Research Article - (2011) Volume 1, Issue 2
Department of Chemistry, Industrial Chemistry Section, School of Physical Sciences, University of Cape Coast, Cape Coast, Ghana
The neem plant (Azadirachta indica) was used in this research to ascertain its effectiveness in controlling insects and pests. The leaves of the plant were applied. The neem plant has an active insecticidal ingredient, Azadirachtin (C35 H44 O16). The active ingredient in the leaf was extracted for the toxicological analysis in rabbits. The extracts were daily administered orally by gavage to male and female rabbits for two weeks at dose levels of 5 and 24mg/kg, respectively. Irrespective of dose/sex, effects on serum biochemistry parameters was not significant (P> 0.05) but the ALT in males was significantly higher than in females. All rabbits in the high-dose groups, irrespective of sex, showed significant increase (P > 0.05) in haematological parameters. RBC and PCV increased significantly in males than in females group. Mild splenomegaly was common to carcasses of all rabbits. Histopatholgy also revealed mild cellular infiltration of the hepatocytes, for both genders, there was no-observed-adverse-effect level for the neem leaved extracts based on this subacute study in rabbits was considered to be 2.5 mg/kg/day. Hence, the toxicological analysis conducted on the rabbits showed there was no mortality or clinical signs among the treatment groups and control. However, there was however increase in body weight gain.
Azadirachta indica, Azadirachtin, Insecticide, Phytochemicals, Metabolite, Aspartate Amino Transferase, Alannine Amino Tranferase, Histopathology, Toxicity, Alkaline Phosphatase.
In developing countries, losses of crops do happen due to pest, plant disease and competition from weeds is great. In households, pest and insects such as mosquitoes, cockroaches, mice etc pose risks such as; the destruction of furniture, clothing and also contribute to the dissemination of various diseases, most especially malaria.
The neem tree is drought resistant and thrives normally in areas with sub-arid to sub-humid conditions, with an annual rainfall between 400-1200mm. It is a life giving tree, especially for the dry coastal, southern districts. It has been observed that the various medicinal values of neem tree are its constituent of phytochemicals present. The seed kernels of neem yield about 90% of a fixed oil comprised primarily of glycerides. The yellow, bitter oil has a garlic-like odor and contains approximately 2% of bitter principles including nimbidin, nimbrin, nimbinin, nimbidol and other related minor limonoid triterpense [1]. All parts of the tree yield beta-sitosterol. Azdirachtin is the most complex secondary metabolite present in the plant.
The neem is a bitter tonic herb that reduces inflammation and clears toxins, while promoting healing and improving all body functions. Apart from this, it destroys a wide range of parasitic organisms and is also insecticidal and spermicidal [2]. There are other several comprehensive toxicological studies on some natural extract [3-8].
The acute oral toxicity in rats fed with technical grade azadirachtin ranged from 3,540 mg/kg to greater than 5000mg/kg, the highest dose tested when administered undiluted to albino rats. The single dose oral toxicity LD50 of the formulated product Azatin-EC fed to rats was 4,241mg/kg; considered practically non-toxic. This acute inhalation toxicity study in rats exposed to technical grade azadirachtin showed that the acute inhalation LD50 is greater than 2.41mg/L per animal, the highest dose tested. Although this figure is below the 5.0mg/L limit test dose for an acute inhalation study, the reported concentration was the maximum dose possible under the test conditions.
A 90 day oral toxicity study in rats fed with levels of 500, 2500, and 10000 ppm of azadirachtin showed no signs of overt systematic toxicity at any dose level after 90 days of feeding. Mean body weight was significantly decreased in the 10000ppm in males and females at weeks 3 and 4, respectively. This persisted for the duration of 90 days feeding period [9].
Work on technical azadirachtin was evaluated by [10] for the potential to cause gene mutations in the S.typhimurium strains at any dose (5, 50, 500, 5000 mg/plate) with or without s-9 activation. The Ames test was negative with or without metabolic activation for the formulated product.
