Research Article - (2023) Volume 14, Issue 9
Received: 01-May-2023, Manuscript No. AASRFC-23-16338; Editor assigned: 04-May-2023, Pre QC No. AASRFC-23-16338 (PQ); Reviewed: 18-May-2023, QC No. AASRFC-23-16338; Revised: 03-Jul-2023, Manuscript No. AASRFC-23-16338 (R); Published: 10-Jul-2023
The aim of the present study was to produce biodiesel from low cost chicken feather meal. The biodiesel was produced through transesterification process using potassium hydroxide as catalyst by single variable at a time approach. The obtained biodiesel was characterized by the assessment of quality according to ASTM D6751 standard and identification of functional groups by FT-IR analysis. The recovery of chicken fat was found to be 520 g from 6 kg of chicken feather meal and the yield of chicken fat was found between 8%-9%. The maximum yield of biodiesel production (74.4%) was exhibited at various optimized parameters such as, reaction time of 120 minutes, reaction temperature of 60°C, catalyst concentration of 1% w/v and methanol fat molar ratio of 6:1. The physiochemical properties of obtained biodiesel met ASTM D6751 Std criteria. Subsequently, the various functional groups of biodiesel were confirmed by Fourier-Transform Infrared Spectroscopy (FTIR) analysis. The results stated that the fat extracted from chicken feather meal consists of processed chicken feathers and it seems to be a low cost feed stock and could be used to produce biodiesel to meet the current demand of fuel as well as minimizes environmental pollution.
Chicken feather meal; Trans-esterification; Fatty acid methyl ester; ASTM D6751; Fourier transform infrared spectroscopy
Primarily, biodiesel is a fuel constituting of mono-alkyl esters of long chain fatty acids derived from renewable feed stocks such as vegetable oils or animal fats [1-3]. Day by day biodiesel is getting more attractive attention due to non-toxic, biodegradable, renewable, high flash point, eminent reduction in green house emission profile and eco-friendly fuel characteristic features [4-7]. It is predominant, known fact that the cetane number of biodiesel is larger than the diesel fuel. This constitutes almost no sulphur, no aromatics and does not contribute to greenhouse gases because of their closed carbon cycle [8-10]. However, the most important disadvantage is the high cost of biodiesel for its commercialization (reachability to its end users). Therefore, low cost effective feed stocks are most required at this instant. Chicken fat shown as a promising feed stock for biodiesel production in a cost basis comparing to high grade vegetable oils [11-14]. It is extracted from feather meal which is obtained from chicken wastes chicken feathers, offal, blood and trims after rendering process. Feather meal constitutes significant amount of chicken fat. The fat content of the feather meal lies between the range of 2% to 12% and it depends upon the kind of feathers used [15,16].
Globally, the consumption of chicken as a food source by human being is found to be very high. Hence, the poultry farming wastes could emerge in to major solid wastes (environmental pollutants). The poultry industries are at the door of extinct to face the huge emerging challenges in disposal of the accumulated waste [17-19]. It is significant and a social urge for the poultry industries to facilitate the disposal methods to mitigate their threat on the environment. The predominant poultry wastes as like poultry feathers, offal and litter have diverse field applications in industrial sector. The useful by product conversions like feather meal, biodiesel, biodegradable plastic and fertilizer which is obtained from the poultry feathers through conversion [20]. The utilization of feather fat for biodiesel production is a best suitable alternative to recycle these wastes. Therefore, the current study is intended to investigate the simple, low cost production of biodiesel from chicken feather meal by transesterification process, further, emphasize on physiochemical evaluation and optimization of biodiesel from poultry waste.
Sample Collection
Commercially available chicken feather meal sample was collected from poultry farm of Palladam, Tamil Nadu, and India. Sample was collected in sterile plastic bags and it was carried to the laboratory and the samples were stored.
Extraction and Separation of Fat from Chicken Feather Meal
The 75 g of chicken feather meal sample was stirred continuously in 1000 ml of distilled water at 90℃ for 45 minutes on magnetic stirrer with hot plate and cooled to room temperature. The fat floated on the surface of water layer was separated through centrifugation at 10000 rpm for 30 min. The bottom water layer was discarded and the top layer of fat was collected.
Saponification and Trans-Esterification of Fat of Chicken Feather Meal
The collected fat that contain 10 ml of 1% alkali solution of KOH was centrifuged at 1000 rpm for 20 min to remove Free Fatty Acid (FFA) in the form of soap. The saponified fat was heated at 50℃ for 20 min to remove traces of water. The saponified fat was subjected to trans-esterification process in a 250 ml conical flask which was suspended in 75 g of fat, molar ratio of methanol to fat of 5:1 and catalyst (KOH) concentration of 0.5%. The flask was then incubated at 30℃ for 90 min in an orbital shaker maintained at 180 rpm.
