TEM image of as-prepared AuNPs. (b) UV-Vis spectrum of asprepared AuNPs.
Colorimetric detection of AFP
In order to establish aptamer-based unmodified gold nanoparticle probes for AFP detection, the colorimetric detection of AFP was described as follow: In brief, lyophilized aptamer was dissolved in 10 mM PBS buffer with 0.1 mM MgCl2. A 50 μl solution containing aptamer or containing aptamer and various concentrations of AFP were incubated at 4°C for 20 min. Next, 200 μl of AuNPs was added to the above solution and allowed to stay at room temperature for another 5 min. Then 50 μl of NaCl was added into the mixed solution, and UV-Vis spectra values were collected at the range of 300 to 700 nm after 5 min incubation.
Selectivity of AFP assay
To ensure the selectivity of this colorimetric method, different serum markers of HCC acted as interferential factors were tested. We prepared two kinds of serum markers of HCC and one compared protein. Concentrations of AFP, GP73 [31], DKK1 [32] and BSA were set to 1000 ng/ml. UV-Vis spectra values were also collected at the range of 300 to 700 nm.
Treatment and determination of serum samples
After obtaining the healthy serum from the hospital, the serum were placed at 4°C for 2 hours and then centrifuged at 3000 rpm for 10 min to obtain about 1 ml of supernatant. The separated serum is stored at -20°C until using. During the detection, a 50 μl solution containing aptamer and serum were mixed for the next experiment according to procedure above.
Results and Discussion
Colorimetric determination of AFP using AuNPs and specific aptamer
The AuNPs were synthesized according to reduction method. In our colorimetric system, as shown in Scheme 1, dissociative aptamers are adsorbed on the surface of AuNPs to prevent the aggregation between nanoparticles in NaCl environment [16,23,26]. This adsorption is mainly due to the non-covalent bond interaction between ssDNA and AuNPs. However, based on the interaction between aptamers and targets, aptamers bind with AFP tightly to form aptamer-target complex that do not prevent the aggregation of AuNPs. An obvious difference in color between red and blue was observed by naked eyes with the absence of AFP and presence of AFP, respectively.
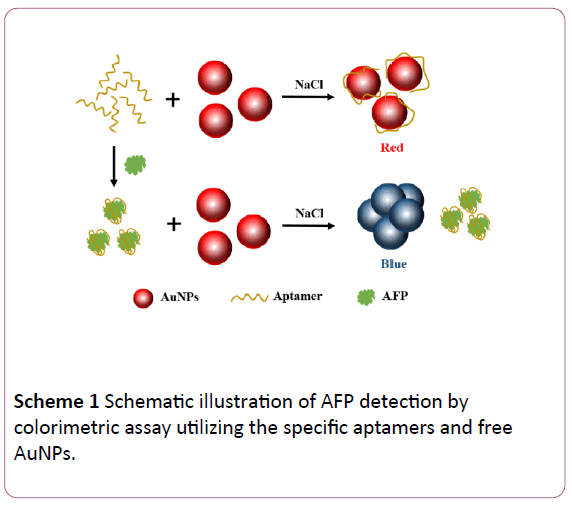
Scheme 1: Schematic illustration of AFP detection by colorimetric assay utilizing the specific aptamers and free AuNPs.
Feasibility and mechanism of detection for AFP
In order to ensure the feasibility for detection of AFP, UV–Vis spectra of the AuNPs under different experimental conditions were showed in the Figure 2a. Once the addition of NaCl (500 mM) to unmodified AuNPs, UV–Vis absorption range is widened and a new peak appeared at 650 nm. However, there is no change in AuNPs containing aptamer (500 nM) after the addition of salt. More importantly, the changes of absorption and peak at 650 nm will happen again when the AFP was added to the AuNPs solution containing aptamer and NaCl. Correspondingly, the color of the AuNPs solution under different experimental conditions was showed in the Figure 2b.
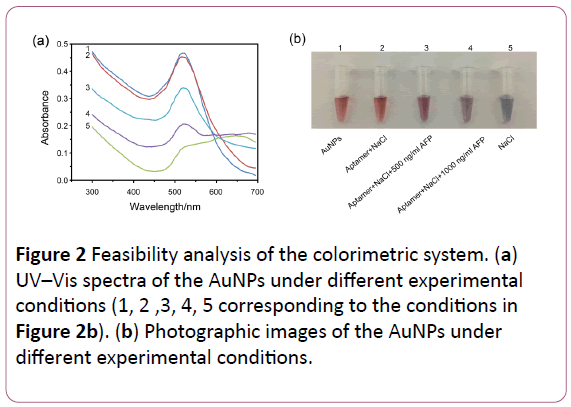
Figure 2: Feasibility analysis of the colorimetric system. (a) UV–Vis spectra of the AuNPs under different experimental conditions (1, 2 ,3, 4, 5 corresponding to the conditions in (b) Photographic images of the AuNPs under different experimental conditions.