Sampling
The research was conducted in the University of Cape Coast, Cape Coast, Ghana, West Africa. The toxicological analysis was carried out at the laboratories of the Accra Regional Veterinary Clinic, Labadi-Accra.
The samples were obtained from Apewosika, Kokoado and Amamoma; communities around the University of Cape Coast campus. The plant was identified according to the description by Felter and Lloyd (1998) and later authenticated by comparing with voucher specimens at the herbarium section of the University of Cape Coast.
All the apparatus were cleaned very well to remove as much dirt as possible. A quantity of the fresh leaves was pounded using a mortar and pestle and then blended with a blender to obtain a homogeneous mixture. The fresh blended leaves were used for the extraction.
Extraction of the neem leaves
About 1000g of homogenous mixture of fresh neem leaves blended was transferred into a cleaned container. 5.00 litres of distilled water was added to enhance the extraction. It stood for 24 hours in the refrigerator to allow extraction. Sieving was done using a semi-permeable white cloth, squeezing to extract all the liquid from the leaves chaff. It was then further allowed to settle for some time and the clear extract on top was decanted carefully and filtered using whatman filter paper to obtain the crude insecticide.
Preparation of stock solution
Two concentrations of the extract was prepared; 1mg/ml and 5mg/ml. The dried crude insecticide was weighed and dissolved in the corresponding amount of distilled water to make up to the desired volume of each concentration. The stock solution was stored in the refrigerator.
Groupings for the experimental animals
The rabbits selected for the toxicological analysis were grouped into five of two each and labeled MN1, MN2, FN1, FN2 AND CN. M denotes male, F for female, N for neem, C for control while 1 and 2 are the first and second duplicate samples respectively.
Doses Administered
The administration of the crude extract was carried out in the premises of a young rabbit farmer in Kaneshie with residential address B565/7, Asafoatse Amar St. MN1 and FN1 received a higher dose of the extract (5mg/kg body weight) while the groups MN1 FN2 received a lower dose of the extract ( 2.5 mg/kg body weight). No extract was administered to the control group. The experimental rabbits only were daily administered with regular doses of the extract by gavage. The various weights were used to determine the respective volumes of the extract to be administered using equation 1:
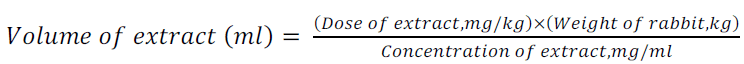 (1)
(1)
Collection of blood samples
Collection of blood samples was carried out on day 1 before the extract administration and a day after the administration of the extract. Blood samples were collected from each of the rabbits in the test and the control group using the method described by the Canadian Council on Animal Care, CCAC (1984) [11]. Marginal ear vein puncture was used to obtain venous blood samples. The hair directly over the vein was shaved and 70% alcohol applied to clean and sterilize the area. To distend the vein, the venous return was occluded by digital pressure on the marginal ear vein.
Haematological Studies
Blood samples collected for hematological studies were analyzed for packed cell volume (PCV), Hemoglobin concentration, White blood cell count (WBC), Platelet count, Differential Leucocytes count and Blood picture.
Determination of PCV (Haematocrit)
The blood samples were shaken gently by repeated inversions to ensure it was well mixed and uniform. It was then allowed to flow into a capillary tube by capillary attraction until it was ¾ full. The tube was sealed with plastacin and then centrifuged at a speed of 10,000 RPM for 5 minutes using the haematocrit centrifuge. The packed cell volume was then read in percentage with the aid of haematocrit reader [12].
Determination of Haemoglobin Concentration (Hb)
The spectrometer was allowed to warm up by switching it on for some time. Three test tubes were labeled as blank, standard and test. 5ml of Drabkins solution was pipette into all the tubes. The blood sample was mixed thoroughly, followed by the pipetting of 0.020 ml of distilled water and the blood sample into the labeled test tubes. The mixture was mixed further by inverting the tubes several times. The tubes with their contents were incubated at room temperature for complete conversion.