One Factor at a Time Experiment
In this conventional method, one independent variable is studied while keeping all the other factors at a constant level. The effect of temperature for biodiesel production was carried out at five different temperatures ranging from 30℃ to 70℃ at an interval of 10℃, respectively. The effect of incubation time on yield of FAME was determined by varying the reaction time (30, 60, 90, 120 and 150 min). The influence of catalyst on yield of FAME was carried out by supplying various concentration of KOH at 0.5% interval in the range of 0.5%-2.5% by weight. The effect of molar ratio of methanol to fat for the production of biodiesel was carried out at five different molar ratios of methanol to fat from 4:1 to 8:1 ratio respectively.
Separation of Biodiesel from Glycerol
The obtained reaction mixture after trans-esterification process was transferred to a separating funnel and kept overnight. The glycerol layer settled at the bottom was decanted and the top biodiesel layer was collected. The biodiesel was washed with warm water (40℃ to 45℃) and also with acidified water (20 ml of 0.1 N HCL solution) to remove excess amount of methanol and catalyst. After washing, the biodiesel was subjected to heating at 80°C through heating mantle to evaporate excess amount of methanol and water and filtered. The yield of biodiesel was calculated using the formula:
Yield percent=(weight of FAME (gm)/weight of fat from chicken feather meal (gm)) × 100
Physiochemical Characterization of Biodiesel Fuel
The physiochemical parameters such as, acid value, kinematic viscosity, specific gravity, pH, water and sediment, methanol test, emulsification and visibility were assessed as per American Society of Testing and Material (ASTM D6751, 2004).
FT-IR Analysis of Biodiesel
The FTIR were recorded in the mid-IR region 4000-400 cm-1 at room resolution 4 cm-1 with scanning using the functional group present in the purified biodiesel was identified as per IR spectral data recorded by KBr pellet (sandwich) sampling technique using Nicolet b6700 FT-IR spectrophotometer.
Extraction of Biodiesel
The amount of fat extracted from chicken feather meal sample that was found from 124.80 g to 127.25 g per kg of chicken feather meal. Further, from the extracted fat after saponification process the recovered fat was evaluated whose value is lying between 84.40 g to 86.60 g/kg. In the present study the yield of fat revealed to be 8% to 9%. A total amount of 520 g of fat was recovered by using 6 kg of chicken feather meal sample. Kondamudi, et al.; Purandaradas, et al., also reported the yield of extracted fat from chicken feather meal was 6.9 g and 218.98 mg respectively.
Optimization of Biodiesel Production
At the varying temperatures tested for biodiesel production the maximum yield of biodiesel was obtained at 60℃ and the yield was 70.4% (Figure 1a). Temperature beyond 60℃ showed a steady decrease in biodiesel production that could be because of higher reaction temperature will accelerate the saponification of triglycerol. Similarly, the optimum temperature of 50℃ and the ultimate yield of 72.8% were observed by Agbajelola, et al., in chicken fat. Moreira, et al., reviewed in most of the publications the poultry waste as a prime source towards biodiesel production have revealed that trans-esterification process cought be performed between 60°C to 70°C. The incubation time is a prime success in bioprocess technology. So as to determine the optimal harvesting time for biodiesel production different incubation periods were examined. In the present study the highest biodiesel yield (70.0%) has been obtained at 120 minutes of incubation (Figure 1b). The yield of biodiesel was lower at the beginning because the reaction proceeds slowly in the initial stage due to the mixing and dispersion of alcohol into the fat as stated by Freedman, et al. Then the reaction proceeds very fast until the attainment of maximum yield. Similar to the present study, earlier Patil and Deng reported the maximum amount of biodiesel production at 120 minutes of reaction time and the yield was 95% from non-edible Jatropha curcas oil.