The widening of absorption and appearance of peak at 650 nm were mainly due to the aggregation of AuNPs induced by NaCl. Aptamers can prevent the aggregation of AuNPs. Furthermore, the amount of AFP added to the detection system was positively correlated with the aggregation degree of AuNPs. These results demonstrated that this colorimetric probs can be used for the detection of AFP.
To display the changes of AuNPs morphology in the absence or presence of AFP, AuNPs were further characterized by TEM. The image in Figure 3a show uniform dispersion of AuNPs and provide powerful evidence of the protection from aptamer in high salt environment with the absence of AFP. In the presence of AFP, the aggregation of AuNPs was triggered by NaCl without the protection of free aptamer (Figure 3b). The TEM results were consistent with the mechanism shown in Scheme 1.

Figure 3: TEM images of the AuNPs mixed with AFP aptamer (500 nM) in the absence (a) or presence (b) of AFP (500 ng/ml) under 300 mM NaCl.
Optimization of key variables
In order to obtain optimal results, the key variables should be optimized. First, the aggregation of AuNPs was controlled by the concentration of NaCl in the reaction solution. It was found that low concentrations of NaCl could not cause color change, while the high concentrations of NaCl could make the color quick turns from red to blue. So, the optimal concentrations of NaCl should be selected to next experiments. Herin, 50 μl various concentrations (50, 100, 150, 200, 250, 300 mM) of NaCl and 200 μl AuNPs were mixed for 5 min to research the optimal concentration, which showed in Figure 4a, and the UV-Vis spectra collected from 300 to 700 nm. It was discovered that the maximal ratio of the absorbance of 520 and 650 nm (A650/ A520) was shown in Figure 4b when the concentration of NaCl reached to 250 mM. It is indicated that the 250 mM was maximum reaction signal of NaCl. So, 250 mM was the best choice of NaCl concentration to induce obvious color change whether the presence of AFP or not.
Next, the concentration of aptamer was deemed to be the another key variable that affected the sensitivity of the probes. The smaller the concentration of aptamer, the less the gold nanoparticles adsorbed on the gold surface, the easier the gold nanoparticles aggregate after addition of NaCl. Conversely, after the over-saturation of adsorption of aptamer on the surface of AuNPs, the solution still remain red even in the high NaCl environment, which reduce the sensitivity of probes. Hence, the appropriate concentration of aptamer should be optimized to allow the few free aptamers and strong protection for AuNPs from aggregation. The different concentrations (20, 40, 80, 160, 320, 640 nM) of aptamer were used in the solution and the UVVis spectra were also collected from 300 to 700 nm (Figure 4c). As is shown in Figure 4d, A650/A520 decreases as the concentration of aptamer increases. When the aptamer is increased to 320 nM, the ratio of A650/A520 tends to be stable. So, 320 nM was chosen as the perfect concentration because of the stable ratio of absorbance values.
Last, the reaction time of AFP and aptamer was optimized. In order to investigated the effect of incubation time between the aptamer and AFP during the detection, the adsorption was measured from 5 to 20 min at the 500 ng/ml of AFP Figure 4e. As is shown in Figure 4f, the A650/A520 in 5 min is smaller than other groups, while the A650/A520 in 10, 15 and 20 min are very close. These results demonstrated that 10 min is enough to form stable aptamer-AFP complex. Thus, incubation time of 10 min was chosen to use in the later experiments.