Measurement was then done by recording the readings of the absorbance spectrophotometrically against the reagent blank at 540nm wavelength. The Hb content was then calculated by the equation 2 below:
 (2)
(2)
Determination of white blood cell count (WBC)
Following thorough mixing of blood, 0.02ml of it was pipette into 0.38ml of Turk’s solution. The blood was further mixed and allowed to be converted in two minutes [12].
Cover slip was fixed onto an improved Neubeaur counting chamber, ensuring that the Newton’s rings were seen. The chamber was then filled by means of a Pasteur pipette and allowed to settle for 5 mins. Number of cells was counted in each of the sixteen boxes of the four large corners square under the low power (x 10 objective) microscope. The total WBC was calculated as follows by equation 3:
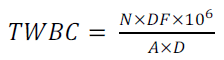 (3)
(3)
Where
N = Number of cells counted
DF = Dilution factor
106 = Converting factor into cells per litre (l)
A = Area of chamber
D = Depth of chamber
Differential Leukocyte Count and Blood Picture
The blood sample was mixed by inverting it repeatedly. A thin blood film was made on a clean grease free slide. The film was allowed to air dry and thereafter, over flowed with Leishman’s stain and allowed to fix for about 2 minutes.
The Leishman’s stain on the film was diluted with 2mls of buffered distilled water (pH 6.8) and allowed for 8 minutes to stain. Washing and differentiation was then done on the film by using distilled water followed by cleaning the back of the slide and air drying.
The prepared slide was then viewed under the microscope using x100 oil immersion objective lens. White cells were observed and recorded in percentage.
Biochemical Assay
The sera samples from various groups were analyzed for serum enzymes activities; Aspartate Amino Transferase (AST), Alanine Amino Tranferase (ALT), and Alkaline Phosphatase (ALP) were analysed [13].
Determination of Aspartate Amino Transferase (AST)
The method of Reithman and Frankel of 1957 was used. 0.5ml of AST substrate was pipette into four test tubes labeled Test, Test blank, STD and STD blank respectively and incubated at 37°c for 5 minutes. 0.2ml of serum was then added to Test, 0.2ml of Pyruvate standard and distilled water were added to STD and STD blank respectively, it was mixed and incubated at 37°c for 30 minutes. 0.5ml of 2,4-DNPH was then added into all test tubes; 0.2ml of serum was added into test blank only and left at room temperature for 30minutes. The reaction was stopped by the addition of 0.5ml of 0.4ml NaOH to all the test tubes was mixed and kept at room temperature for 5 minutes and absorbance was read spectrophotometrically at 510nm. The instrument was zero using the following formulae:
The AST value was calculated from the following equations 4 and 5:
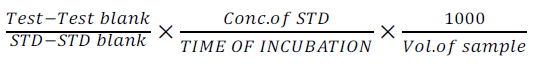 (4)
(4)
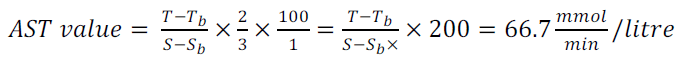 (5)
(5)
Determination of Alanine Amino Transferase (ALT)
The method of Reithman and Frankel of 1957 was also employed here [14].
The procedure is same as for AST except for using ALT substrate and 0.1ml of serum. Absorbance was read at 510nm. The instrument was zero using distilled water. ALT activity was calculated from the formula as above.
Determination of alkaline phosphate (ALP)
The alkaline phosphate was assayed according to the method of King Armstrong with slight modification 1964.