The effect of KOH concentration was studied in the range of 0.5%-2.5% whereby the other parameters remains constant. This is evident from the study that the KOH concentration influenced the biodiesel yield in a positive manner only up to the level of 1.0% with the maximum FAME yield of 73.2% (Figure 1c). Also, it is inferred that beyond this concentration the yield of biodiesel was reduced with increase in KOH concentration. Kondamudi, et al., has a similar view and reported that 1% concentration of KOH catalyst gave maximum yield of biodiesel from chicken feather meal and chicken fat respectively. The production of biodiesel is vitally enhanced by molar ratio of alcohol. This study revealed that the molar ratio of methanol to fat influenced the biodiesel yield in a positive manner only up to the ratio of 6:1 with the highest biodiesel yield of 72.8% beyond this ratio the biodiesel yield decreased with increase in molar ratio of methanol to fat concentration (Figure 1d). Similar to the present study, earlier Nuhu, et al., reported the molar ratio of methanol to fat oil of 6:1 is optimum to convert chicken fat to biodiesel and the yield was 93%. The results revealed that the optimum condition for maximum yield of biodiesel (74.4%) was found to be catalyst concentration of 1% w/v, reaction temperature of 60℃, reaction time of 120 minutes and molar ratio of methanol to fat is 6:1. Nagaraja, et al., observed the maximum CFME yield of 94.76% at 35°C reaction temperature, 9:1 molar ratio of chicken fat to methanol, reaction time of 45 min and 1% w/v potassium hydroxide catalyst.
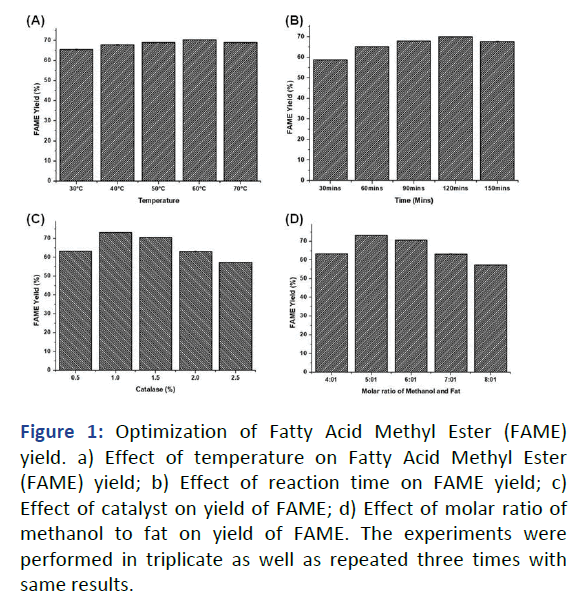
Figure 1: Optimization of Fatty Acid Methyl Ester (FAME) yield. a) Effect of temperature on Fatty Acid Methyl Ester (FAME) yield; b) Effect of reaction time on FAME yield; c) Effect of catalyst on yield of FAME; d) Effect of molar ratio of methanol to fat on yield of FAME. The experiments were performed in triplicate as well as repeated three times with same results.
Characterization of Extracted Biodiesel
In the present study, the acid value of biodiesel was determined by volumetric titration method and it was 0.32 mg KOH g-1. Universal constraints and limits that the maximum value should be 0.50 mg KOH g-1 as per ASTM D6751 standard. This property impact aging of fuel and the low acid value of biodiesel revealed good quality of biodiesel. Alptekin, et al., reported that acid value of Chicken Fat Methyl Ester (CFME) was 0.30 mg KOH g-1 and 0.364 mg KOH g-1 respectively. The viscosity of biodiesel is inferred to be 4.98 mm2/S from the experimentation. As per “ASTM D6751 standard” the acceptable viscosity ranges for biodiesel from 3.5 mm2/s-5.9 mm2/s. The determination of kinematic viscosity is most important parameter for biodiesel because higher viscosity tends to negative influence on a locomotive fuel injection pump and will increase excessive exhaust smoke. Similarly, Amber, et al., observed values of kinematic viscosity ranging between 4.35 mm2/s to 4.65 mm2/s in chicken feather meal sample. The specific gravity of biodiesel was analyzed using specific gravity bottle at a room temperature of 30℃ and it was 879 kg/m3. As per ASTM standard the specific gravity of biodiesel value must be up to 900 kg/m3. The value of specific gravity of biodiesel was in the range between 874.5 kg/m3-881 kg/m3 as reported by Amber, et al., in the chicken feather meal sample. The specific gravity of FAME from poultry waste was found to be 868 kg/m3. In a pH meter, the pH value of biodiesel sample was experimented and it was pH 7.8 and as per ASTM standard the pH value of biodiesel should be pH 7.0. Abraham Peele, et al., assessed the pH value of biodiesel from chicken feather meal sample and it was between 7.23-8.0.