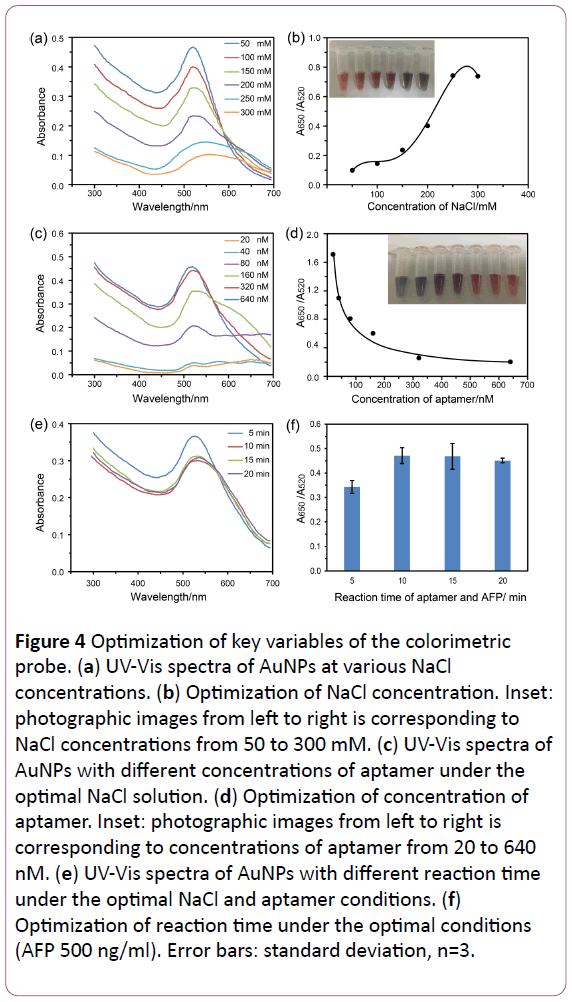
Figure 4: Optimization of key variables of the colorimetric probe. (a) UV-Vis spectra of AuNPs at various NaCl concentrations. (b) Optimization of NaCl concentration. Inset: photographic images from left to right is corresponding to NaCl concentrations from 50 to 300 mM. (c) UV-Vis spectra of AuNPs with different concentrations of aptamer under the optimal NaCl solution. (d) Optimization of concentration of aptamer. Inset: photographic images from left to right is corresponding to concentrations of aptamer from 20 to 640 nM. (e) UV-Vis spectra of AuNPs with different reaction time under the optimal NaCl and aptamer conditions. (f) Optimization of reaction time under the optimal conditions (AFP 500 ng/ml). Error bars: standard deviation, n=3.
Aptamer-based colorimetric assay for AFP
Under the optimal conditions, we developed the aptamerbased colorimetric assay using the unmodified gold nanoparticles for the detection of AFP. The absorbance of AuNPs with different concentrations of AFP were measured. Figure 5a showed the UV–Vis absorption spectra of colorimetric assay for AFP in different concentrations. As shown in Figure 5b, A650/ A520 continued to increase with the increment of AFP concentration, which lead to the color change from red to blue (Figure 5b, inset). It also showed good linear correlation to concentration of AFP at the range of 10 to 2000 ng/ml. The limit of detection was calculated to 5 ng/ml according to 3sblank criterion. In addition, the whole experiment takes less than half an hour, showing evidence of speediness.
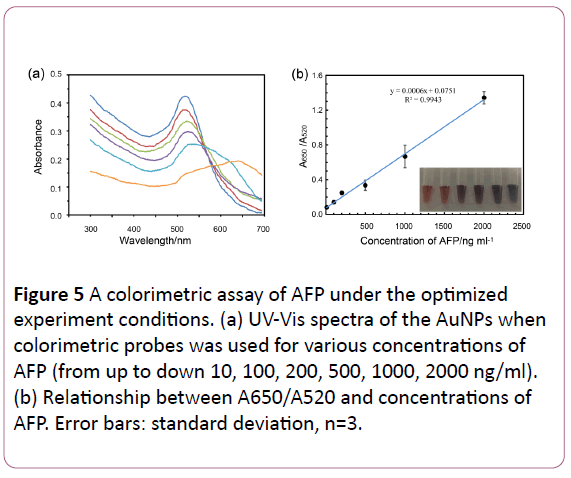
Figure 5: A colorimetric assay of AFP under the optimized experiment conditions. (a) UV-Vis spectra of the AuNPs when colorimetric probes was used for various concentrations of AFP (from up to down 10, 100, 200, 500, 1000, 2000 ng/ml). (b) Relationship between A650/A520 and concentrations of AFP. Error bars: standard deviation, n=3.
Selectivity for the detection
The selectivity of this probe was examined through replacing other markers of HCC such as GP73 and DKK1. As shown in Figure 6, only AFP showed a higher absorbance value in 650 nm and a lower absorbance value in 520 nm compared to others because of the aggregation of AuNPs. It was obvious that the depletion of free aptamers reduces the ability to prevent the aggregation of AuNPs. And soution turns to blue after adding the NaCl into AFP group (Figure 6, inset). These results clearly showed a high selectivity for the detection of AFP.