1ml of Alkaline buffer was added in 4 test tubes labeled T (Test), TB (Test blank), STD (standard) and STDB (Standard blank), also 1 ml of phenyl substrate was added to the test tubes labeled test and test blank, 0.1ml phenol standard and distilled water was added into STD and STDB respectively, 0.8ml 0.4N NaOH was then added to test tube labeled TB, STD and STDB it was mixed and incubated at 37 °C for 5 minutes. 0.1ml of serum was then added into test and test blank respectively and was incubated at 37 °C for 15minutes, 0.8ml of 0.4N NaOH was added to test tube labeled test followed by the addition of 1.2ml of 0.5M NaHCO3 all test tubes respectively, 1 ml of 4- amino antipyrine and potassium ferriccyanide was added successively into all test tubes was mixed and read immediately at 510nm.
ALT activity is calculated from equation 6 below:
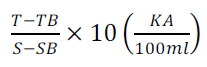 (6)
(6)
The value was converted to international Unit by multiplying with 7.1
Serum albumin and total protein analysis
Determination of total Protein
0.1ml of sample/ protein standard/ distilled water was mixed with 5ml of burette solution in test tube label test/standard and blank. It was mixed incubated at 37°C for 10 minutes which was the absorbance was read spectrometrically at 540nm by this relation.
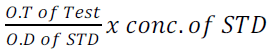 (7)
(7)
Concentration of the STD was 60g/is.
Determination of Serum Albumin
0.1ml of serum was added to 5ml of BCG; it was mixed together and incubated at 37°c for 10minutes. This was read at 625nm while the instrument was set to zero with (BCG) blank.
The level of albumin was calculated from the equation 8 as follows:
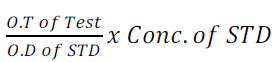 (8)
(8)
Concentration of the STD was 40g/is.
Urea and creatinine estimation
Determination of Serum Urea Level
In determining urea levels, 5ml of distilled water was place into each of 11test tubes, 10 for the test (sample) and 1 for the standard. 50μl of each corresponding serum was then added into each of the 10 tubes for the test, a corresponding 50μl of the standard solution added into the tube for the standard only. Contents of each tube were then mixed thoroughly. Next, 0.5 ml each of the diluted sera and standard were pipette into fresh tubes respectively, making a 1 in 10 serial dilution. Also a new tube was included for the blank. Into this new tube was placed 0.5 ml of distilled water. Therefore, 1.0 ml of the mixed acid reagent was added into all 12 tubes (10 for the diluted test and 1 for standard and the last 1 for the blank). Also 1.0 ml of the colour reagent was added into all 12 tubes. Content of each tube was mixed thoroughly and plugged with cotton wool. All the rag bearing the tubes was placed into boiling water for 30 minutes in dark, after which it was placed in cold water to cool but still in the dark. Finally the absorbance of the content of each tube was read at wavelength 520nm and recorded. Water blank was used to zero the spectrophotometer [15].
Determination of Creatinine Level
In the same manner, 1.5ml of distilled water is placed into each of the 10 tubes for the test, and 2.0 ml into the tube for the blank. Next, 0.5 ml or 50μl of serum was added into the tubes for the test. 50μl of 10% sodium tungstate, followed by 50μl of 2/3 N H2 SO4 was added into each of the tubes, mixed and centrifuged for 10 minutes. Next, 1.5 of the supernatant from each tube was transferred into fresh tubes. One more tube was included for the standard, and into this tube, 1.5 ml of distilled water followed by 1.5 ml of the standard solution was placed. Thereafter, into all 12 tubes was added 50μl of 0.75 N NaOH, followed by 50μl of picric acid. Each tube was then mixed well and let stand for 15 minutes, and absorbance read at 500nm and recorded. Water blank was equally used to zero the spectrophotometer.
Post Mortem Examination
These examinations involved both gross and microscopic examination of tissues.