In the present study, the water and sedimentation test was carried in a biodiesel by centrifugation process and the corresponding value was 325 mg/kg. As per ASTM standard the value should be up to the level of 500 mg/kg. The biodiesel should be clear, bright in appearance and would be free from water and sediment because the excess amount of water will corrode the engine. Amber, et al., also found similar results in chicken feather meal sample and the corresponding value was 445 mg/kg. The obtained biodiesel was blended together with methanol and it resulted clear bright phase. This shows that the sample is fully dissolved in methanol. Ultimately, the emulsification test was carried over and the equal amount of biodiesel and water were blended. The biodiesel sample exhibited positive result and meets with an ASTM standard, that is biodiesel on top layer and water with no impurity at the bottom. This is because of the obtained biodiesel has been rinsed with HCL and warm water. Hence, it infers that the obtained biodiesel is a good quality. Visibly, the derived biodiesel was amber in color the Amber color of biodiesel is due to golden (or) transparent yellow color of chicken fat. Similar inferences were also reported by Amber, et al. Hence, it is to be noted that the assessed physiochemical properties of obtained biodiesel is comparatively matching all the properties as per the ASTM standard (Table 1).
| Properties | Unit | ASTM D6751 | Biodiesel from chicken feather meal |
|---|---|---|---|
| Acid value | mg KOH g-1 | 0.50 max | 0.32 |
| Kinematic viscosity at 40℃ | mm2/S | 3.5-5.9 | 4.98 |
| Specific gravity | kg/m3 | Up to 900 | 878 |
| pH | - | 7 | 7.8 |
| Water and sediment | mg/kg | Up to 500 | 325 |
| Methanol test | - | N/A | Biodiesel completely dissolved in methanol. |
| Emulsification | - | N/A | Biodiesel on a top and water at bottom. |
| Visibility | - | Amber | Amber |
Table 1: Characterization of biodiesel from chicken feather meal by ASTM standard values.
FTIR Analysis
FT-IR spectra exhibits bands between the regions 700 cm-1 to 2900 cm-1 reveals the possibilities of presence of all the required functional moieties of the diesel derived from the biological source. A clear stretching band at 1735 cm-1 is due to the presence of C=O stretching infers the presence of either of the functional group ester (or) carbonyl groups in the derived biodiesel. A strong bond at 2922 cm-1 reveals the presence of aromatic C-H stretching in the compound and cluster of bands in the finger print region between 900 cm-1 to 1400 cm-1 is due to overtones and combination bands of CH chemical moiety in the sample. A strong band at 1460 may be due to the presence of C-O-C (ether moiety) in the derived sample. The above said analytical data coincide with the literature data confirm the derived sample could be a biodiesel (Figure 2). Sabrina Rabelo, et al., also observed the FT-IR spectra inferred that the region from 1800 cm-1-1700 cm-1; shall be observed peaks that can be attributed to the stretching of C=O, typical of esters. The region from 1500 cm-1-900 cm-1 known as fingerprint region. The peak at 1446 cm-1 correspond to the asymmetric stretching of -CH3. The stretching of O-CH3 represented by the absorbance at 1196 cm-1 is typical of biodiesel.
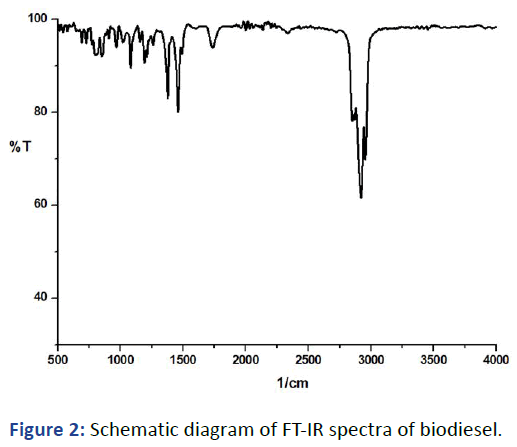
Figure 2: Schematic diagram of FT-IR spectra of biodiesel.
Biodiesel has become alternative fuel due to its environmental assistances and easily accessible renewable resources. In this present study, fat from chicken feather meal which is a waste ingredient of poultry farm that provides us a low cost feed stock to produce biodiesel and this will spare as major renewable fuel resource in forth coming era. As fine as the eminence of obtained samples were estimated according to FTIR and also determined the physical properties of biodiesel which was accessible that all the measured parameters results were in suitable range.
Authors are thankful to the department of biochemistry, Vivekanandha college of arts and sciences for women for providing necessary facilities to carry out this research work.
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Duraipandian M, Prabhu N, Kowsalya R (2023) Utilization of Chicken Feather Meal for Effective Production Economical Biodiesel and Its Characterization Studies. Adv Appl Sci Res. 14:42.
Copyright: © 2023 Duraipandian M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.