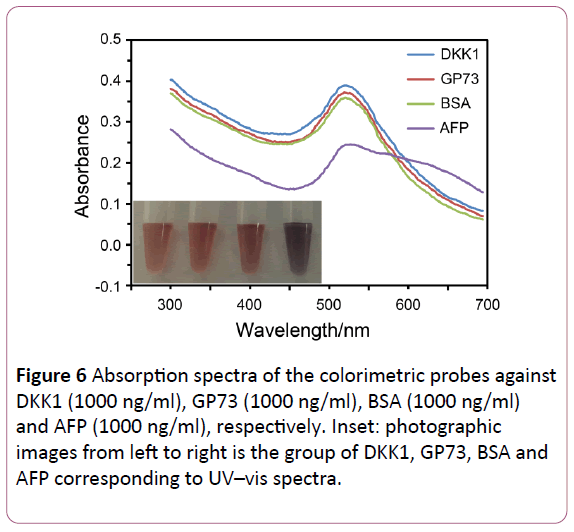
Figure 6: Absorption spectra of the colorimetric probes against DKK1 (1000 ng/ml), GP73 (1000 ng/ml), BSA (1000 ng/ml) and AFP (1000 ng/ml), respectively. Inset: photographic images from left to right is the group of DKK1, GP73, BSA and AFP corresponding to UV–vis spectra.
Application in analysis of simulative HCC serum
To further ensure the feasibility of this colorimetric assay in HCC serum, the detection of AFP in serum samples was implemented. As demonstrated in Figure 7, aptamer bind to the AFP in the diluted (1% diluted and 5% diluted) healthy human serum, which form the aptamer-AFP complex, inducing the decrease of free aptamer.
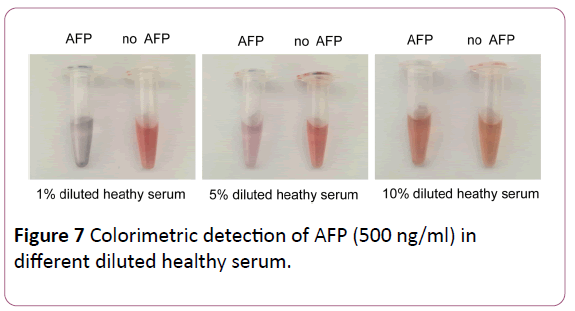
Figure 7: Colorimetric detection of AFP (500 ng/ml) in different diluted healthy serum.
The AuNPs lose the protection from free aptamer and was leaded to aggregration. In contrast, the diluted healthy serum without AFP did not affect the amount of free aptamers. AuNPs were protected by free aptamers and remained red. However, we found that there is no obvious change of color in the 10% diluted healthy human serum. This phenomenon exhibited that the complex organic compounds in serum can prevent the aggregation of AuNPs [20], which result in a high background signal. The results suggest that the colorimetric assay for AFP could be applied to the detection of serum samples but its boundedness shows that serum samples need to be treated to reduce background signal.
Conclusion
In summary, we have developed a simple and rapid colorimetric method for detection of AFP using specific aptamers and free gold nanoparticles. It could obtain quite sensitive results without expensive instruments and complicated operating procedures. The result of the obvious color change is visible to the naked eye, and the whole process takes only less than half an hour. An applicable linear range and a good detection limit were determined to be 10 to 2000 ng/ml and 5 ng/ml respectively, which are obtained in the case that the concentration of salt, the amount of aptamer and the time of binding of aptamer and AFP are optimal. More importantly, this colorimetric assay has been applied to the detection of AFP in diluted serum. An obvious change in color from red to blue due to the presence of AFP can be observed in the 1% and 5% diluted serum by the naked eye. This promising detection method for AFP shows a good potential for rapid diagnosis of HCC, even for the large-scale rapid preliminary screening in developing areas of medicine.
Acknowledgment
This work is supported by the Key Project of National Natural Scientific Foundation of China (No. 81430055), the Program for Changjiang Scholars and Innovative Research Team in University of Ministry of Education of China (No. IRT_15R13).
Abbreviations
AFP, alpha-fetoprotein; AuNPs, gold nanoparticles; HCC, hepatocellular carcinoma; ELISA, enzyme linked immunosorbent assay; SELEX, systematic evolution of ligands by exponential enrichment; SPR, surface plasmon resonance; GP73, golgiprotein 73; DKK1, dickkopf-1; BSA, bovine serum albumin; TEM, transmission electron microscope.