Necropsy
Each rabbit was placed on the postmortem table in a dorso-ventral position and a midline incision was made through the skin from the mandible to the anus. The medial attachments of the limbs were severed and the abdominal wall reflected. The thorax was exposed and the ribs cut fairly high on both sides using a pair of scissors. All the major organs in the oral cavity, pharynx, thorax and abdomen were then examined insitu noting their relationship to one another. The crop which includes the tongue and thoracic organs was excised and laid on the postmortem tray. In the same way, the abdominal organs were excised but the urogential organs were left in the abdomen. This was followed by individual inspection of all the organs in position, shape, colour and consistency. Blocks of tissues including the lungs, liver, heart, intestines, kidneys and the brain were taken and preserved in 10% buffered formalin for histopathology [16].
Histopathology
The method of Baker and Stevenson (1957) was applied;
Statistical Analysis
Data were analyzed statistically using students’t-test. The results were expressed at the mean± S.D. the significance of the differences between control and the test groups were determined by the students’t-test and the values of P<0.05 were taken to be statistical significant (Parker, 1979). The stat graphics software was employed in the analysis of the data.
Toxicological Analysis
Clinical signs: During the period of the administration of the extract, the animals were inactive and dull. Tables 1 to 8 give a summary of Histopathology results of the animals after and before extract administration.
In the toxicological analysis, the rabbits used were administered with the neem extracts from low to high doses to test for the level of toxicity on an acute basis. Clinical signs of toxicity and various tests or parameters were carried out before and after the administration of the extract to authenticate either the effect or non- effectiveness of the extracts.
Although, there was progressive increase in body weight in both the test and control animals, during the entire duration of the administration of the extract, there was no observed sign of toxicity in both groups. The effect of the bitter nature of the extract was seen when the animals became dull and lost appetite.
The results of the haematology revealed a mild leucocytosis in both the test animals and the control animals after the two weeks of the administration as compared to the pretest values. This is an indication of the likely presence of a mild infection. However, the values still were within the normal reference range for rabbits and there was no significant difference between the control and the test animals (P> 0.05). Increases also RBC, Hb, and PCV of both test and control rabbits indicates there was no anaemia and blood production was not affected. Thus the crude extract of the neem leaves had no acute toxic effect on haematological parameters.
The results of the biochemical assay of ALT, AST, ALP, protein, urea, and creatinine showed that there was no significant difference between the test and control animals after the two weeks of the administration of the extract. This means that the crude aqueous extract of the neem leaves had no significant changes in the biochemical parameters assayed in the test.
In addition, the gross and histopathological lesions observed did not reveal any significant pathology to any of the organs as a result of the crude extract administered. The pulmonary congestion observed was common to both test and control animals. It may have been hypostatic congestion due to the time that lapsed between death and necropsy as the observation of pulmonary congestion increased in rabbits that were opened up afterwards. Also, histopathology did not reveal any significant lesion in the heart. Lesions in the brain were observed both in the test and the control animals indicating that it was not as a result of the extract administered.
No sign of observable toxicity was detected during the experimental period. Rabbits treated with various doses of the crude extracts of neem leaves for 14 days had progressive weight gain.
In the same study after 14 days dosage of the extract, there were no significant changes in ALT, AST, and ALP activities in the serum samples. There were also no significant changes in serum albumin and total protein concentrations.
The results show that the systems of the animals were able to detoxify the toxic material and excrete it without allowing its bioaccumulation which could lead to toxicoses.
The research has shown that insecticides made from natural botanical sources are less toxic as compared to those from the synthetic origin hence it is safer to use insecticides prepared from natural raw materials since they have little or no adverse effects on human health.
However, the results obtained from the various tests carried out, the No Observed Adverse Effect Level (NOAEL) of crude aqueous extract of neem leaves in rabbits was put at 5mg/kg body weight irrespective of sex.
The NOAEL of the crude aqueous extract of neem leaves can be applied in subsequent long term studies e.g. chronic toxicity study to determine whether it can induce any chronic or carcinogenic changes.
Moreover, studies can be carried out to test the efficacy of the leaves at this dose level in the treatment of various ailments. This will aid in standardization of local pharmaceutical preparations derived from neem leaves.